Abstract
Exercise training in combination with optimal nutritional support is an effective strategy to maintain or increase skeletal muscle mass. A single bout of resistance exercise undertaken with adequate protein availability increases rates of muscle protein synthesis and, when repeated over weeks and months, leads to increased muscle fiber size. While resistance-based training is considered the ‘gold standard’ for promoting muscle hypertrophy, other modes of exercise may be able to promote gains in muscle mass. High-intensity interval training (HIIT) comprises short bouts of exercise at or above the power output/speed that elicits individual maximal aerobic capacity, placing high tensile stress on skeletal muscle, and somewhat resembling the demands of resistance exercise. While HIIT induces rapid increases in skeletal muscle oxidative capacity, the anabolic potential of HIIT for promoting concurrent gains in muscle mass and cardiorespiratory fitness has received less scientific inquiry. In this review, we discuss studies that have determined muscle growth responses after HIIT, with a focus on molecular responses, that provide a rationale for HIIT to be implemented among populations who are susceptible to muscle loss (e.g. middle-aged or older adults) and/or in clinical settings (e.g. pre- or post-surgery).


Similar content being viewed by others
References
Hawley JA, Hargreaves M, Joyner MJ, Zierath JR. Integrative biology of exercise. Cell. 2014;159(4):738–49. https://doi.org/10.1016/j.cell.2014.10.029.
Beaudart C, Zaaria M, Pasleau F, Reginster JY, Bruyere O. Health outcomes of sarcopenia: a systematic review and meta-analysis. PLoS ONE. 2017;12(1):e0169548. https://doi.org/10.1371/journal.pone.0169548.
Woo T, Yu S, Visvanathan R. Systematic literature review on the relationship between biomarkers of sarcopenia and quality of life in older people. J Frailty Aging. 2016;5(2):88–99. https://doi.org/10.14283/jfa.2016.93.
Fisher G, Brown AW, Bohan Brown MM, Alcorn A, Noles C, Winwood L, et al. High intensity interval- vs moderate intensity-training for improving cardiometabolic health in overweight or obese males: a randomized controlled trial. PLoS ONE. 2015;10(10):e0138853. https://doi.org/10.1371/journal.pone.0138853.
Mayhew AJ, Amog K, Phillips S, Parise G, McNicholas PD, de Souza RJ, et al. The prevalence of sarcopenia in community-dwelling older adults, an exploration of differences between studies and within definitions: a systematic review and meta-analyses. Age Ageing. 2019;48(1):48–56. https://doi.org/10.1093/ageing/afy106.
Falcon LJ, Harris-Love MO. Sarcopenia and the new icd-10-cm code: Screening, staging, and diagnosis considerations. Fed Pract. 2017;34(7):24–32.
Goates S, Du K, Arensberg MB, Gaillard T, Guralnik J, Pereira SL. Economic impact of hospitalizations in us adults with sarcopenia. J Frailty Aging. 2019;8(2):93–9. https://doi.org/10.14283/jfa.2019.10.
Norman K, Otten L. Financial impact of sarcopenia or low muscle mass - a short review. Clin Nutr. 2019;38(4):1489–95. https://doi.org/10.1016/j.clnu.2018.09.026.
Frontera WR, Hughes VA, Lutz KJ, Evans WJ. A cross-sectional study of muscle strength and mass in 45- to 78-year-old men and women. J Appl Physiol (1985). 1991;71(2):644–50.
Roubenoff R, Hughes VA. Sarcopenia: current concepts. J Gerontol A Biol Sci Med Sci. 2000;55(12):M716–24.
Macaluso A, De Vito G. Muscle strength, power and adaptations to resistance training in older people. Eur J Appl Physiol. 2004;91(4):450–72. https://doi.org/10.1007/s00421-003-0991-3.
von Haehling S, Morley JE, Anker SD. An overview of sarcopenia: facts and numbers on prevalence and clinical impact. J Cachexia Sarcopenia Muscle. 2010;1(2):129–33. https://doi.org/10.1007/s13539-010-0014-2.
