Abstract
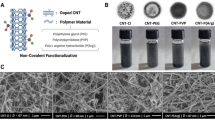
An advanced biosensor assembly based on glass fiber paper (GFP) electrodes modified with nanocomposites comprising of poly (3,4-ethylene dioxythiophene) polystyrene sulfonate (PEDOT: PSS) as the main matrix with functionalized gold nanoparticles (GFP/PEDOT: PSS-AuNP) for selective and sensitive detection of procalcitonin (PCT). PCT is a biomarker that is specifically used to detect bacterial infections, and it has received a lot of interest as a potential answer to the challenges with selecting when to use antibiotics. Field emission scanning electron microscopy, ultraviolet–visible spectroscopy and energy dispersive X-ray analysis characterization techniques were used to characterize the prepared PEDOT: PSS-AuNP nanocomposite. For the electrochemical characterization and response study, electrochemical impedance studies study have been presented because it is more sensitive and result accuracy can be obtained through it. This conductive paper-based biosensor exhibits improved detection 0.10 pg mL−1 performance with a linear detection range (1–15 pg mL−1), high sensitivity, and a 24-day life span for PCT antigen detection. In addition, the inclusion of AuNP in PEDOT: PSS resulted in improved electrochemical performance and signal stability. This PEDOT: PSS-AuNP@GFP platform based was used to immobilize anti-PCT antigenic protein for PCT quantification. The results of electrochemical response studies showed that this conductive paper bioelectrode had a good reproducibility sensitivity in physiological ranges (1–15 pg mL−1). Further, the proposed bioelectrode an alternative choice for point-of-care PCT detection has been shown.






Similar content being viewed by others
References
Abbaszadeh S, Nikaeen G, Yousefinejad S (2021) Carbon nanomaterials as promising substrates in the design of sensors for SARS-CoV-2 and new emerging viral infections. Nanomedicine 16(23):2033–2037. https://doi.org/10.2217/nnm-2021-0154
Al-Kazazz FFM, Al-Imarah KAF, Alhasnawi SI, Agelmashotjafar L, Abdul-Majeed BA (2013) A Simple method for synthesis, purification and concentration stabilized goldnanoparticles. Int J Eng Res Appl 3:21
Banerjee R, Jaiswal A (2018) Recent advances in nanoparticle-based lateral flow immunoassay as a point-of-care diagnostic tool for infectious agents and diseases. Analyst 143(9):1970–1996. https://doi.org/10.1039/c8an00307f
Boonkaew S, Jang I, Noviana E, Siangproh W, Chailapakul O, Henry CS (2021) "Electrochemical paper-based analytical device for multiplexed, point-of-care detection of cardiovascular disease biomarkers. Sens Actuators B Chem 330:129336. https://doi.org/10.1016/j.snb.2020.129336
Dong J, Carpinone PL, Pyrgiotakis G, Demokritou P, Moudgil BM (2020) Synthesis of precision gold nanoparticles using Turkevich method. Kona 37:224–232. https://doi.org/10.14356/kona.2020011
Gangopadhyay R, Das B, Molla MR (2014) How does PEDOT combine with PSS? Insights from structural studies. RSC Adv 4(83):43912–43920. https://doi.org/10.1039/C4RA08666J
Gao N, Yu J, Tian Q, Shi J, Zhang M, Chen S, Zang L (2021) Application of PEDOT: PSS and its composites in electrochemical and electronic chemosensors. Chemosensors. https://doi.org/10.3390/chemosensors9040079
Ge X-Y, Zhang J-X, Feng Y-G, Wang A-J, Mei L-P, Feng J-J (2022) Label-free electrochemical biosensor for determination of procalcitonin based on graphene-wrapped Co nanoparticles encapsulated in carbon nanobrushes coupled with AuPtCu nanodendrites. Microchim Acta 189(3):110. https://doi.org/10.1007/s00604-022-05179-8
Gupta Y, Ghrera AS (2021) Recent advances in gold nanoparticle-based lateral flow immunoassay for the detection of bacterial infection. Archi Microbiol 203(7):3767–3784. https://doi.org/10.1007/s00203-021-02357-9
Gupta Y, Ghrera AS (2022) Disposable paper-based biosensing platform for procalcitonin detection. Anal Bioanal Chem Res 10(1):15–23
Gupta Y, Kalpana K, Ghrera AS (2022a) Electrochemical studies of lateral flow assay test results for procalcitonin detection. J Electrochem Sci Technol 12(2):265–274. https://doi.org/10.5599/jese.1127
