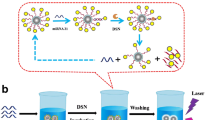
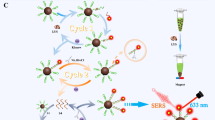
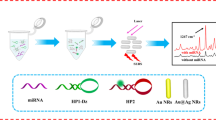
Abstract
Magnetic core gold shell nanostructures which integrate both SERS activity and superparamagnetism are widely utilized in SERS-based detection as SERS substrates, sample separation and preconcentration operators, as well as external magnetic field controlled directional carrier. However, most of the reported gold shells coated on the magnetic cores had smooth surfaces rather than branched nanostructures with enhanced SERS activity. Here, a novel type of Fe3O4-Au core-shell nanoparticles with branched gold shell was prepared by a seed-mediated method together with the shape induction agent AgNO3, and their growth process and mechanism, properties, as well as morphologically controlled synthesis were also investigated. The branched gold coated magnetic nanoparticles (B-GMNPs) with improved SERS performance were further utilized to build superparamagnetic and SERS-active capturers by assembling tetrahedral DNA onto their surfaces for sandwich-structured detection of cancer-related biomarker miRNA-21. The experimental results indicate that highly sensitive and specific detections can be obtained by the proposed SERS sensing system including B-GMNPs and tetrahedral DNA, and the limit of detection (LOD) of miRNA-21 in serum is 623 amol L−1. These B-GMNPs can be used as good SERS substrates with the functions of external magnetic field controlled sample separation and directional enrichment for effective SERS-based biochemical sensing and detections.
摘要
复合SERS活性和超顺磁性的磁核金壳纳米颗粒在SERS传感、样品分离与富集、定向输运等方面已有广泛应用. 已报道的基于磁 核制备的金纳米壳大多为平滑的金壳, 基于磁核生长枝杈状金壳可以提升颗粒的SERS增强性能, 但相关报道甚少. 本文基于种子生长法 并引入定向生长控制剂AgNO3, 合成了一种新型的磁核枝杈状金壳纳米颗粒, 系统地研究了金壳的生长过程与机制、表面形貌的控制生 长以及对SERS性能的影响. 此外, 设计并构建了具有良好的结构刚性、稳定性和空间定位能力的四面体DNA探针, 并将其修饰到磁核枝 杈状金壳纳米颗粒表面构建得到检测基底. 通过在金纳米颗粒表面修饰单链DNA构建SERS探针, 利用碱基互补配对形成“检测基底-目标 核酸-SERS探针”三明治结构复合物, 借助外加磁场分离检测液中的复合物并富集后进行SERS检测, 实现了对于血清中肿瘤核酸标志物 miRNA-21的高灵敏、特异性检测, 检测限达到623 amol L−1.
Article PDF
Similar content being viewed by others
References
Yang D, Ma J, Zhang Q, et al. Polyelectrolyte-coated gold magnetic nanoparticles for immunoassay development: toward point of care diagnostics for syphilis screening. Anal Chem, 2013, 85: 6688–6695
Zheng J, Dong Y, Wang W, et al. In situ loading of gold nanoparticles on Fe3O4@SiO2 magnetic nanocomposites and their high catalytic activity. Nanoscale, 2013, 5: 4894–4901
Lu AH, Salabas EL, Schüth F. Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed, 2007, 46: 1222–1244
Espinosa A, Di Corato R, Kolosnjaj-Tabi J, et al. Duality of iron oxide nanoparticles in cancer therapy: amplification of heating efficiency bymagnetic hyperthermia and photothermal bimodal treatment. ACS Nano, 2016, 10: 2436–2446
Tintoré M, Mazzini S, Polito L, et al. Gold-coated superparamagnetic nanoparticles for single methyl discrimination in DNA aptamers. IJMS, 2015, 16: 27625–27639
Chuah K, Lai LMH, Goon IY, et al. Ultrasensitive electrochemical detection of prostate-specific antigen (PSA) using gold-coated magnetic nanoparticles as “dispersible electrodes”. Chem Commun, 2012, 48: 3503–3505
Qi D, Zhang H, Tang J, et al. Facile synthesis of mercaptophenylboronic acid-functionalized core-shell structure Fe3O4@C@Au magnetic microspheres for selective enrichment of glycopeptides and glycoproteins. J Phys Chem C, 2010, 114: 9221–9226
Guven B, Basaran-Akgul N, Temur E, et al. SERS-based sandwich immunoassay using antibody coated magnetic nanoparticles for Escherichia coli enumeration. Analyst, 2011, 136: 740–748
