Abstract
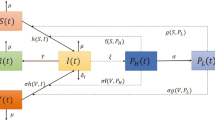
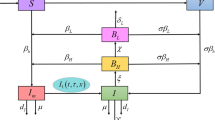
In this paper, a mathematical model is proposed to study the spread of pathogen-induced cholera disease and its control by vaccination. It is assumed in the model that cholera vaccine-induced immunity has a temporary effect and imperfect dose of vaccine does not protect the recipients. From the model, a threshold for the disease dynamics the vaccinated reproduction number \(R_V \) is derived, which is compared with the basic reproduction number \( R_0 \) for without vaccination system. It has been shown that the disease will tend to extinction when \( R_V < 1 \). The disease-free equilibrium point is asymptotically stable when \( R_V < 1 \) and unstable when \( R_V > 1 \). Further, we have also proved that a unique endemic equilibrium point exists when \( R_V > 1 \). Also used Pontryagin Minimum Principle to find out the optimal rate of vaccination and death rate of pathogen population for the control of cholera disease. Sensitivity analysis of system parameters is performed to show their relative importance to disease transmission and prevalence. Finally, numerical simulations are provided to support the analytical results.













Similar content being viewed by others
References
Azman, A.S., Rudolph, K.E., Cummings, D., Lessler, J.: The incubation period of cholera: a systematic review. J. Infect. 66(5), 432–438 (2013)
Braks, M.A., de Roda Husman, A.M.: Dimensions of effects of climate change on water-transmitted infectious diseases. Air Water Borne Dis. 2(1), 2 (2013)
Chitnis, N., Hyman, J.M., Cushing, J.M.: Determining important parameters in the spread of malaria through the sensitivity analysis of a mathematical model. Bull. Math. Biol. 70(5), 1272–96 (2008)
Clemens, J., Harris, J., Khan, M., Kay, B., Yunus, M., Svennerholm, A.: Field trial of oral cholera vaccines in bangladesh. Lancet 2(8499), 124–127 (1986)
Codeco, C.: Endemic and epidemic dynamics of cholera: the role of the aquatic reservoir. BMC Infect. Dis. 1, 1 (2001)
Diekmann, O., Heesterbeek, J., Metz, J.: On the definition and the computation of the basic reproduction ratio \(r_0\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28, 365–382 (1990)
Epstein, P.R., Ford, T.E., Colwell, R.R.: Cholera and the environment. Lancet 342, 1216–1219 (1993)
Funari, E., Manganelli, M., Sinisi, L.: Impact of climate change on water-borne diseases. Ann. Ist. Super. Sanita 48(4), 473–487 (2012)
Hartley, D.M., Morris, J.G., Smith, D.L.: Hyper infectivity: a critical element in the ability of V. cholerae to cause epidemics? Plos Med. 3, 63–69 (2006)
Heffernan, J., Smith, R., Wahl, L.: Perspectives on the basic reproductive ratio. J. R. Soc. Interface 2, 281–293 (2005)
Jeuland, M., Whittingtona, D.: Cost–benefit comparisons of investments in improved water supply and cholera vaccination programs. Vaccine 27, 3109–3120 (2009)
Lemos-Paiao, A.P., Cristiana, J.S., Delfim, F.T.: An epidemic model for cholera with optimal control treatment. J. Comput. Appl. Math. 318, 168–180 (2017)
Lenhart, S., Wortman, J.: Optimal Control Applied to Biological Models. Taylor and Francis, Boca Raton (2007)
Mahoney, R., Maynard, J.: The introduction of new vaccines into developing countries. Vaccine 17, 646–652 (1999)
Meili, L., Junling, M., van den Driessche, P.: Model for disease dynamics of a waterborne pathogen on a random network. Math. Biol. 71(4), 961–977 (2014)