Gupta Y, Pandey CM, Ghrera AS (2022b) Reduced graphene oxide-gold nanoparticle nanohybrid modified cost-effective paper-based biosensor for procalcitonin detection. Chem Select 7(41):e202202642. https://doi.org/10.1002/slct.202202642
Huang Y, Kormakov S, He X, Gao X, Zheng X, Liu Y, Sun J, Wu D (2019) Conductive polymer composites from renewable resources: An overview of preparation, properties, and applications. Polymers. https://doi.org/10.3390/polym11020187
Jiang Y, Wu J (2019) Recent development in chitosan nanocomposites for surface-based biosensor applications. Electrophoresis 40(16–17):2084–2097. https://doi.org/10.1002/elps.201900066
Kariuki J, Ervin E, Olafson C (2015) Development of a novel, low-cost, disposable wooden pencil graphite electrode for use in the determination of antioxidants and other biological compounds. Sensors. https://doi.org/10.3390/s150818887
Kumar S, Sen A, Kumar S, Augustine S, Yadav BK, Mishra S, Malhotra BD (2016) Polyaniline modified flexible conducting paper for cancer detection. App Phy Lett 108(20):203702. https://doi.org/10.1063/1.4950961
Kumawat N, Soman SS, Vijayavenkataraman S, Kumar S (2022) Rapid and inexpensive process to fabricate paper based microfluidic devices using a cut and heat plastic lamination process. Lab Chip. https://doi.org/10.1039/D2LC00452F
Li H, Song S, Wen M, Bao T, Wu Z, Xiong H, Zhang X, Wen W, Wang S (2019) "A novel label-free electrochemical impedance aptasensor for highly sensitive detection of human interferon-gamma based on target-induced exonuclease inhibition. Biosens Bioelectrons 142:111532. https://doi.org/10.1016/j.bios.2019.111532
Li X, Qin Z, Fu H, Li T, Peng R, Li Z, Rini JM, Liu X (2021) Enhancing the performance of paper-based electrochemical impedance spectroscopy nanobiosensors: an experimental approach. Biosens Bioelectrons 177:112672. https://doi.org/10.1016/j.bios.2020.112672
Liu F, Xiang G, Yuan R, Chen X, Luo F, Jiang D, Huang S, Li Y, Pu X (2014) Procalcitonin sensitive detection based on graphene–gold nanocomposite film sensor platform and single-walled carbon nanohorns/hollow Pt chains complex as signal tags. Biosens Bioelectrons 60:210–217. https://doi.org/10.1016/j.bios.2014.03.071
Maity N, Dawn A (2020) Conducting polymer grafting: recent and key developments. Polymers. https://doi.org/10.3390/polym12030709
Moussa M, El-Kady MF, Dubal D, Tung TT, Nine MJ, Mohamed N, Kaner RB, Losic D (2020) Self-assembly and cross-linking of conducting polymers into 3D hydrogel electrodes for supercapacitor applications. ACS Appl Energy Mater 3(1):923–932. https://doi.org/10.1021/acsaem.9b02007
Namsheer K, Rout CS (2021) Conducting polymers: a comprehensive review on recent advances in synthesis, properties and applications. RSC Adv 11(10):5659–5697. https://doi.org/10.1039/D0RA07800J
Narang J, Malhotra N, Singhal C, Mathur A, Chakraborty D, Anil A, Ingle A, Pundir CS (2017) Point of care with microfluidic paper-based device integrated with nano zeolite–graphene oxide nanoflakes for electrochemical sensing of ketamine. Biosen Bioelectrons 88:249–257. https://doi.org/10.1016/j.bios.2016.08.043
Nie J, Zhang Y, Lin L, Zhou C, Li S, Zhang L, Li J (2012) Low-cost fabrication of paper-based microfluidic devices by one-step plotting. Anal Chem 84(15):6331–6335. https://doi.org/10.1021/ac203496c
Nie S, Li Z, Yao Y, Jin Y (2021) Progress in synthesis of conductive polymer poly(3,4-ethylenedioxythiophene). Front Chem 9:803509. https://doi.org/10.3389/fchem.2021.803509
Nikaeen G, Abbaszadeh S, Yousefinejad S (2020) Application of nanomaterials in treatment, anti-infection and detection of coronaviruses. Nanomedicine 15(15):1501–1512. https://doi.org/10.2217/nnm-2020-0117
Nishat S, Jafry AT, Martinez AW, Awan FR (2021) Paper-based microfluidics: simplified fabrication and assay methods. Sens Actuators B Chem 336:129681. https://doi.org/10.1016/j.snb.2021.129681
Olowu RA, Arotiba O, Mailu SN, Waryo TT, Baker P, Iwuoha E (2010) Electrochemical aptasensor for endocrine disrupting 17β-estradiol based on a poly(3,4-ethylenedioxylthiopene)-gold nanocomposite platform. Sensors 10(11):9872–9890. https://doi.org/10.3390/s101109872