Sun M, Xu L, Ma W, et al. Hierarchical plasmonic nanorods and upconversion core-satellite nanoassemblies for multimodal imaging-guided combination phototherapy. Adv Mater, 2016, 28: 898–904
Wang Y, Yan B, Chen L. SERS tags: novel optical nanoprobes for bioanalysis. Chem Rev, 2013, 113: 1391–1428
Wang Y, Chen L, Liu P. Biocompatible triplex Ag@SiO2@mTiO2 core-shell nanoparticles for simultaneous fluorescence-SERS bimodal imaging and drug delivery. Chem Eur J, 2012, 18: 5935–5943
Wang M, Meng G, Huang Q, et al. CNTs-anchored egg shell membrane decorated with Ag-NPs as cheap but effective SERS substrates. Sci China Mater, 2015, 58: 198–203
Duan C, Ren B, Liu H, et al. Flexible SERS active detection from novel Ag nano-necklaces as highly reproducible and ultrasensitive tips. Sci China Mater, 2016, 59: 435–443
Zhen S, Wu T, Huang X, et al. Facile synthesis of gold nanoflowers as SERS substrates and their morphological transformation induced by iodide ions. Sci China Chem, 2016, 59: 1045–1050
Song C, Yang B, Yang Y, et al. SERS-based mercury ion detections: principles, strategies and recent advances. Sci China Chem, 2016, 59: 16–29
Xu Z, Hou Y, Sun S. Magnetic core/shell Fe3O4/Au and Fe3O4/Au/Ag nanoparticles with tunable plasmonic properties. J Am Chem Soc, 2007, 129: 8698–8699
Kouassi GK, Irudayaraj J. Magnetic and gold-coated magnetic nanoparticles as a DNA sensor. Anal Chem, 2006, 78: 3234–3241
Cho SJ, Shahin AM, Long GJ, et al. Magnetic and Mössbauer spectral study of core/shell structured Fe/Au nanoparticles. Chem Mater, 2006, 18: 960–967
Jin Y, Jia C, Huang SW, et al. Multifunctional nanoparticles as coupled contrast agents. Nat Commun, 2010, 1: 41–48
Hu Y, Meng L, Niu L, et al. Facile synthesis of superparamagnetic Fe3O4@polyphosphazene@Au shells for magnetic resonance imaging and photothermal therapy. ACS Appl Mater Interfaces, 2013, 5: 4586–4591
Goon IY, Lai LMH, Lim M, et al. Fabrication and dispersion of gold-shell-protected magnetite nanoparticles: systematic control using polyethyleneimine. Chem Mater, 2009, 21: 673–681
Song C, Min L, Zhou N, et al. Ultrasensitive detection of carcino-embryonic antigen by using novel flower-like gold nanoparticle SERS tags and SERS-active magnetic nanoparticles. RSC Adv, 2014, 4: 41666–41669
Moraes Silva S, Tavallaie R, Sandiford L, et al. Gold coated magnetic nanoparticles: from preparation to surface modification for analytical and biomedical applications. Chem Commun, 2016, 52: 7528–7540
Li F, Yu Z, Zhao L, et al. Synthesis and application of homogeneous Fe3O4 core/Au shell nanoparticles with strong SERS effect. RSC Adv, 2016, 6: 10352–10357
Tang X, Dong R, Yang L, et al. Fabrication of Au nanorod-coated Fe3O4 microspheres as SERS substrate for pesticide analysis by near-infrared excitation. J Raman Spectrosc, 2015, 46: 470–475
Chen J, Pang S, He L, et al. Highly sensitive and selective detection of nitrite ions using Fe3O4@SiO2/Aumagnetic nanoparticles by surface- enhanced Raman spectroscopy. Biosens Bioelectron, 2016, 85: 726–733
Mo AH, Landon PB, Gomez KS, et al. Magnetically-responsive silica-gold nanobowls for targeted delivery and SERS-based sensing. Nanoscale, 2016, 8: 11840–11850
Zhang H, Harpster MH, Wilson WC, et al. Surface-enhanced Raman scattering detection of DNAs derived from virus genomes using Au-coated paramagnetic nanoparticles. Langmuir, 2012, 28: 4030–4037
Sharma B, Frontiera RR, Henry AI, et al. SERS: materials, applications, and the future. Mater Today, 2012, 15: 16–25
