Abstract
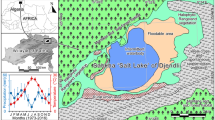
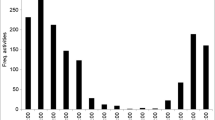
Life in desert ecosystems drives animals to adapt their surface locomotor activity according to environmental conditions. In this study, the hourly and monthly variations in the surface activity patterns of the terrestrial crustacean Hemilepistus reaumurii were investigated. The surface activity of H. reaumurii at the population scale was observed by collecting the hourly active individuals from the sunrise to the sunset of the studied day in each month of 2013 in the Bchachma locality, Tunisia. The collected active individuals were put in perspex boxes (on which we labeled the hourly time interval in which the individuals were collected) in the field and then transported to the laboratory for analysis. Individuals were counted, sexed, and measured in the laboratory. Despite desert conditions in the studied site, H. reaumurii was characterized by a diurnal surface activity, showing a bimodal pattern during the warm months (i.e., May to October). However, it exhibited a unimodal surface activity pattern in the cold months (i.e., February, March, and November). The surface activity was significantly correlated with sunrise and sunset. Moreover, a significant quadratic effect of temperature on the surface activity of H. reaumurii was observed. Furthermore, the study showed that the most important surface activity was recorded in March. The daily exploitation of the temporal niches was significantly different as a function of months. The body size of males was larger than that of females, and the body size of active individuals changed with months. All these behavioural changes in the surface activity represent an adaptive strategy of life in the arid environment.
Similar content being viewed by others
References
Alpato A M, Rietveld W J, Oryntaeva L B, et al. 1994. Properties of the two-peak free running circadian rhythm of locomotor activity of the sand desert beetle Trigonoscelis gigas reitt. Biological Rhythm Research, 25(2): 153–167.
Alpatov A M, Zotov V A, Tshernyshev W B, et al. 1999. Endogenous circadian rhythm is a crucial tool for survival of the sand-desert tenebrionid beetle Trigonoscelis gigas Reitter. Biological Rhythm Research, 30(1): 104–109.
Aschoff J. 1986. Anticipation of daily meal: a process of learning due to entrainment. Monitore Zoologico Italiano, 20: 195–219.
Ayari A, Jelassi R, Ghemari C, et al. 2015. Effect of age, sex, and mutual interaction on the locomotor behavior of Orchestia gammarellus in the supralittoral zone of Ghar El Melh lagoon (Bizerte, Tunisia). Biological Rhythm Research, 46(5): 703–714.
Ayari A, Raimond M, Souty-Grosset C, et al. 2016a. Hierarchical organization of the cuticle of the subsocial desert isopod, Hemilepistus reaumurii. Journal of Structural Biology, 193(2): 115–123.
Ayari A, Richard F J, Souty-Grosset C, et al. 2016b. Family identity of the sub-social desert terrestrial isopod Hemilepistus reaumurii. Journal of Arid Environments, 134: 10–16.
Ayari A, Jelassi R, Ghemari C, et al. 2017. Locomotor activity pattern in pairs of the subsocial desert isopod Hemilepistus reaumuri. Biological Rhythm Research, 48(3): 449–457.
Baker M B. 2005. Experience influences settling behaviour in desert isopods, Hemilepistus reaumuri. Animal Behaviour, 69(5): 1131–1138.
Blanckenhorn W U. 2007. Case studies of the differential-equilibrium hypothesis of sexual size dimorphism in two dung fly species. In: Fairbairn D J, Blanckenhorn W U, Szekély T. Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism. Oxford, UK: Oxford University Press, 106–114.
Bohli D, Morgan E, Charfi-Cheikhrouha F, et al. 2006. Study of locomotor activity of Talitrus saltator (Crustacea, Amphipoda) from Zouarâa beach (Tunisia). In: Scapini F. Proceedings of the MEDCORE International Conference, 10th–14th November 2005. Florence: Firenze University Press, 209–219. (in Italian)
Bohli-Abderrazak D, Ayari A, Morgan E, et al. 2012. Towards a characterization of the locomotor activity rhythm of the supralittoral isopod Tylos europaeus. Chronobiology International, 29(2): 166–174.
Chelazzi L, Colombini I. 1989. Zonation and activity patterns of two species of the genus Phaleria Latreille (Coleoptera Tenebrionidae) inhabiting an equatorial and a Mediterranean sandy beach. Ethology Ecology & Evolution, 1(4): 313–321.
Cloudsley-Thompson J L. 1990. Thermal ecology and behaviour of Physadesmia globosa (Coleoptera: Tenebrionidae) in the Namib desert. Journal of Arid Environments, 19(3): 317–324.
Cloudsley-Thompson J L. 1991. Ecophysiology of Desert Arthropods and Reptiles. Heidelberg, Berlin, Tokyo: Springer-Verlag, 203.
Cloudsley-Thompson J L. 2001. Thermal and water relations of desert beetles. Naturwissenschaften, 88(11): 447–460.
Cros S, Cerdá X, Retana J. 1997. Spatial and temporal variations in the activity patterns of Mediterranean ant communities. Écoscience, 4(3): 269–278.
Dubinsky Z, Steinberger Y, Shachak M. 1979. The survival of the desert isopod Hemilepistus reaumurii (Audouin) in relation to temperature (Isopoda, Oniscoidea). Crustaceana, 36(2): 147–154.