Sayer AA, Syddall H, Martin H, Patel H, Baylis D, Cooper C. The developmental origins of sarcopenia. J Nutr Health Aging. 2008;12(7):427–32.
Stokes T, Hector AJ, Morton RW, McGlory C, Phillips SM. Recent perspectives regarding the role of dietary protein for the promotion of muscle hypertrophy with resistance exercise training. Nutrients. 2018. https://doi.org/10.3390/nu10020180.
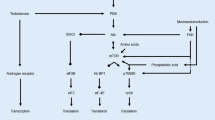
McGlory C, Devries MC, Phillips SM. Skeletal muscle and resistance exercise training; the role of protein synthesis in recovery and remodeling. J Appl Physiol (1985). 2017;122(3):541–8. https://doi.org/10.1152/japplphysiol.00613.2016.
Bigaard J, Frederiksen K, Tjonneland A, Thomsen BL, Overvad K, Heitmann BL, et al. Body fat and fat-free mass and all-cause mortality. Obes Res. 2004;12(7):1042–9. https://doi.org/10.1038/oby.2004.131.
Li R, Xia J, Zhang XI, Gathirua-Mwangi WG, Guo J, Li Y, et al. Associations of muscle mass and strength with all-cause mortality among us older adults. Med Sci Sports Exerc. 2018;50(3):458–67. https://doi.org/10.1249/mss.0000000000001448.
Merom D, Pye V, Macniven R, van der Ploeg H, Milat A, Sherrington C, et al. Prevalence and correlates of participation in fall prevention exercise/physical activity by older adults. Prev Med. 2012;55(6):613–7. https://doi.org/10.1016/j.ypmed.2012.10.001.
Humphries B, Duncan MJ, Mummery WK. Prevalence and correlates of resistance training in a regional australian population. Br J Sports Med. 2010;44(9):653–6. https://doi.org/10.1136/bjsm.2008.048975.
Lin CY, Park JH, Hsueh MC, Sun WJ, Liao Y. Prevalence of total physical activity, muscle-strengthening activities, and excessive tv viewing among older adults; and their association with sociodemographic factors. Int J Environ Res Public Health. 2018. https://doi.org/10.3390/ijerph15112499.
Bennie JA, Pedisic Z, van Uffelen JG, Charity MJ, Harvey JT, Banting LK, et al. Pumping iron in australia: prevalence, trends and sociodemographic correlates of muscle strengthening activity participation from a national sample of 195,926 adults. PLoS ONE. 2016;11(4):e0153225. https://doi.org/10.1371/journal.pone.0153225.
Schneider J. Age dependency of oxygen uptake and related parameters in exercise testing: an expert opinion on reference values suitable for adults. Lung. 2013;191(5):449–58. https://doi.org/10.1007/s00408-013-9483-3.
Ekblom-Bak E, Ekblom B, Soderling J, Borjesson M, Blom V, Kallings LV, et al. Sex- and age-specific associations between cardiorespiratory fitness, cvd morbidity and all-cause mortality in in 266.109 adults. Prev Med. 2019;127:105799. https://doi.org/10.1016/j.ypmed.2019.105799.
Trost SG, Owen N, Bauman AE, Sallis JF, Brown W. Correlates of adults’ participation in physical activity: review and update. Med Sci Sports Exerc. 2002;34(12):1996–2001. https://doi.org/10.1249/01.mss.0000038974.76900.92.
Thompson WR. Worldwide survey of fitness trends for 2019. ACSM’s Health Fit J. 2018;22(6):10–7. https://doi.org/10.1249/fit.0000000000000438.
Weston KS, Wisløff U, Coombes JS. High-intensity interval training in patients with lifestyle-induced cardiometabolic disease: a systematic review and meta-analysis. Br J Sports Med. 2014;48(16):1227–34. https://doi.org/10.1136/bjsports-2013-092576.
Gibala MJ, Little JP, Macdonald MJ, Hawley JA. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J Physiol. 2012;590(5):1077–84. https://doi.org/10.1113/jphysiol.2011.224725.
Gibala M. Molecular responses to high-intensity interval exercise. Appl Physiol Nutr Metab (Physiologie appliquee, nutrition et metabolisme). 2009;34(3):428–32. https://doi.org/10.1139/h09-046.
