Abstract
The hazardous compound di-2-ethylhexyl phthalate (DEHP) is widely used as polyvinyl chloride plasticizer. The present research studied the fungal biodegradation of DEHP contained in blood and urine bags. Soil-plate method was used for fungal isolation from heavily plastic polluted soil using Martin’s and Sabouraud’s agar media, where DEHP was the sole carbon source. Isolated fungal species were identified morphologically according to Moubasher (Soil Fungi in Qatar and Arab Countries University of Qatar, Qatar The Center for Scientific and Research, 1993) as Aspergillus nidulans, Aspergillus niger and Rhizopus nigricans. DEHP concentrations were determined in 1 g of soil, urine bags and blood bags to be 0.92, 2.5 and 2.6 g/l, respectively. Samples of both bags (as a sole carbon source) were artificially inoculated with the isolated fungi and incubated for 20 days. As the time increased, the growth increased where Rhizopus nigricans obtained the highest dry weight in urine bags after 20 days of incubation, while A. nidulans had the highest dry weight in blood bags. Also, a sharp declining of initial pH (6.8) reached 4.7 in urine bags with A. niger growth, while reached 2.5 after A. nidulans growth in blood bags. DEHP% decreased as time increased indicating a continuous DEHP utilization by the three fungal species. Aspergillus niger was the most DEHP degrading fungal species in both bags. Scanning electron microscope examination showed an uniform plastic network in both bags before fungal treatment. While, a microporous network was observed on the plastic surfaces in both bags after fungal treatment due to DEHP utilization. The most DEHP metabolizing fungal species were further identified molecularly using internal transcribed spacer primers to be Aspergillus niger and Aspergillus nidulans with accession numbers MZ832174 and MT919276, respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
A wide variety of medicinal devices (MDs) used in hospitals are made of flexible plasticized polyvinyl chloride (PVC). Polyvinyl chloride is a polymer in which chlorine resembles more than 50% of its content. However due to the low cost and well performance of di-2-ethylhexyl phthalate (DEHP) and other phthalate plasticizers they are still commonly used in several applications. DEHP is used to soften the rigid PVC and increase its flexibility and workability but it is not chemically bound in plastic materials and they can be leaked into the environment easily (Fierens et al. 2012; Pradeep and Benjamin 2012). Plasticizers are added to PVC to make the plastics flexible, which are the most required properties for medicinal products (Koch et al. 2003; Zhang et al. 2009). Harmful effect of using phthalates as plasticizers in PVC is that it acts as simulators for the performance of human natural hormone system. Inoue et al. (2003) and Vesterberg et al. (2005) stated that DEHP is not chemically bound to the PVC polymer; it may be leaked when PVC medicinal devices come into contact with blood, urine and drugs. They also showed that DEHP from medicinal tubing is leaked into drug solutions in different quantities. DEHP is a developmental and reproductive poison in animals and supposed to be endocrine modulator in humans. DEHP may transfer from PVC storage bags to blood and blood products such as plasma, platelets and red blood cells (RBCs). Leaching of DEHP and mono-2-ethylhexyl phthalate (MEHP) into RBC units depends on the storage period (Münch et al. 2020). DEHP is hydrolyzed quickly into MEHP in human. For this reason, both DEHP (leaked from blood bags) and MEHP levels in blood products should be measured. DEHP pollution originating in several laboratory products, room air and plastic tubing may pose the contamination risk and therefore an over-estimation of DEHP levels (Inoue et al. 2005). In general population, a widespread exposure to DEHP is found. Toxic kinetic and metabolic information on DEHP in humans are very important not only for the toxicological assessment of DEHP but also for exposure assessments based on human bio monitoring information (Koch et al. 2006). Since transformation of DEHP to MEHP takes place firstly in the intestinal tract, ingestion of DEHP may be more dangerous than exposure intravenously. A number of studies displayed that DEHP causes liver cancer in laboratory animals (Gonzalez et al. 1998). It can be occurred through peroxisome proliferation induction, leading to oxidative stress and production of electrophilic free radicals, which leading to DNA damage indirectly (Silva et al. 2006). This involves the propagation of cellular organelles that contain enzymes showing catalytic activity toward fatty acids oxidation and the cholesterol biosynthesis in the liver (Dzehekova-stojkova et al. 2001; Reddy and Lalwai 1983).
