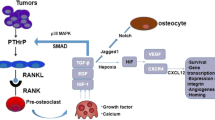
Abstract
Purpose of Review
The more our knowledge on cancer biology progresses, the more it becomes evident that studying the cancer cell isolated from the cancer environment is reductive. Therefore, a better understanding of cancer biology needs a better characterization of the interactions a cancer cell can establish with the surrounding environment. The purpose of this review is to focus on one of the most intriguing cancer/healthy tissue interactions, which occurs in bone during breast cancer-bone metastasis.
Recent Findings
Bone and bone marrow represent a very peculiar environment populated by a variety of cells that cross-communicate. Bone is also by far the most common metastatic site in breast cancer. Breast cancer cells not only colonize the bone, but also alter its metabolism inducing osteoclast-mediated osteolysis. Recent findings further support the relevance of the pathological cross-talk at the basis of the breast cancer-induced bone metastasis, called the vicious cycle. In fact, targeting molecules essential for this cross-talk is already an effective therapeutic strategy, proven to be more constructive than targeting the cancer cells alone.
Summary
We will dissect in this article the vicious cycle and describe the interactions that happen in bone metastasis.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Kohler BA, et al. Annual report to the nation on the status of Cancer, 1975-2011, featuring incidence of breast Cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107(6):djv048.
•• Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271–89. This paper presents a summary of epidemiology and statistics about cancer occurrence and success treatment rate.
Forman D, Bray F, Brewster DH, Gombe Mbalawa C, et al. Cancer incidence in five continents, vol. X. vol. 164. IARC Sci Publ; 2014.
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–86.
• Raphael J, Verma S. Overall survival (OS) endpoint: an incomplete evaluation of metastatic breast cancer (MBC) treatment outcome. Breast Cancer Res Treat. 2015;150(3):473–8. This works provides an overview on pathogenetic and therapeutic aspects for breast cancer metastases.
Williams F, Jeanetta S, James AS. Geographical location and stage of breast Cancer diagnosis: a systematic review of the literature. J Health Care Poor Underserved. 2016;27(3):1357–83.
Brook N, Brook E, Dharmarajan A, Dass CR, Chan A. Breast cancer bone metastases: pathogenesis and therapeutic targets. Int J Biochem Cell Biol. 2018;96:63–78.
van der Pol CB, et al. Breast cancer and bone metastases: the association of axial skeleton MRI findings with skeletal-related events and survival. Breast Cancer Res Treat. 2014;146(3):583–9.
Paget S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev. 1989;8(2):98–101.
•• Cappariello A, Ponzetti M, Rucci N. The ‘soft’ side of the bone: unveiling its endocrine functions. Horm Mol Biol Clin Investig. 2016;28(1):5–20. This paper highlights the endocrine role of skeleton and its function beyond mechanical properties.
Capulli M, Paone R, Rucci N. Osteoblast and osteocyte: games without frontiers. Arch Biochem Biophys. 2014;561:3–12.
Cappariello A, Maurizi A, Veeriah V, Teti A. Reprint of: the great beauty of the osteoclast. Arch Biochem Biophys. 2014;561:13–21.
• Coutu DL, Kokkaliaris KD, Kunz L, Schroeder T. Three-dimensional map of nonhematopoietic bone and bone-marrow cells and molecules. Nat Biotechnol. 2017;35(12):1202–10. This paper describes the spatial distribution of cellular and extracellular components of bone marrows other than hematopoietic lineage.
Sanders JL, Chattopadhyay N, Kifor O, Yamaguchi T, Butters RR, Brown EM. Extracellular calcium-sensing receptor expression and its potential role in regulating parathyroid hormone-related peptide secretion in human breast Cancer cell lines 1. Endocrinology. 2000;141(12):4357–64.
Buijs JT, Stayrook KR, Guise TA. The role of TGF-β in bone metastasis: novel therapeutic perspectives. Bonekey Rep. 2012;1:96.
