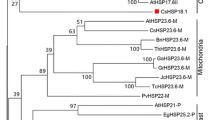
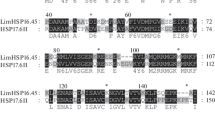
Abstract
Abiotic stresses such as drought, heat and salinity are responsible for decelerating agricultural productivity all over the world and the destructive aftermaths are likely to increase further due to changing global climate. Small heat shock proteins (sHsps) are synthesized ubiquitously and play diverse roles in abiotic stress responses of plants. sHsps are reported to be involved in preventing aggregation, stabilizing non-native proteins. In the present study, Hsp17.9 gene from Prosopis cineraria was cloned, characterized and expression was studied in prokaryotic system. In silico analysis was carried out to gain insights in to protein structure and its interaction with other molecules. Overexpression of the gene in Escherichia coli BL21 (DE3) cells was analyzed on SDS–PAGE, followed by western blotting using Anti His-tag antibody and purified by nickel–nitrilo triacetic acid resin based columns. The single purified band was confirmed by peptide mass fingerprinting. Two dimensional protein analysis of bacterial protein showed the presence of overexpressed protein spot at the desired place. Further, the recombinant E. coli cells carrying pET28a-PcHsp17.9 construct were able to tolerate abiotic stresses better than the cells carrying pET28a vector alone. The cloned PcHsp17.9 gene may be useful to generate abiotic stress tolerant transgenic crops for a climate smart agriculture.




Similar content being viewed by others
Abbreviations
- SDS–PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- E. coli :
-
Escherichia coli
- sHsps :
-
Small heat shock proteins
- Ni–NTA:
-
Nickel–nitrilo triacetic acid
- IPTG:
-
Isopropyl-β-D thiogalactoside
- PVDF:
-
Polyvinylidene difluoride
References
Ben-Zvi, A. P., & Goloubinoff, P. (2001). Review: Mechanisms of disaggregation and refolding of stable protein aggregates by molecular chaperones. Journal of Structural Biology. https://doi.org/10.1006/jsbi.2001.4352.
Bertioli, D. J., Cannon, S. B., Froenicke, L., Huang, G., Farmer, A. D., Cannon, E. K. S., et al. (2016). The genome sequences of Arachis duranensis and Arachis ipaensis, the diploid ancestors of cultivated peanut. Nature Genetics, 48(4), 438–446. https://doi.org/10.1038/ng.3517.
Chaurasia, N., Mishra, Y., & Rai, L. C. (2008). Cloning expression and analysis of phytochelatin synthase (pcs) gene from Anabaena sp. PCC 7120 offering multiple stress tolerance in Escherichia coli. Biochemical and Biophysical Research Communications, 376(1), 225–230. https://doi.org/10.1016/j.bbrc.2008.08.129.
Diep, P. H., Vishwakarma, H., Solanke, A., Kumar, U., & Padaria, J. C. (2017). Identification of drought stress responsive genes from Prosopis cineraria (L.) Druce for climate resilient agriculture. Biotech Today: An International Journal of Biological Sciences, 7(1), 25. https://doi.org/10.5958/2322-0996.2017.00002.3.
Feder, M. E., & Hofmann, G. E. (1999). Heat-shock proteins, molecular chaperones, and the stress response: Evolutionary and ecological physiology. Annual Review of Physiology, 61(1), 243–282. https://doi.org/10.1146/annurev.physiol.61.1.243.
Georgopoulos, C., & Welch, W. J. (1993). Role of the major heat shock proteins as molecular chaperones. Annual Review of Cell Biology, 9(1), 601–634. https://doi.org/10.1146/annurev.cb.09.110193.003125.
Gupta, K., Agarwal, P. K., Reddy, M. K., & Jha, B. (2010). SbDREB2A, an A-2 type DREB transcription factor from extreme halophyte Salicornia brachiata confers abiotic stress tolerance in Escherichia coli. Plant Cell Reports, 29(10), 1131–1137. https://doi.org/10.1007/s00299-010-0896-7.
Lee, G. J., & Vierling, E. (2000). A small heat shock protein cooperates with heat shock protein 70 systems to reactivate a heat-denatured protein. Plant Physiology, 122(1), 189–198. https://doi.org/10.1104/pp.122.1.189.
Liu, Y., & Zheng, Y. (2005). PM2, a group 3 LEA protein from soybean, and its 22-mer repeating region confer salt tolerance in Escherichia coli. Biochemical and Biophysical Research Communications, 331(1), 325–332. https://doi.org/10.1016/j.bbrc.2005.03.165.
Lovell, S. C., Davis, I. W., Arendall, W. B., de Bakker, P. I. W., Word, J. M., Prisant, M. G., et al. (2003). Structure validation by Cα geometry: ϕ, ψ and Cβ deviation. Proteins: Structure, Function, and Bioinformatics, 50(3), 437–450. https://doi.org/10.1002/prot.10286.
Mayer, M. P., & Bukau, B. (2005). Hsp70 chaperones: Cellular functions and molecular mechanism. Cellular and Molecular Life Sciences. https://doi.org/10.1007/s00018-004-4464-6.
Nakamoto, H., & Vígh, L. (2007). The small heat shock proteins and their clients. Cellular and Molecular Life Sciences. https://doi.org/10.1007/s00018-006-6321-2.
Padaria, J. C., Thuy, N. T., Tarafdar, A., & Yadav, R. (2015). Development of a heat-responsive cDNA library from Prosopis cineraria and molecular characterisation of the Pchsp17.9 gene. The Journal of Horticultural Science & Biotechnology, 90(3), 318–324. https://doi.org/10.1080/14620316.2015.11513189.
