Abstract
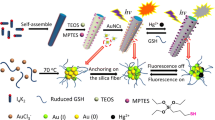
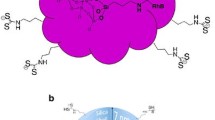
Au nanoparticle (Au NP)@SiO2@TDA-Eu nanocomposites were prepared by a two-step process: Au NP@SiO2 nanocomposites were prepared by a modified onepot process. Then the europium coordination polymer was deposited on the surface of the Au NP@SiO2 by mixing 2,2’-thiodiacetic acid [S(CH2COO)22-, TDA] and Eu(NO3)3·6H2O in ethanol via a hydrothermal method. The maximum fluorescent enhancement factor of the nanocomposites was 6.81 at 30 nm thickness of silica between the core of the Au NP and the shell of TDA-Eu. The prepared nanocomposites exhibit more sensitive monitoring of reactive oxygen species.
摘要
本论文通过两步工艺成功合成了Au NP@SiO2@TDA-Eu复合纳米材料:采用改进的一锅法制备了纳米复合材料Au NP@SiO2,然后 利用水热法, 在无水乙醇中通过亚硫基二乙酸[S(CH2COO)22-, TDA]和EUNO3)3·6H2O反应, 生成铕的配位聚合物, 并将其沉积在Au NP@SiO2的表面合成复合纳米材料Au NP@SiO2@TDA-Eu.该复合材料在二氧化硅层厚度为30 nm时, 荧光增强最大, 增强因子为6.81,在 对活性氧的检测中,表现出高灵敏度.
Article PDF
Similar content being viewed by others
References
Fuller SJ, Wragg FPH, Nutter J, et al. Comparison of on-line and off-line methods to quantify reactive oxygen species (ROS) in atmospheric aerosols. Atmos Environ, 2014, 92: 97–103
Sameenoi Y, Koehler K, Shapiro J, et al. Microfluidic electrochemical sensor for on-line monitoring of aerosol oxidative activity. J Am Chem Soc, 2012, 134: 10562–10568
Meyer LV, Schönfeld F, Müller-Buschbaum K. Lanthanide based tuning of luminescence in MOFs and dense frameworks–from mono- and multimetal systems to sensors and films. Chem Commun, 2014, 50: 8093–8108
Wang X, Chang H, Xie J, et al. Recent developments in lanthanidebased luminescent probes. Coord Chem Rev, 2014, 273-274: 201–212
Syamchand SS, Sony G. Europium enabled luminescent nanoparticles for biomedical applications. J Lumin, 2015, 165: 190–215
Wang D, Wang R, Liu L, et al. Down-shifting luminescence of water soluble NaYF4:Eu3+@Ag core-shell nanocrystals for fluorescence turn-on detection of glucose. Sci China Mater, 2017, 60: 68–74
Bhunia A, Gotthardt MA, Yadav M, et al. Salen-based coordination polymers of manganese and the rare-earth elements: synthesis and catalytic aerobic epoxidation of olefins. Chem Eur J, 2013, 19: 1986–1995
Müller-Buschbaum K, Beuerle F, Feldmann C. MOF based luminescence tuning and chemical/physical sensing. Microporous Mesoporous Mater, 2015, 216: 171–199
Decadt R, Van Hecke K, Depla D, et al. Synthesis, crystal structures, and luminescence properties of carboxylate based rare-earth coordination polymers. Inorg Chem, 2012, 51: 11623–11634
Le Natur F, Calvez G, Daiguebonne C, et al. Coordination polymers based on heterohexanuclear rare earth complexes: toward independent luminescence brightness and color tuning. Inorg Chem, 2013, 52: 6720–6730
Hasegawa Y, Nakanishi T. Luminescent lanthanide coordination polymers for photonic applications. RSC Adv, 2015, 5: 338–353
Jankovic V, Yang YM, You J, et al. Active layer-incorporated, spectrally tuned Au/SiO2 core/shell nanorod-based light trapping for organic photovoltaics. ACS Nano, 2013, 7: 3815–3822
Chu Z, Yin C, Zhang S, et al. Surface plasmon enhanced drug efficacy using core–shell Au@SiO2 nanoparticle carrier. Nanoscale, 2013, 5: 3406–3411
Zhang Y, Jiang H, Wang X. Cytidine-stabilized gold nanocluster as a fluorescence turn-on and turn-off probe for dual functional detection of Ag+ and Hg2+. Anal Chem Acta, 2015, 870: 1–7
Acuna GP, Bucher M, Stein IH, et al. Distance dependence of single-fluorophore quenching by gold nanoparticles studied on DNA origami. ACS Nano, 2012, 6: 3189–3195
Abadeer NS, Brennan MR, Wilson WL, et al. Distance and plasmon wavelength dependent fluorescence of molecules bound to silica-coated gold nanorods. ACS Nano, 2014, 8: 8392–8406
Feng AL, You ML, Tian L, et al. Distance-dependent plasmonenhanced fluorescence of upconversion nanoparticles using polyelectrolyte multilayers as tunable spacers. Sci Rep, 2015, 5: 7779