Common techniques for degradation of plastic materials are incineration, land filling, hydrolysis, photodecomposition, recycling, and biodegradation (Hopewell et al. 2009; Savinova et al. 2022). Hydrolysis pathway chiefly occurs in aquatic environment but sometimes, no complete degradation of PAEs occurs. Photodecomposition is very slow (the half-life ranges from many years to many centuries) (Savinova et al. 2022). Land filling and incineration techniques need huge amount of space and are harmful to the environment. However, recycling technique is an effective method making plastic can be reused. Nevertheless, biodegradation is an environmental friendly technique that degrades a large number of pollutants using different microbial enzymes in a relatively short time (Purnomo et al. 2013; Yastrebova et al. 2019). The DEHP metabolic breakdown by microorganisms is considered to be one of the main techniques of ecological decomposition for this widespread contaminant due to its low rate of photolysis and hydrolysis. Plastics are stated to be decomposed in about 1000 years under the natural environment (Sangale et al. 2012). This slow rate of plastic wastes natural deterioration performs environmental threatens. Among the different common techniques to manage with the plastic wastes, biodeterioration technique by algae, actinomycetes, bacteria and fungi is considered as the most established and ecofriendly technique (Rutkowska et al. 2002). Biodeterioration of plastic wastes leads to complete degradation of the plastic into nonpoisonous materials as water, CH4, CO2, and other biological materials. Agaricus bisporus, Aspergillus flavus, Aspergillus glaucus, Aspergillus nidulans, Aspergillus nomius, Aspergillus oryzae, Bjerkandera adusta, Cladosporium cladosporioides, Penicillium griseofulvum, Phanerochaete chrysosporium, Pleurotus abalones, Pleurotus eryngii, Pleurotus ostreatus, etc., displayed efficient remediation of plastics (Srikanth et al. 2022).
This study aimed at isolation and identification of fungi from heavily plastic polluted soil and investigating their activity in degradation of DEHP plasticizer in both blood and urine bags.
2 Materials and Methods
2.1 Collection of Soil Samples
Soil samples were collected randomly from different sites of heavily plastic polluted areas (10 cm depth) in Garbage dumps in Fifth assembly.
2.2 Extraction and Determination of DEHP Concentration in Soil, Urine and Blood Bags
One gram of soil and plastic pieces (urine or blood bags) was immersed in n-hexane and left for two days then the concentration of DEHP was determined using spectrophotometer at wavelength 275 nm with standard curve of commercial DEHP (Pradeep and Benjamin 2012).
2.3 Isolation of DEHP Degrading Fungi from Plastic Polluted Soil by Soil-Plate Method
Soil-plate method was used to isolate DEHP degrading fungi from plastic polluted soil. Two isolation media (Martin’s and Sabouraud’s media) were used, where filtered-sterilized DEHP was substituted carbon source in these media. Sabouruad’s medium is composed of (g\L): glucose 40, peptone 10 and agar 18; while martin’s medium composed of (g\L): glucose 10, peptone 5, KH2PO4 1, MgSo4.7H2O 0.5 and agar 18. Soil samples (1 g) were added into sterilized petri dishes then media were poured and incubated at 27 °C for five days. Pure cultures of the growing fungi were obtained by repeated sub-culturing then maintained on the same media in refrigerator.
2.4 Morphological Identification of Isolated Fungi
The isolated fungal species were identified morphologically and microscopically according to Moubasher (1993). Cultures of 4 days old were examined for the phenotypic properties as shapes and colors of colonies, and then light microscope (5, 10 & 40X) was used for the examination of conidiophores and spores.
2.5 Artificial Inoculation of Urine and Blood Bags Pieces with Isolated Fungi
One gram of plastic pieces from blood and urine bags were putted in 50 ml of liquid martin media (pH 6.8) then inoculated with the fungal species isolated from polluted soil and incubated at 27 °C for 20 days.
2.6 Determination of Fungal Dry Biomass, pH of the Media and DEHP Percentage
Dry biomasses of fungi were determined at five days’ intervals after drying in oven until constant weight. The pH values of the media were also measured using pH meter. Furthermore, the percentages of DEHP were determined. Plastic pieces were washed with distilled water and left to dry then moved into n-hexane for two days. Then, the percentage of DEHP was determined by using spectrophotometer at wavelength 275 nm.