Song CY, Zhou N, Yang BY, et al. Facile synthesis of hydrangea flower-like hierarchical gold nanostructures with tunable surface topographies for single-particle surface-enhanced Raman scattering. Nanoscale, 2015, 7: 17004–17011
Song CY, Yang BY, Chen WQ, et al. Gold nanoflowers with tunable sheet-like petals: facile synthesis, SERS performances and cell imaging. J Mater Chem B, 2016, 4: 7112–7118
Hu J, Zhang Y, Liu B, et al. Synthesis and properties of tadpoleshaped gold nanoparticles. J Am Chem Soc, 2004, 126: 9470–9471
Rodriguez-Lorenzo L, Alvarez-Puebla RA, de Abajo FJG, et al. Surface enhanced Raman scattering using star-shaped gold colloidal nanoparticles. J Phys Chem C, 2010, 114: 7336–7340
Bakr OM, Wunsch BH, Stellacci F. High-yield synthesis of multibranched urchin-like gold nanoparticles. Chem Mater, 2006, 18: 3297–3301
Mitchell N, Schlapak R, Kastner M, et al. A DNA nanostructure for the functional assembly of chemical groups with tunable stoichiometry and defined nanoscale geometry. Angew Chem Int Ed, 2009, 48: 525–527
Li Z, Zhao B, Wang D, et al. DNA nanostructure-based universal microarray platform for high-efficiency multiplex bioanalysis in biofluids. ACS Appl Mater Interfaces, 2014, 6: 17944–17953
Pei H, Lu N,Wen Y, et al. A DNA nanostructure-based biomolecular probe carrier platform for electrochemical biosensing. Adv Mater, 2010, 22: 4754–4758
Pei H, Liang L, Yao G, et al. Reconfigurable three-dimensional DNA nanostructures for the construction of intracellular logic sensors. Angew Chem, 2012, 124: 9154–9158
Wang S, Zhang L, Wan S, et al. Aptasensor with expanded nucleotide using DNA nanotetrahedra for electrochemical detection of cancerous exosomes. ACS Nano, 2017, 11: 3943–3949
Wen Y, Pei H, Shen Y, et al. DNA nanostructure-based interfacial engineering for PCR-free ultrasensitive electrochemical analysis of microRNA. Sci Rep, 2012, 2: 867
Chen X, Zhou G, Song P, et al. Ultrasensitive electrochemical detection of prostate-specific antigen by using antibodies anchored on a DNA nanostructural scaffold. Anal Chem, 2014, 86: 7337–7342
Song C, Min L, Zhou N, et al. Synthesis of novel gold mesoflowers as SERS tags for immunoassaywith improved sensitivity. ACSAppl Mater Interfaces, 2014, 6: 21842–21850
Michota A, Bukowska J. Surface-enhanced Raman scattering (SERS) of 4-mercaptobenzoic acid on silver and gold substrates. J Raman Spectrosc, 2003, 34: 21–25
Zhang X, Zhu Y, Yang X, et al. Multifunctional Fe3O4@TiO2@Au magnetic microspheres as recyclable substrates for surface-enhanced Raman scattering. Nanoscale, 2014, 6: 5971–5979
Shao M, Ning F, Zhao J, et al. Preparation of Fe3O4@SiO2@layered double hydroxide core-shell microspheres for magnetic separation of proteins. J Am Chem Soc, 2012, 134: 1071–1077
Jana NR, Gearheart L,Murphy CJ. Evidence for seed-mediated nucleation in the chemical reduction of gold salts to gold nanoparticles. Chem Mater, 2001, 13: 2313–2322
Hong JW, Lee SU, Lee YW, et al. Hexoctahedral Au nanocrystals with high-index facets and their optical and surface-enhanced Raman scattering properties. J Am Chem Soc, 2012, 134: 4565–4568
Personick ML, Langille MR, Zhang J, et al. Shape control of gold nanoparticles by silver underpotential deposition. Nano Lett, 2011, 11: 3394–3398
You H, Ji Y,Wang L, et al. Interface synthesis of gold mesocrystals with highly roughened surfaces for surface-enhanced Raman spectroscopy. J Mater Chem, 2012, 22: 1998–2006
You H, Ding C, Song X, et al. In situ studies of different growth modes of silver crystals induced by the concentration field in an aqueous solution. Cryst Eng Comm, 2011, 13: 4491–4495