Padaria, J. C., Vishwakarma, H., Biswas, K., Jasrotia, R. S., & Singh, G. P. (2014). Molecular cloning and in silico characterization of high temperature stress responsive pAPX gene isolated from heat tolerant Indian wheat cv. Raj 3765. BMC Research Notes, 7(1), 713. https://doi.org/10.1186/1756-0500-7-713.
Padaria, J. C., Yadav, R., Tarafdar, A., Lone, S. A., Kumar, K., & Sivalingam, P. N. (2016). Molecular cloning and characterization of drought stress responsive abscisic acid-stress-ripening (AsrI) gene from wild jujube, Ziziphus nummularia (Burm.f.) Wight & Arn. Molecular Biology Reports, 43(8), 849–859. https://doi.org/10.1007/s11033-016-4013-z.
Pandurangaiah, M., Reddy, K. E., Lokanadha Rao, G., Sivakumar, M., Sudhakarbabu, O., Nareshkumar, A., et al. (2013). Cloning and expression analysis of MuNAC4 transcription factor protein from horsegram (Macrotyloma uniflorum (Lam.) Verdc.) conferred salt stress tolerance in Escherichia coli. Acta Physiologiae Plantarum, 35(1), 139–146. https://doi.org/10.1007/s11738-012-1056-1.
Rai M. K., Shekhawat J. K., Kataria V. & Shekhawat N. S. (2017). De novo assembly of leaf transcriptome, functional annotation and genomic resources development in Prosopis cineraria, a multipurpose tree of Indian Thar Desert. Plant Gene. https://doi.org/10.1016/j.plgene.2017.09.002
Ramoliya, P. J., Patel, H. M., Joshi, J. B., & Pandey, A. N. (2006). Effect of salinization of soil on growth and nutrient accumulation in seedlings of Prosopis cineraria. Journal of Plant Nutrition, 29(2), 283–303. https://doi.org/10.1080/01904160500476806.
Saidi, Y., Finka, A., Muriset, M., Bromberg, Z., Weiss, Y. G., Maathuis, F. J. M., et al. (2009). the heat shock response in moss plants is regulated by specific calcium-permeable channels in the plasma membrane. Plant Cell, 21(9), 2829–2843. https://doi.org/10.1105/tpc.108.065318.
Schmutz, J., Cannon, S. B., Schlueter, J., Ma, J., Mitros, T., Nelson, W., et al. (2010). Genome sequence of the palaeopolyploid soybean. Nature, 463(7278), 178–183. https://doi.org/10.1038/nature08670.
Shevchenko, A., Tomas, H., Havlis, J., Olsen, J. V., & Mann, M. (2007). In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nature Protocols, 1(6), 2856–2860. https://doi.org/10.1038/nprot.2006.468.
Sun, W., Van Montagu, M., & Verbruggen, N. (2002). Small heat shock proteins and stress tolerance in plants. Biochimica et biophysica acta, 1577(1), 1–9.
Tajrishi, M. M., Vaid, N., Tuteja, R., & Tuteja, N. (2011). Overexpression of a pea DNA helicase 45 in bacteria confers salinity stress tolernace. Plant Signaling and Behaviour, 6(9), 1271–1275. https://doi.org/10.4161/psb.6.9.16726.
Varshney, R. K., Chen, W., Li, Y., Bharti, A. K., Saxena, R. K., Schlueter, J. A., et al. (2012). Draft genome sequence of pigeonpea (Cajanus cajan), an orphan legume crop of resource-poor farmers. Nature Biotechnology, 30(1), 83–89. https://doi.org/10.1038/nbt.2022.
Varshney, R. K., Song, C., Saxena, R. K., Azam, S., Yu, S., Sharpe, A. G., et al. (2013). Draft genome sequence of chickpea (Cicer arietinum) provides a resource for trait improvement. Nature Biotechnology, 31(3), 240–246. https://doi.org/10.1038/nbt.2491.
Vierling, E. (1991). The roles of heat shock proteins in plants. Annual Review of Plant Physiology and Plant Molecular Biology, 42(1), 579–620. https://doi.org/10.1146/annurev.pp.42.060191.003051.
Vishwakarma, H., Junaid, A., Manjhi, J., Singh, G. P., Gaikwad, K., & Padaria, J. C. (2018). Heat stress transcripts, differential expression, and profiling of heat stress tolerant gene TaHsp90 in Indian wheat (Triticum aestivum L.) cv C306. PLoS ONE, 13(6), e0198293. https://doi.org/10.1371/journal.pone.0198293.
Acknowledgements
Authors are thankful to the Project Director, NRCPB (National Research Centre on Plant Biotechnology), Delhi, India for providing facilities to carry out research work and Director, IARI (Indian Agricultural Research Institute) for providing National Phytotron Facility, IARI, New Delhi, India. The funds provided by the ICAR (Indian Council of Agricultural Research under NICRA (National Innovations in Climate Resilient Agriculture) project is duly acknowledged
Author information
Authors and Affiliations
Contributions
Experimentation: AK, HV. Writing original draft, data curation, formal analysis: AK, HV, AM. Conceptualization, resources, supervision, review and editing, project administration: JCP. Review and editing: JCP
Corresponding author
Ethics declarations
Conflict of interest
All the authors have read the manuscript and declared that no conflict interest exists.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaur, A., Vishwakarma, H., Maibam, A. et al. Cloning, characterization and in silico studies on abiotic stress responsive Hsp17.9 from Prosopis cineraria. Ind J Plant Physiol. 23, 731–740 (2018). https://doi.org/10.1007/s40502-018-0414-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-018-0414-4