Dunn LK, Mohammad KS, Fournier PGJ, McKenna CR, Davis HW, Niewolna M, et al. Hypoxia and TGF-β drive breast Cancer bone metastases through parallel signaling pathways in tumor cells and the bone microenvironment. PLoS One. 2009;4(9):e6896.
Sugatani T, Vacher J, Hruska KA. A microRNA expression signature of osteoclastogenesis. Blood. 2011;117(13):3648–57.
Ell B, Mercatali L, Ibrahim T, Campbell N, Schwarzenbach H, Pantel K, et al. Tumor-induced osteoclast miRNA changes as regulators and biomarkers of osteolytic bone metastasis. Cancer Cell. 2013;24(4):542–56.
Lee DY, Deng Z, Wang C-H, Yang BB. MicroRNA-378 promotes cell survival, tumor growth, and angiogenesis by targeting SuFu and Fus-1 expression. Proc Natl Acad Sci. 2007;104(51):20350–5.
Si M-L, Zhu S, Wu H, Lu Z, Wu F, Mo Y-Y. miR-21-mediated tumor growth. Oncogene. 2007;26(19):2799–803.
Pollari S, Leivonen S-K, Perälä M, Fey V, Käkönen S-M, Kallioniemi O. Identification of MicroRNAs inhibiting TGF-β-induced IL-11 production in bone metastatic breast Cancer cells. PLoS One. 2012;7(5):e37361.
Zhu S, Si M-L, Wu H, Mo Y-Y. MicroRNA-21 targets the tumor suppressor gene tropomyosin 1 (TPM1). J Biol Chem. 2007;282(19):14328–36.
Frankel LB, Christoffersen NR, Jacobsen A, Lindow M, Krogh A, Lund AH. Programmed cell death 4 (PDCD4) is an important functional target of the microRNA miR-21 in breast cancer cells. J Biol Chem. 2008;283(2):1026–33.
•• Clément-Demange L, Clézardin P. Emerging therapies in bone metastasis. Curr Opin Pharmacol. 2015;22:79–86. This paper gives an overview on new and potential therapeutical strategies for bone metastases.
Atkins GJ, Kostakis P, Pan B, Farrugia A, Gronthos S, Evdokiou A, et al. RANKL expression is related to the differentiation state of human osteoblasts. J Bone Miner Res. 2003;18(6):1088–98.
Galli C, Fu Q, Wang WF, Olsen BR, Manolagas SC, Jilka RL, et al. Commitment to the osteoblast lineage is not required for RANKL gene expression. J Biol Chem. 2009;284(19):12654–62.
Kozlow W, Guise TA. Breast Cancer metastasis to bone: mechanisms of Osteolysis and implications for therapy. J Mammary Gland Biol Neoplasia. 2005;10(2):169–80.
•• Rao S, Cronin SJF, Sigl V, Penninger JM. RANKL and RANK: from mammalian physiology to Cancer treatment. Trends Cell Biol. 2018;28(3):213–23. This work describes the importance and the role of RANK/RANKL axis in the organogenesis and the involvement in cancer progression.
Schuster C, Mo H, Shen CL, Gollahon L (2017) RANK/RANKL/OPG: The Axis of Breast Cancer Bone Metastasis Evil? Ann Breast Cancer Res 2(1): 1008
Nolan E, et al. RANK ligand as a potential target for breast cancer prevention in BRCA1-mutation carriers. Nat Med. 2016;22(8):933–9. This paper highlights the importance of RANKL in breast cancer onset and the potential therapeutics advantages of a target strategy.
• Vallet S, Bashari MH, Fan FJ, Malvestiti S, Schneeweiss A, Wuchter P, et al. Pre-osteoblasts stimulate migration of breast Cancer cells via the HGF/MET pathway. PLoS One. 2016;11(3):e0150507. This paper describes how important are the stromal-osteoblast cell in the distribution and migration of breast cancer cells during dissemination and metastasization processes.
Ishimi Y, et al. IL-6 is produced by osteoblasts and induces bone resorption. J Immunol. 1990;145(10):3297–303.
