Abstract
Background
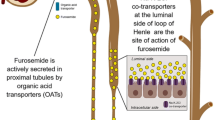
This study assessed the contribution of intracorporeal (IC) and extracorporeal clearance (EC) of furosemide in patients with septic acute kidney injury (AKI), and the relationship between plasma concentrations and urine volume.
Methods
Prospective cohort observational study of 15 patients with septic AKI undergoing continuous veno-venous hemodiafiltration (CVVHDF) divided according to urine volume (< 500 ml/12 h, Oliguria group, n = 5; > 500 ml/12 h, Diuresis group, n = 10) during continuous infusion of furosemide (120 mg/12 h) at steady-state condition. Plasma and effluent furosemide concentrations were determined by high-performance liquid chromatography (HPLC)-mass spectrometry every 12 h for 48 h.
Results
Furosemide plasma concentrations and total body clearance (TBC) were 6.14 mg/l and 22.1 ml/min for the Oliguria group, and 2.63 mg/l and 54.4 ml/min for the Diuresis group, respectively (p < 0.05). When urine volume was < 500 ml/24 h, the furosemide plasma concentrations peaked at the potentially toxic value of 13.0 mg/l. Furosemide EC was not relevant for the Diuresis group, but it represented 18% of TBC for the Oliguria group. Furosemide plasma concentrations correlated positively with dose infusion for both groups (r = 0.728 and 0.685, p < 0.05), and negatively with urine volume only for the Diuresis (r = − 0.578, p < 0.01) but not for the Oliguria group (r = − 0.089, p = 0.715).
Conclusions
For patients with urine volume > 500 ml/12 h continuous infusion of furosemide up to 480 mg/24 h leads to increasing urine volume, which can predict furosemide plasma levels within its safety range. When the urine volume is lower, the furosemide plasma levels are increased beyond any further diuretic efficacy.




Similar content being viewed by others
Abbreviations
- AKI:
-
Acute kidney injury
- TBC:
-
Total body clearance
- EC:
-
Extracorporeal clearance
- IC:
-
Intracorporeal clearance
- KDIGO:
-
Kidney Disease Improving Global Outcomes
- BCrC:
-
Blood creatinine clearance
- CRRT:
-
Continuous renal replacement therapy
- CVVHDF:
-
Continuous veno-venous hemodiafiltration
- ACN:
-
Acetonitrile
References
Bagshaw SM, Bellomo R, Kellum JA (2008) Oliguria, volume overload, and loop diuretics. Crit Care Med 36(4 Suppl):S172–S178
Coppolino G, Presta P, Saturno L, Fuiano G (2013) Acute kidney injury in patients undergoing cardiac surgery. J Nephrol 26:32–40
Omotoso BA, Abdel-Rahman EM, Xin W, Ma JZ, Scully KW, Arogundade FA, Balogun RA (2016) Dialysis requirement, long-term major adverse cardiovascular events (MACE) and all-cause mortality in hospital acquired acute kidney injury (AKI): a propensity-matched cohort study. J Nephrol 29:847–855
Pozzoli S, Simonini M, Manunta P (2018) Predicting acute kidney injury: current status and future challenges. J Nephrol 31:209–223
Ponto LL, Schoenwald RD (1990) Furosemide (frusemide). A pharmacokinetic-pharmacodynamic review (Part I). Clin Pharmacokinet 18:381–408
Kaojarern S, Day B, Brater DC (1982) The time course of delivery of furosemide into urine: an independent determinant of overall response. Kidney Int 22:69–74
Yeh DD, Van Der Wilden GM, Cropano C, Chang Y, King DR, De Moya M, Fagenholz P, Kaafarani H, Lee J, Velmahos G (2015) Goal-directed diuresis: a case–control study of continuous furosemide infusion in critically ill trauma patients. J Emerg Trauma Shock 8:34–38
Alqahtani F, Koulouridis I, Susantitaphong P, Dahal K, Jaber BL (2014) A meta-analysis of continuous vs intermittent infusion of loop diuretics in hospitalized patients. J Crit Care 29:10–17
Beermann B, Dalén E, Lindström B, Rosén A (1975) On the fate of furosemide in man. Eur J Clin Pharmacol 9:51–61
Vree TB, van der Ven AJ (1999) Clinical consequences of the biphasic elimination kinetics for the diuretic effect of furosemide and its acyl glucuronide in humans. J Pharm Pharmacol 51:239–248
Van Wart SA, Shoaf SE, Mallikaarjun S, Mager DE (2014) Population-based meta-analysis of furosemide pharmacokinetics. Biopharm Drug Dispos 35:119–133
Wilcox CS (2002) New insights into diuretic use in patients with chronic renal disease. J Am Soc Nephrol 13:798–805
Silbert BI, Ho KM, Lipman J, Roberts JA, Corcoran TB, Morgan DJ, Pavey W, Mas E, Barden AE, Mori TA (2016) Determinants of urinary output response to IV furosemide in acute kidney injury: a pharmacokinetic/pharmacodynamics study. Crit Care Med 44:e923–e929
Kellum JA, Lameire N; KDIGO AKI Guideline Work Group (2013) Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care 17(1):204
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC (2016) The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 315:801–810
Mariano F, Morselli M, Bergamo D, Hollo Z, Scella S, Maio M, Tetta C, Dellavalle A, Stella M, Triolo G (2011) Blood and ultrafiltrate dosage of citrate as a useful and routine tool during continuous venovenous haemodiafiltration in septic shock patients. Nephrol Dial Transplant 26:3882–3888
Mariano F, Leporati M, Carignano P, Stella M, Vincenti M, Biancone L (2015) Efficient removal of colistin A and B in critically ill patients undergoing CVVHDF and sorbent technologies. J Nephrol 28:623–631
Leporati M, Bua RO, Mariano F, Carignano P, Stella M, Biancone L, Vincenti M (2014) Determination by LC–MS/MS of colistins A and B in plasma and ultrafiltrate from critically ill patients undergoing continuous venovenous hemodiafiltration. Ther Drug Monit 36:182–191
Leporati M, Salomone A, Gole’ G, Vincenti M (2016) Determination of anticoagulant rodenticides and α-chloralose in human hair. Application to a real case. J Anal Toxicol 40:277–285
Chawla LS, Ronco C (2016) Renal stress testing in the assessment of kidney disease. Kidney Int Rep 1:57–63
Mills CD, Whitworth C, Rybak LP, Henley CM (1997) Quantification of furosemide from serum and tissues using high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 701:65–70
Cutler RE, Blair AD (1979) Clinical pharmacokinetics of frusemide. Clin Pharmacokinet 4:279–296
Beermann B, Dalen E, Lindstrom B (1977) Elimination of furosemide in healthy subjects and in those with renal failure. Clin Pharmacol Ther 22:70–78
Hsu CW, Lin SL, Sun SF, Chu KA, Chung HM, Chang HW (2006) Comparison of the diuretic effect of furosemide mixed with human albumin or fresh frozen plasma for patients with hypoalbuminemia in the intensive care unit. J Nephrol 19:621–627
Pichette V, Geadah D, du Souich P (1999) Role of plasma protein binding on renal metabolism and dynamics of furosemide in the rabbit. Drug Metab Dispos 27:81–85
Funding
None declared.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individuals who participated in the study.
Rights and permissions
About this article
Cite this article
Mariano, F., Leporati, M., Carignano, P. et al. Urine volume as a predicting factor for furosemide clearance during continuous infusion in AKI septic shock patients on hemodiafiltration. J Nephrol 31, 889–897 (2018). https://doi.org/10.1007/s40620-018-0537-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-018-0537-8