Mukandavire, Z., Liao, S., Wang, J., Gaff, H., Smith, D .L., Morris, J.G.: Estimating the reproductive numbers for the 2008–2009 cholera outbreak in Zimbabwe. Sci. Rep. 108, 8767–8772 (2011)
Mukandavire, Z., Mutasa, F., Hove-Musekwa, S., Dube, S., Tchuenche, J.: Mathematical analysis of a cholera model with carriers: assessing the effects of treatment, chapt. 4. In: Mathematical Biology Research Trends, pp. 109–145. Nova Science Publisher, Inc. (2008)
Mukandavire, Z., Smith, D.L., Morris Jr., J.G.: Cholera in haiti: reproductive numbers and vaccination coverage estimates. Sci. Rep. 3(997), 1–8 (2013)
Neilan, R., Schaefer, E., Gaff, H., Fister, H., Lenhart, S.: Modeling optimal intervention strategies for cholera. Bull. Math. Biol. 72(8), 2004–18 (2010)
Njagarah, J.B.H., Nyabadza, F.: Modelling optimal control of cholera in communities linked by migration. Comput. Math. Methods Med. 2015, 1–12 (2015)
Pandey, P.K., Kass, P.H., Soupir, M.L., Biswas, S., Singh, V.P.: Contaminationof water resources by pathogenic bacteria. AMB Express 4(51), 1–16 (2014)
Park, K.: Preventive and Social Medicine. M/S Banarsi Das Bhanot Publishers, Jabalpur (2005)
Pascual, M., Chaves, L., Cash, B., Rodo, X., Yunus, M.: Predicting endemic cholera: the role of climate variability and disease dynamics. Clim. Res. 36, 131–140 (2008)
Pontryagin, L.S., Boltyanskii, V.G., Gamkrelidze, R.V., Mishchenko, E.: The Mathematical Theory of Optimal Processes. Wiley, New York (1962)
Robertson, S.L., Eisenberg, M.C., Tien, J.H.: Heterogeneity in multiple transmission pathways: modeling the spread of cholera and other waterborne disease in networks with a common water source. J. Biol. Dyn. 7, 254–275 (2013)
Sanches, R.P., Claudia, P.F., Kraenkel, R.A.: The role of immunity and seasonality in cholera epidemics. Bull. Math. Biol. 73, 2916–2931 (2011)
Sanchez, J., Vasquez, B.: Protective efficacy of oral whole-cell/recombinant-bsubunit cholera vaccine in peruvian military. Lancet 344(8932), 1273–1276 (1994)
Shuai, Z., van den Driessche, P.: Global dynamics of cholera models with differential infectivity. Math. Biosci. 234, 118–126 (2011)
Sun, G.-Q., Xie, J.-H., Huang, S.-H., Jin, Z., Li, M.-T., Liu, L.: Transmission dynamics of cholera: mathematical modeling and control strategies. Commun. Nonlinear Sci. Numer. Simul. 45, 235–244 (2017)
Tchuenche, J.M., Mwasa, A.: Mathematical analysis of a cholera model with public health interventions. Biosystems 105, 190–200 (2011)
Tian, J.P., Wang, J.: Global stability for cholera epidemic models. Math. Biosci. 232, 31–41 (2011)
Tien, J.H., Earn, D.J.: Multiple transmission pathways and disease dynamics in a waterborne pathogen model. Bull. Math. Biol. 72, 1506–33 (2010)
van den Driesche, P., Watmough, J.: Reproduction numbers and sub-threshold endemic equilibria for the compartmental models of disease transmission. Math. Biosci. 180, 29–48 (2002)
Wang, J., Modnak, C.: Modeling cholera dynamics with controls. Can. Appl. Math. Q. 19(3), 255–273 (2011)
Wang, Y., Cao, J.: Global dynamics of a network epidemic model for waterborne diseases spread. Appl. Math. Comput. 237, 474–488 (2014)
WHO, Cholera factsheet 107. http://www.who.int/mediacentre/factsheets/fs107/en/ (2015). Accessed 11 Oct 2016
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sisodiya, O.S., Misra, O.P. & Dhar, J. Pathogen Induced Infection and Its Control by Vaccination: A Mathematical Model for Cholera Disease. Int. J. Appl. Comput. Math 4, 74 (2018). https://doi.org/10.1007/s40819-018-0506-x
Published:
DOI: https://doi.org/10.1007/s40819-018-0506-x