2.7 Examination of the Urine and Blood Bags Pieces Using the Scanning Electron Microscope (SEM)
The surface of the plastic pieces were examined before and after treatment with fungal species using scanning electron microscope (model: JSM-5500 LV; JEOL Ltd-japan) and high vacuum mode.
2.8 Molecular Identification of Fungi
The most active DEHP metabolizing fungal species were further identified by amplification and sequencing of internal transcribed spacer (ITS) region. Briefly, genomic DNA was isolated and purified using Wizard Genomic DNA purification Kit (Promega Corporation, USA) according to manufacture instructions. Polymerase chain reactions (PCR) were prepared to a final volume of 50 µl containing reaction buffer, 1.5 mM MgCl2, 200 µM of each dNTPase, 2.5 unit of Dream Taq DNA polymerase (Thermo Scientific, USA), 30 ng DNA template and 50 pmol of each primer (ITS1 and ITS4). An initial denaturation step for 5 min at 95 °C was followed by 30 cycles of denaturation at 94 °C for 40 s, annealing at 58 °C for 40 s and extension at 72 °C for 40 s, with a final extension at 72 °C for 10 min. The amplicons were purified using GeneJET™ PCR Purification Kit according the manufacture protocol. The nucleotide sequences of the purified PCR products were determined at macrogen (Seoul, South Korea). The sequences were analyzed by BLAST tool and were submitted to the GenBank. Phylogenetic trees in the context of ITS sequences were conducted. Accession numbers of the identified fungal species were also obtained.
3 Experimental Results
3.1 Determination of DEHP Concentration in Soil, Urine and Blood Bags
The concentration of DEHP in plastic polluted soil sample was found to be 0.92 g/l while it was found in urine bag sample to be 2.5 g/l, and in blood bags to be 2.6 g/l.
3.2 Isolation and Identification of DEHP Degrading Fungi from Plastic Polluted Soil
Only, three fungal species were isolated from the heavily plastic contaminated soil. They could metabolize DEHP and grow on media containing it as sole carbon source. They were identified morphologically as Aspergillus nidulans, Aspergillus niger and Rhizopus nigricans according to Moubasher (1993).
3.3 Determination of Fungal dry Biomass, pH of the Media and DEHP Utilization Percentage
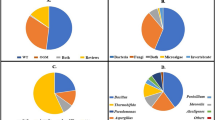
Assaying the ability of the isolated soil fungi to grow on plastic pieces declared that the three fungal species could grow well on the plastic pieces containing medium as a sole carbon source. The growth increased as the time period was increased to reach its maximum value after 20 days of incubation (Fig. 1). Rhizopus nigricans obtained the highest dry weight followed by Aspergillus nidulans then Aspergillus niger in urine bags. Whereas in blood bags A. nidulans had the highest dry weight followed by R. nigricans then A. niger.
The pH value of medium was altered by fungal growth on plastic pieces. The initial pH of the medium was 6.8. The value decreased to acidic side by increasing time with fungal growth. pH reached its minimum value after 20 days of A. niger incubation to be 4.7 in urine bags. In blood bags, pH was altered to 2.5 after 20 days of A. nidulans incubation (Fig. 2).
The concentration of DEHP extracted from plastic pieces of urine and blood bags was determined using UV spectrophotometer after growth of the three fungal species. The decline in DEHP extracted from both blood and urine bags gradually increased as the incubation period was increased with maximum decline after 20 days of incubation for the three fungal species (Fig. 3). In case of urine bags, A. niger was the most DEHP degrading fungal species followed by R. nigricans and A. nidulans with about the same efficacy. Also, A. niger was the most DEHP degrading fungal species followed by A. nidulans then R. nigricans in case of blood bags.
3.4 Examination of Blood and Urine Plastic Surface by Scanning Electron Microscope (SEM) Before and After Treatment with Fungi
Before fungal treatment, uniform plastic network was observed in both urine and blood bags. After fungal treatment, the metabolization of DEHP made a microporous network on the plastic surfaces of either urine or blood bag pieces. A. niger was the most efficient in utilizing DEHP followed by A. nidulans then R. nigricans in urine bags, while Aspergillus nidulans was the most effective in blood bags pieces (Figs. 4 and 5).
3.5 Molecular identification of fungi
The most DEHP degrading fungi (A. niger and A. nidulans) were further identified by amplification and sequencing of ITS region. BLAST result showed 100% similarity with that of A. niger which got accession number of MZ832174 (Fig. 6), and 100% similarity with that of A. nidulans which got accession number of MT919276 (Fig. 7).