Batacan RB, Duncan MJ, Dalbo VJ, Tucker PS, Fenning AS. Effects of high-intensity interval training on cardiometabolic health: a systematic review and meta-analysis of intervention studies. Br J Sports Med. 2017;51(6):494–503. https://doi.org/10.1136/bjsports-2015-095841.
Cassidy S, Thoma C, Houghton D, Trenell MI. High-intensity interval training: a review of its impact on glucose control and cardiometabolic health. Diabetologia. 2017;60(1):7–23. https://doi.org/10.1007/s00125-016-4106-1.
Wisloff U, Stoylen A, Loennechen JP, Bruvold M, Rognmo O, Haram PM, et al. Superior cardiovascular effect of aerobic interval training versus moderate continuous training in heart failure patients: a randomized study. Circulation. 2007;115(24):3086–94. https://doi.org/10.1161/circulationaha.106.675041.
Rognmo O, Hetland E, Helgerud J, Hoff J, Slordahl SA. High intensity aerobic interval exercise is superior to moderate intensity exercise for increasing aerobic capacity in patients with coronary artery disease. Eur J Cardiovasc Prev Rehabil. 2004;11(3):216–22.
Moholdt TT, Amundsen BH, Rustad LA, Wahba A, Lovo KT, Gullikstad LR, et al. Aerobic interval training versus continuous moderate exercise after coronary artery bypass surgery: a randomized study of cardiovascular effects and quality of life. Am Heart J. 2009;158(6):1031–7. https://doi.org/10.1016/j.ahj.2009.10.003.
Robinson MM, Dasari S, Konopka AR, Johnson ML, Manjunatha S, Esponda RR, et al. Enhanced protein translation underlies improved metabolic and physical adaptations to different exercise training modes in young and old humans. Cell Metab. 2017;25(3):581–92. https://doi.org/10.1016/j.cmet.2017.02.009.
Wyckelsma VL, Levinger I, McKenna MJ. Preservation of skeletal muscle mitochondrial content in older adults: relationship between mitochondria, fibre type and high-intensity exercise training. J Physiol. 2017;595(11):3345–59. https://doi.org/10.1113/jp273950.
Joanisse S, Gillen JB, Bellamy LM, McKay BR, Tarnopolsky MA, Gibala MJ, et al. Evidence for the contribution of muscle stem cells to nonhypertrophic skeletal muscle remodeling in humans. FASEB J. 2013;27(11):4596–605. https://doi.org/10.1096/fj.13-229799.
Blue MNM, Smith-Ryan AE, Trexler ET, Hirsch KR. The effects of high intensity interval training on muscle size and quality in overweight and obese adults. J Sci Med Sport. 2017. https://doi.org/10.1016/j.jsams.2017.06.001.
Hood MS, Little JP, Tarnopolsky MA, Myslik F, Gibala MJ. Low-volume interval training improves muscle oxidative capacity in sedentary adults. Med Sci Sports Exerc. 2011;43(10):1849–56. https://doi.org/10.1249/MSS.0b013e3182199834.
Little JP, Gillen JB, Percival ME, Safdar A, Tarnopolsky MA, Punthakee Z, et al. Low-volume high-intensity interval training reduces hyperglycemia and increases muscle mitochondrial capacity in patients with type 2 diabetes. J Appl Physiol (1985). 2011;111(6):1554–60. https://doi.org/10.1152/japplphysiol.00921.2011.
Tabata I, Nishimura K, Kouzaki M, Hirai Y, Ogita F, Miyachi M, et al. Effects of moderate-intensity endurance and high-intensity intermittent training on anaerobic capacity and VO2max. Med Sci Sports Exerc. 1996;28(10):1327–30.
Joanisse S, McKay BR, Nederveen JP, Scribbans TD, Gurd BJ, Gillen JB, et al. Satellite cell activity, without expansion, after nonhypertrophic stimuli. Am J Physiol Regul Integr Comp Physiol. 2015;309(9):R1101–11. https://doi.org/10.1152/ajpregu.00249.2015.