Li M, Cushing SK, Wu N. Plasmon-enhanced optical sensors: a review. Analyst, 2015, 140: 386–406
Sun Y, Guo GZ, Liu Y, et al. Effects of noble metal nanoparticles on the luminescent properties of europium complex. Curr Nanosci, 2010, 6: 103–109
Ming T, Chen H, Jiang R, et al. Plasmon-controlled fluorescence: beyond the intensity enhancement. J Phys Chem Lett, 2012, 3: 191–202
Chen J, Zhang R, Han L, et al. One-pot synthesis of thermally stable gold@mesoporous silica core-shell nanospheres with catalytic activity. Nano Res, 2013, 6: 871–879
Li H, Kang J, Yang J, et al. Distance dependence of fluorescence enhancement in Au nanoparticle@mesoporous silica@europium complex. J Phys Chem C, 2016, 120: 16907–16912
Wang HS, Bao WJ, Ren SB, et al. Fluorescent sulfur-tagged europium(III) coordination polymers for monitoring reactive oxygen species. Anal Chem, 2015, 87: 6828–6833
Li Z, Wang L, Wang Z, et al. Modification of NaYF4:Yb,Er@SiO2 nanoparticles with gold nanocrystals for tunable green-to-red upconversion emissions. J Phys Chem C, 2011, 115: 3291–3296
Liz-Marzán LM, Giersig M, Mulvaney P. Synthesis of nanosized gold-silica core-shell particles. Langmuir, 1996, 12: 4329–4335
http://refractiveindex.info/
Zhang J, Fu Y, Lakowicz JR. Luminescent silica core/silver shell encapsulated with Eu(III) complex. J Phys Chem C, 2009, 113: 19404–19410
Saboktakin M, Ye X, Oh SJ, et al. Metal-enhanced upconversion luminescence tunable through metal nanoparticle–nanophosphor separation. ACS Nano, 2012, 6: 8758–8766
Deng W, Jin D, Drozdowicz-Tomsia K, et al. Ultrabright Eu-doped plasmonic Ag@SiO2 nanostructures: time-gated bioprobes with single particle sensitivity and negligible background. Adv Mater, 2011, 23: 4649–4654
Lakowicz JR. Radiative decay engineering 5: metal-enhanced fluorescence and plasmon emission. Anal Biochem, 2005, 337: 171–194
Park W, Lu D, Ahn S. Plasmon enhancement of luminescence upconversion. Chem Soc Rev, 2015, 44: 2940–2962
Gryczynski I, Malicka J, Gryczynski Z, et al. Radiative decay engineering 4. Experimental studies of surface plasmon-coupled directional emission. Anal Biochem, 2004, 324: 170–182
Zhang H, Li Y, Ivanov IA, et al. Plasmonic modulation of the upconversion fluorescence in NaYF4:Yb/Tm hexaplate nanocrystals using gold nanoparticles or nanoshells. Angew Chem, 2010, 122: 2927–2930
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (51702006 and 21501141), the Doctoral research project (ZK2017027) of Baoji University of Arts and Sciences, and the Education Commission of Shaanxi Province (2015JQ6223, 12JS114, 14JS092 and 17JS009).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Huiqin Li received her BSc degree from Shaanxi University of Science and Technology and PhD degree from Northwestern University. Now, she is a lecturer at Baoji University of Arts and Sciences. Her research interest focuses on the functional nanomaterials and luminescent materials.
Jianhui Yang received PhD degree from Changchun Institute of Applied Chemistry, Chinese Academy of Sciences in 2008. Then, he had his post-doc experience from the City University of New York, University of Florida, the University of Texas at San Antonio and the University of Paris. Currently, he is an associate professor at Northwestern University. His research interests include controllable synthesis, assembly and properties of nanomaterials.
Electronic supplementary material
40843_2017_9127_MOESM1_ESM.pdf
Au nanoparticle@silica@europium coordination polymer nanocomposites for enhanced fluorescence and more sensitive monitoring reactive oxygen species
Rights and permissions
About this article
Cite this article
Li, H., Yang, J., Deng, Q. et al. Au nanoparticle@silica@europium coordination polymer nanocomposites for enhanced fluorescence and more sensitive monitoring reactive oxygen species. Sci. China Mater. 61, 401–408 (2018). https://doi.org/10.1007/s40843-017-9127-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-017-9127-0