4 Discussion
Bioremediation is the clean of the polluted environment at minimum cost and ecofriendly techniques using microorganisms. Plastic is used in many activities and it is a good replacement of other materials such as metals or paper and it can be retained for extended period with no deterioration. Plastic is also hydrophobic so it used in situations where water content requires to be avoided. Furthermore, plastic can be softenized by adding specific materials to it such as plasticizers (Azubuike et al. 2016; Liang et al. 2008). These plasticizers can transfer from the PVC bags into infused solutions and thus come into contact with the patient (Niino et al. 2001, 2002). Some of these chemical compounds are harmful to the patient which can affect reproduction and fertility (Hauser and Calafat 2005). Other adverse effects of DEHP exposure in animal studies include inhibited or delayed ovulation, inhibited estradiol production, and produce polycystic ovaries. It also reduces kidney function, causes kidney atrophy, reduces liver function, causes respiratory distress, and decreases heart rate and blood pressure (Gonzalez et al. 1998; Silva et al. 2006). Phthalate esters (PEs) are the most commonly used plasticizers worldwide. Concerns about toxicity of PEs were found because PEs can act as endocrine disrupting chemicals which affect development and reproduction. A talented approach of PEs remediation is their biotransformation by fungal enzymes. White rot fungi are able to degrade phthalates. The efficacy of the biotransformation most probably depends on the enzymatic system of a particular species (Fedorova et al. 2021). However, there are many problems concerning their deterioration, where themanufacture rate of plastic is not balanced with the decomposition rate throughout the world (Shahnawaz et al. 2019).
In the present study, bioremediation of plastic containing the plasticizer DEHP by three fungal species were investigated. Aspergillus nidulans, Aspergillus niger and Rhizopus nigricans isolated from heavily plastic polluted soil samples were found as good utilizers of DEHP. In relation to our study, Ekanayaka et al. (2022) stated that fungal species which degrade plastic are present in 11 classes belonging to the phyla Ascomycota, Basidiomycota and Mucoromycota. Srikanth et al. (2022) reported that Aspergillus nidulans, Aspergillus flavus, Aspergillus oryzae, etc., can grow on plastics causing degradation of them.
The results of this study indicated that the dry biomass of the studied fungal species increased gradually with increasing the incubation period when grown on media containing DEHP as sole carbon source. However, the pH values of the growth media were altered to acidic side and the DEHP% decreased by increasing the incubation time indicating a continuous utilization of DEHP by the fungal species. The results of this study are compatible with earlier studies. Fu and Xiang (2020) studied the influence of Aspergillus niger, Penicillium and yeast on DEHP degradation by single factor test. They found that the optimum conditions for degradation of DEHP were at pH 5.0 and 30 °C. These fungi had the ability to degrade DEHP, where Aspergillus niger was the most able fungus to degrade DEHP (68.75% of degradation within 60 h). Aguilar-Alvarado et al. (2015) demonstrated that fungal species isolated from recycled paper wastes in a paper manufacturing were able to utilize DEHP as the sole carbon source. These fungal isolates can be used for DEHP biodegradation. Savinova et al. (2022) reported that PAEs degradation is depending on fungal enzymes. They also stated that DEHP was degraded by some fungi (Trametes hirsute and Steccherinum ochraceum) with an efficacy of > 99%. Johnson (2014) studied an experiment to determine the effect of fungal growth on the phthalate ester content of the pPVC used as the fronting material of metal building insulation. He showed that the mean content of all tested phthalate esters was decreased in case of fungal growth as compared to pieces of no fungal growth. He found that DEHP had a mean mass content of 319 ng in the pPVC without fungal growth and 278 ng in the pPVC with fungal growth (i.e., 10% decline in phthalate ester mass between the pPVC without fungal growth and pPVC with fungal growth).
In the current study, SEM images showed changes in plastic surface structure. A microporous plastic surface was formed by the decomposition of the urine and blood bags plastic pieces by the tested fungal species. Adamcová et al. (2018) studied the degradation of plastics using SEM analysis. SEM is used for examination of changes occurs in materials morphology at microscale. SEM images proved the microbial activity on bioplastic by formation of cracks and loss of surface structure smoothness.