Scribbans TD, Edgett BA, Vorobej K, Mitchell AS, Joanisse SD, Matusiak JBL, et al. Fibre-specific responses to endurance and low volume high intensity interval training: striking similarities in acute and chronic adaptation. PLoS ONE. 2014;9(6):e98119. https://doi.org/10.1371/journal.pone.0098119.
Burgomaster KA, Howarth KR, Phillips SM, Rakobowchuk M, Macdonald MJ, McGee SL, et al. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J Physiol. 2008;586(1):151–60. https://doi.org/10.1113/jphysiol.2007.142109.
Granata C, Oliveira RS, Little JP, Renner K, Bishop DJ. Training intensity modulates changes in pgc-1alpha and p53 protein content and mitochondrial respiration, but not markers of mitochondrial content in human skeletal muscle. FASEB J. 2016;30(2):959–70. https://doi.org/10.1096/fj.15-276907.
Gibala MJ, Little JP, van Essen M, Wilkin GP, Burgomaster KA, Safdar A, et al. Short-term sprint interval versus traditional endurance training: similar initial adaptations in human skeletal muscle and exercise performance. J Physiol. 2006;575(Pt 3):901–11. https://doi.org/10.1113/jphysiol.2006.112094.
Scalzo RL, Peltonen GL, Binns SE, Shankaran M, Giordano GR, Hartley DA, et al. Greater muscle protein synthesis and mitochondrial biogenesis in males compared with females during sprint interval training. FASEB J. 2014;28(6):2705–14. https://doi.org/10.1096/fj.13-246595.
Metcalfe RS, Babraj JA, Fawkner SG, Vollaard NB. Towards the minimal amount of exercise for improving metabolic health: beneficial effects of reduced-exertion high-intensity interval training. Eur J Appl Physiol. 2012;112(7):2767–75. https://doi.org/10.1007/s00421-011-2254-z.
Metcalfe RS, Tardif N, Thompson D, Vollaard NB. Changes in aerobic capacity and glycaemic control in response to reduced-exertion high-intensity interval training (rehit) are not different between sedentary men and women. Appl Physiol Nutr Metab. 2016;41(11):1117–23. https://doi.org/10.1139/apnm-2016-0253.
Gillen JB, Martin BJ, MacInnis MJ, Skelly LE, Tarnopolsky MA, Gibala MJ. Twelve weeks of sprint interval training improves indices of cardiometabolic health similar to traditional endurance training despite a five-fold lower exercise volume and time commitment. PLoS ONE. 2016;11(4):e0154075. https://doi.org/10.1371/journal.pone.0154075.
Sogaard D, Lund MT, Scheuer CM, Dehlbaek MS, Dideriksen SG, Abildskov CV, et al. High-intensity interval training improves insulin sensitivity in older individuals. Acta Physiol (Oxf). 2017. https://doi.org/10.1111/apha.13009.
Hwang CL, Yoo JK, Kim HK, Hwang MH, Handberg EM, Petersen JW, et al. Novel all-extremity high-intensity interval training improves aerobic fitness, cardiac function and insulin resistance in healthy older adults. Exp Gerontol. 2016;82:112–9. https://doi.org/10.1016/j.exger.2016.06.009.
Mejías-Peña Y, Rodriguez-Miguelez P, Fernandez-Gonzalo R, Martínez-Flórez S, Almar M, de Paz JA, et al. Effects of aerobic training on markers of autophagy in the elderly. Age (Dordr). 2016;38(2):33. https://doi.org/10.1007/s11357-016-9897-y.
Stensvold D, Viken H, Steinshamn SL, Dalen H, Støylen A, Loennechen JP, et al. Effect of exercise training for five years on all cause mortality in older adults—the generation 100 study: randomised controlled trial. BMJ. 2020;371:m3485. https://doi.org/10.1136/bmj.m3485.
Kovacevic A, Fenesi B, Paolucci E, Heisz JJ. The effects of aerobic exercise intensity on memory in older adults. Appl Physiol Nutr Metab. 2020;45(6):591–600. https://doi.org/10.1139/apnm-2019-0495.