• Costa D, Principi E, Lazzarini E, Descalzi F, Cancedda R, Castagnola P, et al. LCN2 overexpression in bone enhances the hematopoietic compartment via modulation of the bone marrow microenvironment. J Cell Physiol. 2017;232(11):3077–87. This paper describes the effect of the proinflammatory cytokine lipocalin 2 in the context of vicious cycle of bone.
Yang J, Bielenberg DR, Rodig SJ, Doiron R, Clifton MC, Kung AL, et al. Lipocalin 2 promotes breast cancer progression. Proc Natl Acad Sci. 2009;106(10):3913–8.
Ören B, Urosevic J, Mertens C, Mora J, Guiu M, Gomis RR, et al. Tumour stroma-derived lipocalin-2 promotes breast cancer metastasis. J Pathol. 2016;239(3):274–85.
Chaudhary LR, Spelsberg TC, Riggs BL. Production of various cytokines by normal human osteoblast-like cells in response to interleukin-1 beta and tumor necrosis factor-alpha: lack of regulation by 17 beta-estradiol. Endocrinology. 1992;130(5):2528–34.
Abuna RPF, De Oliveira FS, Santos TDS, Guerra TR, Rosa AL, Beloti MM. Participation of TNF-α in inhibitory effects of adipocytes on osteoblast differentiation. J Cell Physiol. 2016;231(1):204–14.
Passeri G, Girasole G, Manolagas SC, Jilka RL. Endogenous production of tumor necrosis factor by primary cultures of murine calvarial cells: influence on IL-6 production and osteoclast development. Bone Miner. 1994;24(2):109–26.
Azuma Y, Kaji K, Katogi R, Takeshita S, Kudo A. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J Biol Chem. 2000;275(7):4858–64.
Liu F, Malaval L, Aubin JE. The mature osteoblast phenotype is characterized by extensive plasticity. Exp Cell Res. 1997;232(1):97–105.
Wildemann B, Kadow-Romacker A, Haas NP, Schmidmaier G. Quantification of various growth factors in different demineralized bone matrix preparations. J Biomed Mater Res Part A. 2007;81A(2):437–42.
Rodrigues LR, Teixeira JA, Schmitt FL, Paulsson M, Lindmark-Mänsson H. The role of osteopontin in tumor progression and metastasis in breast cancer. Cancer Epidemiol Biomark Prev. 2007;16(6):1087–97.
Zhang J-H, Tang J, Wang J, Ma W, Zheng W, Yoneda T, et al. Over-expression of bone sialoprotein enhances bone metastasis of human breast cancer cells in a mouse model. Int J Oncol. 2003;23(4):1043–8.
Bengtsson E, Mörgelin M, Sasaki T, Timpl R, Heinegård D, Aspberg A. The leucine-rich repeat protein PRELP binds perlecan and collagens and may function as a basement membrane anchor. J Biol Chem. 2002;277(17):15061–8.
Haglund L, Tillgren V, Önnerfjord P, Heinegård D. The C-terminal peptide of Chondroadherin modulates cellular activity by selectively binding to Heparan sulfate chains. J Biol Chem. 2013;288(2):995–1008.
Rucci N, Capulli M, Ventura L, Angelucci A, Peruzzi B, Tillgren V, et al. Proline/arginine-rich end leucine-rich repeat protein N-terminus is a novel osteoclast antagonist that counteracts bone loss. J Bone Miner Res. 2013;28(9):1912–24.
Rucci N, Capulli M, Olstad OK, Önnerfjord P, Tillgren V, Gautvik KM, et al. The α2β1 binding domain of chondroadherin inhibits breast cancer-induced bone metastases and impairs primary tumour growth: a preclinical study. Cancer Lett. 2015;358(1):67–75.
• Matic I, Matthews BG, Wang X, Dyment NA, Worthley DL, Rowe DW, et al. Quiescent bone lining cells are a major source of osteoblasts during adulthood. Stem Cells. 2016;34(12):2930–42. This paper describes the differentiation process of osteoblasts in adulthood from other source than mesenchymal stem cells.