5 Conclusion
This study obviously demonstrates that fungal species (Aspergillus nidulans, Aspergillus niger and Rhizopus nigricans) isolated from heavily plastic polluted soil can grow on and degrade the plasticizer hazardous compound DEHP contained in blood and urine bags with an efficient degree.
References
Adamcová D, Radziemska M, Zloch J, Dvořáčková H, Elbl J, Kynický J, Brtnicky M, Vaverková MD (2018) SEM analysis and degradation behavior of conventional and bio-based plastics during composting. Acta Univ Agric Silvic Mendel Brun 66(2):349–356. https://doi.org/10.11118/actaun201866020349
Aguilar-Alvarado Y, Báez-Sánchez MdR, Martínez-Carrera DC, Ahuactzin-Pérez M, Cuamatzi-Muñoz M, Sánchez C (2015) Mycelial growth and enzymatic activities of fungi isolated from recycled paper wastes grown on di (2-ethylhexyl) phthalate. Pol J Environ Stud 24(5):1897–1902
Azubuike CC, Chikere CB, Okpokwasili GC (2016) Bioremediation techniques-classification based on site of application: principles, advantages, limitations and prospects. World J Microbial Biotechnol 32:180
Dzehekova-Stojkova S, Bogdanska J, Stojkova Z (2001) Peroxisome proliferators: their biological and toxicological effects. Clin Chem Lab Med 39(6):468–474. https://doi.org/10.1515/CCLM.2001.076
Ekanayaka AH, Tibpromma S, Dai D, Xu R, Suwannarach N, Stephenson SL, Dao C, Karunarathna SC (2022) A review of the fungi that degrade plastic. J Fungi 8(8):772. https://doi.org/10.3390/jof8080772
Fedorova TV, Moiseenko KV, Savinova OS, Glazunova OA, Eremin SA (2021) Biotransformation of phthalates by basidiomycete fungi from different ecophysiological groups. Public Health Toxicol 1(Supplement 1):A1. https://doi.org/10.18332/pht/142008
Fierens T, Servaes K, Van Holerbeke M, Geerts L, De Henauw S, Sioen I, Vanermen G (2012) Analysis of phthalates in food products and packaging materials sold on the Belgian market. Food Chem Toxicol 50:2575–2583
Fu X, Xiang M (2020) Study on the biodegradation of di-ethyl hexyl phthalate (DEHP) by different microorganisms. J Biotech Res 11:133–140
Gonzalez F, Peters M, Cattley R (1998) Mechanism of action of the nongenotoxic peroxisome proliferators: Role of the peroxisome proliferator-activated receptor α. J Natl Cancer I 90:1702–1709
Hauser R, Calafat AM (2005) Phthalates and human health. Occup Environ Med 62(11):806–818. https://doi.org/10.1136/oem.2004.017590
Hopewell J, Dvorak R, Kosior E (2009) Plastics recycling: challenges and opportunities. Philos Trans R Soc Lond B Biol Sci 364(1526):2115–2126. https://doi.org/10.1098/rstb.2008.0311
Inoue K, Higuchi T, Okada F, Iguchi H, Yoshimura Y, Sato A et al (2003) The validation of column-switching LC/MS as a high throughput approach for direct analysis of di (2-ethylhexyl) phthalate released from PVC medical devices in intravenous solution. J Pharm Biomed Anal 31:1145–1152
Inoue K, Kawaguchi M, Yamanaka R, Higuchi T, Ito R, Saito K, Nakazawa H (2005) Evaluation and analysis of exposure levels of di (2-ethylhexyl) phthalate from blood bags. Clin Chim Acta 358(1–2):159–166. https://doi.org/10.1016/j.cccn.2005.02.019
Johnson G (2014) The effect of fungal growth on potential phthalate ester exposures from plasticized polyvinyl chloride. Graduate Theses and Dissertations. https://scholarcommons.usf.edu/etd/5048
Koch HM, Rossbach B, Drexler H, Angerer J (2003) Internal exposure of the general population to DEHP and other phthalates–determination of secondary and primary phthalate monoester metabolites in urine. Environ Res 93(2):177–185
Koch HM, Preuss R, Angerer J (2006) Di (2ethylhexyl) phthalate (DEHP): human metabolism and internal exposure - an update and latest results. Int J Androl 29:155–165
Liang D-W, Zhang T, Fang HHP, He J (2008) Phthalates biodegradation in the environment. Appl Microbiol Biotechnol 80:183–198