Kim HK, Hwang CL, Yoo JK, Hwang MH, Handberg EM, Petersen JW, et al. All-extremity exercise training improves arterial stiffness in older adults. Med Sci Sports Exerc. 2017;49(7):1404–11. https://doi.org/10.1249/mss.0000000000001229.
Oliveira BRR, Santos TM, Kilpatrick M, Pires FO, Deslandes AC. Affective and enjoyment responses in high intensity interval training and continuous training: a systematic review and meta-analysis. PLoS ONE. 2018;13(6):e0197124. https://doi.org/10.1371/journal.pone.0197124.
Martinez-Valdes E, Falla D, Negro F, Mayer F, Farina D. Differential motor unit changes after endurance or high-intensity interval training. Med Sci Sports Exerc. 2017;49(6):1126–36. https://doi.org/10.1249/mss.0000000000001209.
Martinez-Valdes E, Farina D, Negro F, Del Vecchio A, Falla D. Early motor unit conduction velocity changes to high-intensity interval training versus continuous training. Med Sci Sports Exerc. 2018;50(11):2339–50. https://doi.org/10.1249/mss.0000000000001705.
Krustrup P, Soderlund K, Mohr M, Gonzalez-Alonso J, Bangsbo J. Recruitment of fibre types and quadriceps muscle portions during repeated, intense knee-extensor exercise in humans. Pflugers Arch. 2004;449(1):56–65. https://doi.org/10.1007/s00424-004-1304-3.
Edgett BA, Foster WS, Hankinson PB, Simpson CA, Little JP, Graham RB, et al. Dissociation of increases in pgc-1alpha and its regulators from exercise intensity and muscle activation following acute exercise. PLoS ONE. 2013;8(8):e71623. https://doi.org/10.1371/journal.pone.0071623.
Folland JP, Williams AG. The adaptations to strength training : morphological and neurological contributions to increased strength. Sports Med. 2007;37(2):145–68.
Duchateau J, Semmler JG, Enoka RM. Training adaptations in the behavior of human motor units. J Appl Physiol (1985). 2006;101(6):1766–75. https://doi.org/10.1152/japplphysiol.00543.2006.
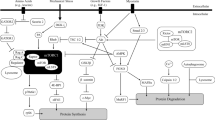
Camera DM, Smiles WJ, Hawley JA. Exercise-induced skeletal muscle signaling pathways and human athletic performance. Free Radic Biol Med. 2016;98:131–43. https://doi.org/10.1016/j.freeradbiomed.2016.02.007.
Rundqvist HC, Montelius A, Osterlund T, Norman B, Esbjornsson M, Jansson E. Acute sprint exercise transcriptome in human skeletal muscle. PLoS ONE. 2019;14(10):e0223024. https://doi.org/10.1371/journal.pone.0223024.
Miyamoto-Mikami E, Tsuji K, Horii N, Hasegawa N, Fujie S, Homma T, et al. Gene expression profile of muscle adaptation to high-intensity intermittent exercise training in young men. Sci Rep. 2018;8(1):16811. https://doi.org/10.1038/s41598-018-35115-x.
Dutka TL, Lamboley CR, McKenna MJ, Murphy RM, Lamb GD. Effects of carnosine on contractile apparatus Ca(2)(+) sensitivity and sarcoplasmic reticulum Ca(2)(+) release in human skeletal muscle fibers. J Appl Physiol. 2012;112(5):728–36. https://doi.org/10.1152/japplphysiol.01331.2011.
Kamm KE, Stull JT. Dedicated myosin light chain kinases with diverse cellular functions. J Biol Chem. 2001;276(7):4527–30. https://doi.org/10.1074/jbc.R000028200.
Gibala MJ, Hawley JA. Sprinting toward fitness. Cell Metab. 2017;25(5):988–90. https://doi.org/10.1016/j.cmet.2017.04.030.
Goody MF, Sher RB, Henry CA. Hanging on for the ride: adhesion to the extracellular matrix mediates cellular responses in skeletal muscle morphogenesis and disease. Dev Biol. 2015;401(1):75–91. https://doi.org/10.1016/j.ydbio.2015.01.002.