Zhang C, Tang W, Li Y. Matrix metalloproteinase 13 (MMP13) is a direct target of osteoblast-specific transcription factor Osterix (Osx) in osteoblasts. PLoS One. 2012;7(11):e50525.
Pivetta E, Scapolan M, Pecolo M, Wassermann B, Abu-Rumeileh I, Balestreri L, et al. MMP-13 stimulates osteoclast differentiation and activation in tumour breast bone metastases. Breast Cancer Res. 2011;13(5):R105.
Hock JM, Krishnan V, Onyia JE, Bidwell JP, Milas J, Stanislaus D. Osteoblast apoptosis and bone turnover. J Bone Miner Res. 2001;16(6):975–84.
Koelink PJ, Sier CFM, Hommes DW, Lamers CBHW, Verspaget HW. Clinical significance of stromal apoptosis in colorectal cancer. Br J Cancer. 2009;101(5):765–73.
Wang R-A, Li QL, Li ZS, Zheng PJ, Zhang HZ, Huang XF, et al. Apoptosis drives cancer cells proliferate and metastasize. J Cell Mol Med. 2013;17(1):205–11.
• Xiong J, Piemontese M, Onal M, Campbell J, Goellner JJ, Dusevich V, et al. Osteocytes, not osteoblasts or lining cells, are the main source of the RANKL required for osteoclast formation in remodeling bone. PLoS One. 2015;10(9):e0138189. This paper highlights the importance of osteocytes in controlling osteoclast differentiation and formation through production of RANKL.
Nakashima T, Hayashi M, Fukunaga T, Kurata K, Oh-hora M, Feng JQ, et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat Med. 2011;17(10):1231–4.
• Lee K, Nam K, Oh S, Lim J, Kim YP, Lee JW, et al. Extracellular matrix protein 1 regulates cell proliferation and trastuzumab resistance through activation of epidermal growth factor signaling. Breast Cancer Res. 2014;16(6):479. This paper showed one important aspect of chemioresistance in cancer cells.
Bucciarelli E, Sidoni A, Bellezza G, Cavaliere A, Brachelente G, Costa G, et al. Low dentin matrix protein 1 expression correlates with skeletal metastases development in breast cancer patients and enhances cell migratory capacity in vitro. Breast Cancer Res Treat. 2007;105(1):95–104.
Suva LJ, Washam C, Nicholas RW, Griffin RJ. Bone metastasis: mechanisms and therapeutic opportunities. Nat Rev Endocrinol. 2011;7(4):208–18.
Weilbaecher KN, Guise TA, McCauley LK. Cancer to bone: a fatal attraction. Nat Rev Cancer. 2011;11(6):411–25.
Martin TJ. Manipulating the environment of cancer cells in bone: a novel therapeutic approach. J Clin Invest. 2002;110(10):1399–401.
Takagaki K, et al. Parathyroid hormone-related protein expression, in combination with nodal status, predicts bone metastasis and prognosis of breast cancer patients. Exp Ther Med. 2012;3(6):963–8.
Lu X, Mu E, Wei Y, Riethdorf S, Yang Q, Yuan M, et al. VCAM-1 promotes osteolytic expansion of indolent bone micrometastasis of breast cancer by engaging α4β1-positive osteoclast progenitors. Cancer Cell. 2011;20(6):701–14.
• Becker A, Thakur BK, Weiss JM, Kim HS, Peinado H, Lyden D. Extracellular vesicles in Cancer: cell-to-cell mediators of metastasis. Cancer Cell. 2016;30(6):836–48. This paper describes the crosstalk of cancer cells based on extracellular vesicles as source of biological messagges in tumor microenvironment.
Lee K, Park H, Lim EH, Lee KW. Exosomes from breast cancer cells can convert adipose tissue-derived mesenchymal stem cells into myofibroblast-like cells. Int J Oncol. 2011;40(1):130–8.
•• Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527(7578):329–35. This work shows the involvment of extracellular vesicles and their pivotal role in determining the organotropism in the dissemination of a primary tumour.
Luga V, Zhang L, Viloria-Petit AM, Ogunjimi AA, Inanlou MR, Chiu E, et al. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast Cancer cell migration. Cell. 2012;151(7):1542–56.
Deng L, Wang Y, Peng Y, Wu Y, Ding Y, Jiang Y, et al. Osteoblast-derived microvesicles: a novel mechanism for communication between osteoblasts and osteoclasts. Bone. 2015;79:37–42.
•• Cappariello A, Loftus A, Muraca M, Maurizi A, Rucci N, Teti A. Osteoblast-derived extracellular vesicles are biological tools for the delivery of active molecules to bone. J Bone Miner Res. 2018;33(3):517–33. This paper shows the involvement of extracellular vesicle in the context of bone physiology between osteoblasts and osteoclasts.
• Harbeck N, Gnant M. Breast cancer. Lancet. 2017;389(10074):1134–50. This paper provides an overview on biological basis of breast cancer and potential therapeutical approaches.
•• McDonald ES, Clark AS, Tchou J, Zhang P, Freedman GM. Clinical diagnosis and Management of Breast Cancer. J Nucl Med. 2016;57(Suppl 1, no. Supplement 1):9S–16S. This work makes a summary on the main diagnostic and therapeutical strategies to treat breast cancer.
• Francis PA, Regan MM, Fleming GF, Láng I, Ciruelos E, Bellet M, et al. Adjuvant ovarian suppression in premenopausal breast Cancer. N Engl J Med. 2015;372(5):436–46. This paper reports the impact of estrogen-bloking chemioadjuvant on premenopausal breast cancer patients.
International Breast Cancer Study Group, et al. Tamoxifen after adjuvant chemotherapy for premenopausal women with lymph node-positive breast cancer: international breast Cancer study group trial 13-93. J Clin Oncol. 2006;24(9):1332–41.
Zallone A. Direct and indirect estrogen actions on osteoblasts and osteoclasts. Ann N Y Acad Sci. 2006;1068(1):173–9.
Klibanski, A., Adams-Campbell, L., Bassford, T., Blair, S. N., Boden, et al. Osteoporosis prevention, diagnosis, and therapy. JAMA, 2001;285(6), 785-795
Zhang Y, Kiel DP, Kreger BE, Cupples LA, Ellison RC, Dorgan JF, et al. Bone mass and the risk of breast Cancer among postmenopausal women. N Engl J Med. 1997;336(9):611–7.
Powles TJ, Diem SJ, Fabian CJ, Neven P, Wickerham DL, Cox DA, et al. Breast cancer incidence in postmenopausal women with osteoporosis or low bone mass using arzoxifene. Breast Cancer Res Treat. 2012;134(1):299–306.
von Minckwitz G, Raab G, Caputo A, Schütte M, Hilfrich J, Blohmer JU, et al. Doxorubicin with cyclophosphamide followed by docetaxel every 21 days compared with doxorubicin and docetaxel every 14 days as preoperative treatment in operable breast cancer: the GEPARDUO study of the German breast group. J Clin Oncol. 2005;23(12):2676–85.
•• Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384(9938):164–72. This paper reports the success rate and the beneficial effect on quality of life for breast cancer patients.
• Anampa J, Makower D, Sparano JA. Progress in adjuvant chemotherapy for breast cancer: an overview. BMC Med. 2015;13(1):195. This paper comment on the current precedures and pharmacological treatment in managing of brest cancer.
Takahashi M, Mizoguchi, T., Uehara, S. et al. Docetaxel inhibits bone resorption through suppression of osteoclast formation and function in different manners. J Bone Miner Metab (2009) 27(1):24–35
Wong M, N. P.-B. C. T. and Therapy, and undefined. Optimal management of bone metastases in breast cancer patients. 2011. ncbi.nlm.nih.gov.
Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate Cancer. Clin Nucl Med. 2013;38(12):1.
Takalkar A, Adams S, Subbiah V. Radium-223 dichloride bone-targeted alpha particle therapy for hormone-refractory breast cancer metastatic to bone. Exp Hematol Oncol. 2014;3(1):23.
Nilsson S, Franzén L, Parker C, Tyrrell C, Blom R, Tennvall J, et al. Bone-targeted radium-223 in symptomatic, hormone-refractory prostate cancer: a randomised, multicentre, placebo-controlled phase II study. Lancet Oncol. 2007;8(7):587–94.
Russell RGG. Bisphosphonates: the first 40years. Bone. 2011;49(1):2–19.
Mundy GR, Yoneda T. Bisphosphonates as anticancer drugs. N Engl J Med. 1998;339(6):398–400.
Santini D, Fratto ME, Galluzzo S, Vincenzi B, Tonini G. Are bisphosphonates the suitable anticancer drugs for the elderly? Crit Rev Oncol. 2009;69:83–94.
Coleman R, de Boer R, Eidtmann H, Llombart A, Davidson N, Neven P, et al. Zoledronic acid (zoledronate) for postmenopausal women with early breast cancer receiving adjuvant letrozole (ZO-FAST study): final 60-month results. Ann Oncol. 2013;24(2):398–405.
Bekker PJ, Holloway DL, Rasmussen AS, Murphy R, Martin SW, Leese PT, et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J Bone Miner Res. 2004;19(7):1059–66.
Martin M, Bell R, Bourgeois H, Brufsky A, Diel I, Eniu A, et al. Bone-related complications and quality of life in advanced breast cancer: results from a randomized phase III trial of Denosumab versus Zoledronic acid. Clin Cancer Res. 2012;18(17):4841–9.
Rosen LS, et al. Long-term efficacy and safety of zoledronic acid compared with pamidronate disodium in the treatment of skeletal complications in patients with advanced multiple myeloma or breast carcinoma: a randomized, double-blind, multicenter, comparative trial. Cancer. 2003;98(8):1735–44.
Bonomi M, Nortilli R, Molino A, Sava T, Santo A, Caldara A, et al. Renal toxicity and osteonecrosis of the jaw in cancer patients treated with bisphosphonates: a long-term retrospective analysis. Med Oncol. 2010;27(2):224–9.
Migliorati CA, et al. A systematic review of bisphosphonate osteonecrosis (BON) in cancer. Support Care Cancer. 2010;18(8):1099–106.
Bamias A, Kastritis E, Bamia C, Moulopoulos LA, Melakopoulos I, Bozas G, et al. Osteonecrosis of the jaw in Cancer after treatment with bisphosphonates: incidence and risk factors. J Clin Oncol. 2005;23(34):8580–7.
Saad F, et al. Incidence, risk factors, and outcomes of osteonecrosis of the jaw: integrated analysis from three blinded active-controlled phase III trials in cancer patients with bone metastases. Ann Oncol Off J Eur Soc Med Oncol. 2012;23(5):1341–7.
Roodman GD. Pathophysiology of bone metastases. Dordrecht: Springer; 2009. p. 31–50.
Yin JJ, Selander K, Chirgwin JM, Dallas M, Grubbs BG, Wieser R, et al. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J Clin Invest. 1999;103(2):197–206.
Lewiecki EM. Odanacatib, a cathepsin K inhibitor for the treatment of osteoporosis and other skeletal disorders associated with excessive bone remodeling. IDrugs. 2009;12(12):799–809.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Alfredo Cappariello reports having patent 16745158.1-1112 pending; Mattia Capulli declares no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Molecular Biology of Bone Metastasis
Rights and permissions
About this article
Cite this article
Cappariello, A., Capulli, M. The Vicious Cycle of Breast Cancer-Induced Bone Metastases, a Complex Biological and Therapeutic Target. Curr Mol Bio Rep 4, 123–131 (2018). https://doi.org/10.1007/s40610-018-0099-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40610-018-0099-5