Moubasher AH (1993) Soil Fungi in Qutar and Arab Countries. University of Qatar, Qatar, The Center for Scientific and Research
Münch F, Göen T, Zimmermann R, Adler W, Purbojo A, Höllerer C, Cesnjevar RA, Rüffer A (2020) Reduction of exposure to plasticizers in stored red blood cell units. Perfusion 35(1):32–38
Niino T, Ishibashi T, Itoh T, Sakai S, Ishiwata H, Yamada T, Onodera S (2001) Monoester formation by hydrolysis of dialkyl phthalate migrating from polyvinyl chloride products in human saliva. J Health Sci 47:318–322
Niino T, Ishibashi T, Itoh T, Sakai S, Ishiwata H, Yamada T, Onodera S (2002) Comparison of diisononyl phthalate migration from polyvinyl chloride products into human saliva in vivo and into saliva stimulant in vitro. J Health Sci 48:277–281
Pradeep S, Benjamin S (2012) Mycelia fungi completely remediate di (2-ethylhexyl) phthalate, the hazardous plasticizer in PVC blood storage bag. J Hazard Mater 235–236:69–77
Purnomo AS, Mori T, Putra SR, Kondo R (2013) Biotransformation of heptachlor and heptachlor epoxide by white-rot fungus Pleurotus ostreatus. Int Biodeterior Biodegrad 82:40–44
Reddy JK, Lalwai ND (1983) Carcinogenesis by hepatic peroxisome proliferators: Evaluation of the risk of hypolipidemic drugs and industrial plasticizers to humans. CRC Crit Rev Toxicol 12(1):1–58. https://doi.org/10.3109/10408448309029317
Rutkowska M, Heimowska A, Krasowska K, Janik H (2002) Biodegradability of polyethylene starch blends in sea water. Polish J Environ Stud 11:267–272
Sangale MK, Shahnawaz M, Ade AB (2012) A review on biodegradation of polythene: the microbial approach. J Bioremed Biodeg 3:1–9. https://doi.org/10.4172/2155-6199.1000164
Savinova OS, Shabaev AV, Glazunova OA, Eremin SA, Fedorova TV (2022) Biodestruction of phthalic acid esters by white rot fungi. Appl Biochem Microbiol 58(5):598–612. https://doi.org/10.1134/S0003683822050143
Shahnawaz M, Sangale MK, Ade AB (2019) Bioremediation technology for plastic waste. Springer Singapore. https://doi.org/10.1007/978-981-13-7492-0
Silva MJ, Samandar E, Preau JL Jr, Needham LL, Calafat AM (2006) Urinary oxidative metabolites of di(2ethylhexyl) phthalate in humans. Toxicology 219:22–32. https://doi.org/10.1016/j.tox.2005.10.018
Srikanth M, Sandeep TSRS, Sucharitha K, Godi S (2022) Biodegradation of plastic polymers by fungi: a brief review. Bioresour Bioprocess 9:42. https://doi.org/10.1186/s40643-022-00532-4
Vesterberg A, Hedenmark M, Vass AM (2005) “PVC in Medical Devices,” in Final report Karolinska University Stockholm, www.karolinska.se
Yastrebova OV, Pyankova AA, Plotnikova EG (2019) Phthalate-degrading bacteria isolated from an industrial mining area and the processing of potassium and magnesium salts. Appl Biochem Microbiol 55:397–404. https://doi.org/10.1134/S000368381904015X
Zhang D, Lui H, Liang Y, Wang C, Liang H, Cai H (2009) Distribution of phthalate esters in the groundwater of Jianghan plain, Hubei. China Front Earth Sci China 3(1):73–79
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
EAMA and TMA A-R contributed to the study conception. Methodology, data collection and analysis were performed by A-SHG. Analyses of molecular results were performed by EAMA. The first draft of the manuscript was written by A-SHG. Reviewing and editing were performed by TMA A-R, EAMA and DMIA. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Human or animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ali, E.A.M., Abdel-Rahman, T.M.A., Ali, D.M.I. et al. Fungal Bioremediation of the Plasticizer Hazardous Compound di-2-Ethylhexyl Phthalate (DEHP) in Urine and Blood Bags. Iran J Sci 47, 673–682 (2023). https://doi.org/10.1007/s40995-023-01466-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-023-01466-y