Hjorth M, Norheim F, Meen AJ, Pourteymour S, Lee S, Holen T, et al. The effect of acute and long-term physical activity on extracellular matrix and serglycin in human skeletal muscle. Physiol Rep. 2015. https://doi.org/10.14814/phy2.12473.
Wilkinson SB, Phillips SM, Atherton PJ, Patel R, Yarasheski KE, Tarnopolsky MA, et al. Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J Physiol. 2008;586(15):3701–17. https://doi.org/10.1113/jphysiol.2008.153916.
Dreyer HC, Fujita S, Glynn EL, Drummond MJ, Volpi E, Rasmussen BB. Resistance exercise increases leg muscle protein synthesis and mtor signalling independent of sex. Acta Physiol (Oxf). 2010;199(1):71–81. https://doi.org/10.1111/j.1748-1716.2010.02074.x.
Short KR, Vittone JL, Bigelow ML, Proctor DN, Nair KS. Age and aerobic exercise training effects on whole body and muscle protein metabolism. Am J Physiol Endocrinol Metab. 2004;286(1):E92-101. https://doi.org/10.1152/ajpendo.00366.2003.
Bell KE, Seguin C, Parise G, Baker SK, Phillips SM. Day-to-day changes in muscle protein synthesis in recovery from resistance, aerobic, and high-intensity interval exercise in older men. J Gerontol A Biol Sci Med Sci. 2015;70(8):1024–9. https://doi.org/10.1093/gerona/glu313.
Brook MS, Wilkinson DJ, Mitchell WK, Lund JN, Szewczyk NJ, Greenhaff PL, et al. Skeletal muscle hypertrophy adaptations predominate in the early stages of resistance exercise training, matching deuterium oxide-derived measures of muscle protein synthesis and mechanistic target of rapamycin complex 1 signaling. FASEB J. 2015;29(11):4485–96. https://doi.org/10.1096/fj.15-273755.
Mitchell CJ, Churchward-Venne TA, Parise G. Acute post-exercise myofibrillar protein synthesis is not correlated with resistance training-induced muscle hypertrophy in young men. PLoS ONE. 2014. https://doi.org/10.1371/journal.pone.0089431.
Mayhew DL, Kim JS, Cross JM, Ferrando AA, Bamman MM. Translational signaling responses preceding resistance training-mediated myofiber hypertrophy in young and old humans. J Appl Physiol (1985). 2009;107(5):1655–62. https://doi.org/10.1152/japplphysiol.91234.2008.
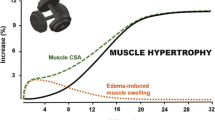
Damas F, Phillips SM, Libardi CA, Vechin FC, Lixandrao ME, Jannig PR, et al. Resistance training-induced changes in integrated myofibrillar protein synthesis are related to hypertrophy only after attenuation of muscle damage. J Physiol. 2016;594(18):5209–22. https://doi.org/10.1113/jp272472.
Damas F, Phillips SM, Lixandrao ME, Vechin FC, Libardi CA, Roschel H, et al. Early resistance training-induced increases in muscle cross-sectional area are concomitant with edema-induced muscle swelling. Eur J Appl Physiol. 2016;116(1):49–56. https://doi.org/10.1007/s00421-015-3243-4.
Joanisse S, Snijders T, Nederveen JP, Parise G. The impact of aerobic exercise on the muscle stem cell response. Exerc Sport Sci Rev. 2018;46(3):180–7. https://doi.org/10.1249/jes.0000000000000153.
Nederveen JP, Joanisse S, Seguin CM, Bell KE, Baker SK, Phillips SM, et al. The effect of exercise mode on the acute response of satellite cells in old men. Acta Physiol (Oxf). 2015;215(4):177–90. https://doi.org/10.1111/apha.12601.
Nederveen JP, Joanisse S, Snijders T, Ivankovic V, Baker SK, Phillips SM, et al. Skeletal muscle satellite cells are located at a closer proximity to capillaries in healthy young compared with older men. J Cachexia Sarcopenia Muscle. 2016;7(5):547–54. https://doi.org/10.1002/jcsm.12105.