Nikoobakht B, El-Sayed MA. Preparation and growth mechanism of gold nanorods (NRs) using seed-mediated growth method. Chem Mater, 2003, 15: 1957–1962
Grubisha DS, Lipert RJ, Park HY, et al. Femtomolar detection of prostate-specific antigen: an immunoassay based on surface-enhanced Raman scattering and immunogold labels. Anal Chem, 2003, 75: 5936–5943
Shrestha YK, Yan F. Determination of critical micelle concentration of cationic surfactants by surface-enhanced Raman scattering. RSC Adv, 2014, 4: 37274–37277
Wang Z, Zong S, Li W, et al. SERS-fluorescence joint spectral encoding using organic-metal-QD hybrid nanoparticles with a huge encoding capacity for high-throughput biodetection: putting theory into practice. J Am Chem Soc, 2012, 134: 2993–3000
Wang G, Lipert RJ, JainM, et al. Detection of the potential pancreatic cancermarkerMUC4in serumusing surface-enhancedRaman scattering. Anal Chem, 2011, 83: 2554–2561
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21475064), Sci-tech Support Plan of Jiangsu Province (BE2014719), Program for Changjiang Scholars and Innovative Research Team in University (IRT_15R37), the Research Innovation ProgramforCollegeGraduates of Jiangsu Province (SJZZ15_0107), the Scientific Research Foundation of Nanjing University of Posts and Telecommunications (NY215075), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (YX03001).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yanjun Yang is a graduate student at the Institute of Advanced Materials, Nanjing University of Posts and Telecommunications (NJUPT) under the supervision of Prof. Lianhui Wang and Prof. Chunyuan Song. She received her Bachelor’s degree from NJUPT in 2014. Her current research focuses on the development of ultrasensitive SERS sensors for multiple biochemical sensing and detections.
Xinyu Jiang is an undergraduate student at NJUPT. Currently, he is working on the Research Innovation Program for College Undergraduates of NJUPT in Prof. Wang’s lab.
Chunyuan Song received his PhD degree in optical engineering from Southeast University in 2012. He is now an associate professor at the Institute of Advanced Materials, NJUPT. His research interest focuses on the synthesis, characterization and application of plasmonic nanomaterials for surface-enhanced Raman scattering based biosensing and imaging.
Lianhui Wang obtained his PhD degree in polymeric chemistry and physics at Zhejiang University in 1998. Then he joined Prof. E. T. Kang’s group at the National University of Singapore (NUS) as a postdoctoral researcher from 1998 to 2000, followed by being a researcher and assistant professor at the Institute ofMolecular and Cell Biology, NUS. Since June 2005, he joined the faculty of Fudan University as a professor and then moved to NJUPT in January 2011. Currently, he is a professor at the Institute of AdvancedMaterials, NJUPT. He was granted the funding of “National Distinguished Young Scholar” in 2004, and was honored as “Yangtze River Scholar Distinguished Professor” in 2011. His research group works on bioelectronics and nanobiology including the synthesis of optoelectronic nanomaterials and their applications for biochemical sensing, multimodal imaging, drug delivery and photothermo/chemo/ photodynamic therapy.
Rights and permissions
About this article
Cite this article
Yang, Y., Jiang, X., Chao, J. et al. Synthesis of magnetic core-branched Au shell nanostructures and their application in cancer-related miRNA detection via SERS. Sci. China Mater. 60, 1129–1144 (2017). https://doi.org/10.1007/s40843-017-9022-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-017-9022-1