Eduard K, Linsenmair C. 1971. Pairing and couple cohesion in the monogamous desert isopod Hemilepistus reaumuri (Crustacea, Isopoda, Oniscoidea). Journal of Animal Psychology, 29(2): 134–155.
Fairbairn D J. 2007. Introduction: the enigma of sexual size dimorphism. In: Fairbairn D J, Blanckenhorn W U, Szekély T. Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism. Oxford, UK: Oxford University Press, 1–10.
Fleissner G, Fleissner G. 1998. Natural photic zeitgeber signals and underlying neuronal mechanisms in Scorpions. In: Touitou Y. Biological Clocks: Mechanisms and Applications. Paris: Elsevier, 171–180.
Fleissner G, Fleissner G. 2002. Perception of natural zeitgeber signals. In: Kumar V. Biological Rhythms. New Delhi, New York: Narosa/Springer, 71–82.
Jayatilaka P, Narendra A, Reid S F, et al. 2011. Different effects of temperature on foraging activity schedules in sympatric Myrmecia ants. Journal of Experimental Biology, 214(16): 2730–2738.
Jelassi R, Ayari A, Nasri-Ammar K. 2013. Seasonal variation of locomotor activity rhythm of Orchestia gammarellus in the supralittoral zone of Ghar Melh lagoon (North-East of Tunisia). Biological Rhythm Research, 44(6): 956–967.
Kennedy F, Naylor E, Jaramillo E. 2000. Ontogenetic differences in the circadian locomotor activity rhythm of the talitrid amphipod crustacean Orchestoidea tuberculata. Marine Biology, 137(3): 511–517.
Lewis R D, Bullivant A G, King V M. 1991. A dual pacemaker model for the circadian system of the insect Hemideina thoracica. Journal of Interdisciplinary Cycle Research, 22(4): 293–309.
Linsenmair K E. 1984. Comparative studies on the social behavior of the desert isopod Hemilepistus reaumuri and of a Porcellio species. In: Sutton S L, Holdich D M. The Biology of Terrestrial Isopods. Oxford: Oxford University Press, 53: 423–454.
Linsenmair K E. 2007. Sociobiology of terrestrial isopods. In: Duffy J E, Thiel M. Evolutionary Ecology of Social and Sexual Systems: Crustaceans as Model Organisms. New York: Oxford University Press, 339–364.
Nasri K, Juchault P, Mocquard J P, et al. 1996. Seasonal reproduction in Hemilepistus reaumuri (Audouin, 1826), terrestrial isopod of semi-arid zones. Crustaceana, 69(2): 223–235.
Nasri-Ammar K, Morgan E. 2005. Preliminary observations on the natural variation in the endogenous rhythm of the desert isopod Hemilepistus reaumurii. European Journal of Soil Biology, 41(3–4): 63–68.
Nasri-Ammar K, Jelassi R, Ayari A, et al. 2016. Seasonal changes of locomotor activity patterns in the desert isopod Hemilepistus reaumuri. Biological Rhythm Research, 47(1): 25–38.
Parmenter R R, Parmenter C A, Cheney C D. 1989. Factors influencing microhabitat partitioning in arid-land darkling beetles (Tenebrionidae): temperature and water conservation. Journal of Arid Environments, 17(1): 57–67.
Pol R, de Casenave J L. 2004. Activity patterns of harvester ants Pogonomyrmex pronotalis and Pogonomyrmex rastratus in the central Monte desert, Argentina. Journal of Insect Behavior, 17(5): 647–661.
Robinson M D, Clayton D A. 2001. Thermal ecology and seasonal activity patterns of Erodius sauditus, an Arabian dune beetle (Tenebrionidae: Erodinae). In: Prakash I. Ecology of Desert Environments. Jodhpur: Scientific Publishers, 281–291.
Shachak M. 1980. Energy allocation and life history strategy of the desert isopod H. reaumuri. Oecologia, 45(3): 404–413.
Shachak M, Yair A. 1984. Population dynamics and the role of Hemilepistus reaumuri in a desert ecosystem. In: Sutton S L, Holdich D M. The Biology of Terrestrial Isopods. Oxford: Oxford University Press, 53: 295–314.
Smith M D, Brockmann H J. 2014. The evolution and maintenance of sexual size dimorphism in horseshoe crabs: an evaluation of six functional hypotheses. Animal Behaviour, 96: 127–139.
Vandel A. 1949. On a collection of terrestrial isopods collected by F. PIERRE, in southern Algeria, and on the presence of a new Irano-Turanian element in the Saharan fauna. Bulletin of the Zoological Society of France, 74: 310–316.
Zotov V A, Alpatov A A. 2004. Ecological aspects of circadian rhythms in six species of omni-seasonal beetles (Coleoptera, Tenebrionidae) inhabiting Kara Kum Desert (Turkmenistan). Biological Rhythm Research, 35(1–2): 93–103.
Acknowledgements
This work was supported by the University of Tunis El Manar. We wish to thank Mrs. Sabiha KHIARI-AYARI, Ms. Nada AYARI, and Mr. Assad ABIDI for their important contribution in accomplishing the field work of this study. We also wish to thank Dr. Julian REYNOLDS for constructive remarks and English revision of the manuscript. We also thank the two anonymous reviewers for their helpful comments on this paper.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ayari, A., Touihri, M., Ghemari, C. et al. Hourly and monthly variations in the surface activity patterns of Hemilepistus reaumurii in arid environments of Tunisia. J. Arid Land 10, 470–481 (2018). https://doi.org/10.1007/s40333-018-0058-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40333-018-0058-2