Khacho M, Slack RS. Mitochondrial activity in the regulation of stem cell self-renewal and differentiation. Curr Opin Cell Biol. 2017;49:1–8. https://doi.org/10.1016/j.ceb.2017.11.003.
Abreu P, Kowaltowski AJ. Satellite cell self-renewal in endurance exercise is mediated by inhibition of mitochondrial oxygen consumption. J Cachexia Sarcopenia Muscle. 2020. https://doi.org/10.1002/jcsm.12601.
Charifi N, Kadi F, Feasson L, Denis C. Effects of endurance training on satellite cell frequency in skeletal muscle of old men. Muscle Nerve. 2003;28(1):87–92. https://doi.org/10.1002/mus.10394.
Verney J, Kadi F, Charifi N, Feasson L, Saafi MA, Castells J, et al. Effects of combined lower body endurance and upper body resistance training on the satellite cell pool in elderly subjects. Muscle Nerve. 2008;38(3):1147–54. https://doi.org/10.1002/mus.21054.
Snijders T, Nederveen JP, Bell KE, Lau SW, Mazara N, Kumbhare DA, et al. Prolonged exercise training improves the acute type ii muscle fibre satellite cell response in healthy older men. J Physiol. 2019;597(1):105–19. https://doi.org/10.1113/jp276260.
Sultana RN, Sabag A, Keating SE, Johnson NA. The effect of low-volume high-intensity interval training on body composition and cardiorespiratory fitness: a systematic review and meta-analysis. Sports Med. 2019. https://doi.org/10.1007/s40279-019-01167-w.
Bagley L, Slevin M, Bradburn S, Liu D, Murgatroyd C, Morrissey G, et al. Sex differences in the effects of 12 weeks sprint interval training on body fat mass and the rates of fatty acid oxidation and VO2max during exercise. BMJ Open Sport Exerc Med. 2016;2(1):e000056. https://doi.org/10.1136/bmjsem-2015-000056.
Bruseghini P, Calabria E, Tam E, Milanese C, Oliboni E, Pezzato A, et al. Effects of eight weeks of aerobic interval training and of isoinertial resistance training on risk factors of cardiometabolic diseases and exercise capacity in healthy elderly subjects. Oncotarget. 2015;6(19):16998–7015. https://doi.org/10.18632/oncotarget.4031.
Cassidy S, Thoma C, Hallsworth K, Parikh J, Hollingsworth KG, Taylor R, et al. High intensity intermittent exercise improves cardiac structure and function and reduces liver fat in patients with type 2 diabetes: a randomised controlled trial. Diabetologia. 2016;59(1):56–66. https://doi.org/10.1007/s00125-015-3741-2.
Dohlmann TL, Hindso M, Dela F, Helge JW, Larsen S. High-intensity interval training changes mitochondrial respiratory capacity differently in adipose tissue and skeletal muscle. Physiol Rep. 2018;6(18):e13857. https://doi.org/10.14814/phy2.13857.
Gahreman D, Heydari M, Boutcher Y, Freund J, Boutcher S. The effect of green tea ingestion and interval sprinting exercise on the body composition of overweight males: a randomized trial. Nutrients. 2016. https://doi.org/10.3390/nu8080510.
Gillen JB, Percival ME, Ludzki A, Tarnopolsky MA, Gibala MJ. Interval training in the fed or fasted state improves body composition and muscle oxidative capacity in overweight women. Obesity (Silver Spring). 2013;21(11):2249–55. https://doi.org/10.1002/oby.20379.
Heydari M, Freund J, Boutcher SH. The effect of high-intensity intermittent exercise on body composition of overweight young males. J Obes. 2012;2012:480467. https://doi.org/10.1155/2012/480467.
Kong Z, Sun S, Liu M, Shi Q. Short-term high-intensity interval training on body composition and blood glucose in overweight and obese young women. J Diabetes Res. 2016;2016:4073618. https://doi.org/10.1155/2016/4073618.
Maillard F, Rousset S, Pereira B, Traore A, de Pradel Del Amaze P, Boirie Y, et al. High-intensity interval training reduces abdominal fat mass in postmenopausal women with type 2 diabetes. Diabetes Metab. 2016;42(6):433–41. https://doi.org/10.1016/j.diabet.2016.07.031.