Park IH, Lee SH, Yu ST, Oh YK (2014) Serum procalcitonin as a diagnostic marker of neonatal sepsis. Korean J Pediatr 57(10):451–456. https://doi.org/10.3345/kjp.2014.57.10.451
Ruecha N, Shin K, Chailapakul O, Rodthongkum N (2019) Label-free paper-based electrochemical impedance immunosensor for human interferon gamma detection. Sens Actuators B Chem 279:298–304. https://doi.org/10.1016/j.snb.2018.10.024
Sahu S, Dutta G (2021) Emerging evidence for serum procalcitonin estimation at point-of-care and advancement in quantitative sensing strategies over the past decade. Sens Int 2:100107. https://doi.org/10.1016/j.sintl.2021.100107
Samsudin I, Vasikaran SD (2017) Clinical utility and measurement of procalcitonin. Clin Biochem Rev 38(2):59–68
Singh V, Kumar T (2019) Study of modified PEDOT:PSS for tuning the optical properties of its conductive thin films. J Sci Adv Mater Dev 4(4):538–543. https://doi.org/10.1016/j.jsamd.2019.08.009
Siwal SS, Zhang Q, Devi N, Thakur VK (2020) Carbon-based polymer nanocomposite for high-performance energy storage applications. Polymers. https://doi.org/10.3390/polym12030505
Suchomel P, Kvitek L, Prucek R, Panacek A, Halder A, Vajda S, Zboril R (2018) Simple size-controlled synthesis of Au nanoparticles and their size-dependent catalytic activity. Sci Rep 8(1):4589. https://doi.org/10.1038/s41598-018-22976-5
Tanak AS, Jagannath B, Tamrakar Y, Muthukumar S, Prasad S (2019) Non-faradaic electrochemical impedimetric profiling of procalcitonin and C-reactive protein as a dual marker biosensor for early sepsis detection. Anal Chim Acta X 3:100029. https://doi.org/10.1016/j.acax.2019.100029
Tang RH, Liu LN, Zhang SF, He XC, Li XJ, Xu F, Ni YH, Li F (2019) A review on advances in methods for modification of paper supports for use in point-of-care testing. Microchim Acta 186(8):521. https://doi.org/10.1007/s00604-019-3626-z
Turkevich J, Stevenson PC, Hillier J (1951) A study of the nucleation and growth processes in the synthesis of colloidal gold. Dis Faraday Soc 11:55–75. https://doi.org/10.1039/DF9511100055
Wang M, Tang X-H, Cai J-H, Wu H, Shen J-B, Guo S-Y (2021) Construction, mechanism and prospective of conductive polymer composites with multiple interfaces for electromagnetic interference shielding: a review. Carbon 177:377–402. https://doi.org/10.1016/j.carbon.2021.02.047
Wen Y, Xu J (2017) Scientific importance of water-processable PEDOT–PSS and preparation, challenge and new application in sensors of Its film electrode: a review. J Polym Sci Part A Polym Chem 55(7):1121–1150. https://doi.org/10.1002/pola.28482
Xiao R, Cho SI, Liu R, Lee SB (2007) Controlled electrochemical synthesis of conductive polymer nanotube structures. J Am Chem Soc 129(14):4483–4489. https://doi.org/10.1021/ja068924v
Xu XX, Lei XL, Ye LY, Song SS, Liu LL, Xu LG, Xu CL, Kuang H (2022) Gold-based paper sensor for sensitive detection of procalcitonin in clinical samples. Chin J Anal Chem 50(4):100062. https://doi.org/10.1016/j.cjac.2022.100062
Yang L, Xue J, Jia Y, Zhang Y, Wu D, Ma H, Wei Q, Ju H (2019) Construction of well-ordered electrochemiluminescence sensing interface using peptide-based specific antibody immobilizer and N-(aminobutyl)-N-(ethylisoluminol) functionalized ferritin as signal indicator for procalcitonin analysis. Biosens Bioelectrons 142:111562. https://doi.org/10.1016/j.bios.2019.111562
Zhao J-J, Lou X-L, Chen H-W, Zhu F-T, Hou Y-Q (2018) Diagnostic value of decoy receptor 3 combined with procalcitonin and soluble urokinase-type plasminogen activator receptor for sepsis. Cell Mol Biol Lett 23(1):22. https://doi.org/10.1186/s11658-018-0087-z
Acknowledgements
A.S. Ghrera thanks financial support received from Science and Engineering Board (DST), India under the Young Scientist project (YSS/2015/001330). The authors wish to thank and acknowledge AIRF- JNU for the TEM characterization and UV -Vis, FESEM characterization from MJU, Jaipur.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gupta, Y., Ghrera, A.S. Conducting Polymer Modified Glass Fiber Paper Substrate Based Biosensor for Procalcitonin Detection. Iran J Sci 47, 327–335 (2023). https://doi.org/10.1007/s40995-023-01432-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-023-01432-8