Sawyer BJ, Tucker WJ, Bhammar DM, Ryder JR, Sweazea KL, Gaesser GA. Effects of high-intensity interval training and moderate-intensity continuous training on endothelial function and cardiometabolic risk markers in obese adults. J Appl Physiol (1985). 2016;121(1):279–88. https://doi.org/10.1152/japplphysiol.00024.2016.
Sculthorpe NF, Herbert P, Grace F. One session of high-intensity interval training (hiit) every 5 days, improves muscle power but not static balance in lifelong sedentary ageing men: a randomized controlled trial. Medicine (Baltimore). 2017;96(6):e6040. https://doi.org/10.1097/md.0000000000006040.
Trapp EG, Chisholm DJ, Freund J, Boutcher SH. The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. Int J Obes (Lond). 2008;32(4):684–91. https://doi.org/10.1038/sj.ijo.0803781.
Ziemann E, Grzywacz T, Luszczyk M, Laskowski R, Olek RA, Gibson AL. Aerobic and anaerobic changes with high-intensity interval training in active college-aged men. J Strength Cond Res. 2011;25(4):1104–12. https://doi.org/10.1519/JSC.0b013e3181d09ec9.
Ravnholt T, Tybirk J, Jorgensen NR, Bangsbo J. High-intensity intermittent “5-10-15” running reduces body fat, and increases lean body mass, bone mineral density, and performance in untrained subjects. Eur J Appl Physiol. 2018;118(6):1221–30. https://doi.org/10.1007/s00421-018-3851-x.
Stensvold D, Tjonna AE, Skaug EA, Aspenes S, Stolen T, Wisloff U, et al. Strength training versus aerobic interval training to modify risk factors of metabolic syndrome. J Appl Physiol (1985). 2010;108(4):804–10. https://doi.org/10.1152/japplphysiol.00996.2009.
Brown EC, Hew-Butler T, Marks CRC, Butcher SJ, Choi MD. The impact of different high-intensity interval training protocols on body composition and physical fitness in healthy young adult females. Biores Open Access. 2018;7(1):177–85. https://doi.org/10.1089/biores.2018.0032.
Osawa Y, Azuma K, Tabata S, Katsukawa F, Ishida H, Oguma Y, et al. Effects of 16-week high-intensity interval training using upper and lower body ergometers on aerobic fitness and morphological changes in healthy men: a preliminary study. Open Access J Sports Med. 2014;5:257–65. https://doi.org/10.2147/oajsm.s68932.
Fex A, Leduc-Gaudet JP, Filion ME, Karelis AD, Aubertin-Leheudre M. Effect of elliptical high intensity interval training on metabolic risk factor in pre- and type 2 diabetes patients: a pilot study. J Phys Act Health. 2015;12(7):942–6. https://doi.org/10.1123/jpah.2014-0123.
Macpherson RE, Hazell TJ, Olver TD, Paterson DH, Lemon PW. Run sprint interval training improves aerobic performance but not maximal cardiac output. Med Sci Sports Exerc. 2011;43(1):115–22. https://doi.org/10.1249/MSS.0b013e3181e5eacd.
Matsuo T, Saotome K, Seino S, Eto M, Shimojo N, Matsushita A, et al. Low-volume, high-intensity, aerobic interval exercise for sedentary adults: VO(2)max, cardiac mass, and heart rate recovery. Eur J Appl Physiol. 2014;114(9):1963–72. https://doi.org/10.1007/s00421-014-2917-7.
Nybo L, Sundstrup E, Jakobsen MD, Mohr M, Hornstrup T, Simonsen L, et al. High-intensity training versus traditional exercise interventions for promoting health. Med Sci Sports Exerc. 2010;42(10):1951–8. https://doi.org/10.1249/MSS.0b013e3181d99203.
Toombs RJ, Ducher G, Shepherd JA, De Souza MJ. The impact of recent technological advances on the trueness and precision of dxa to assess body composition. Obesity (Silver Spring). 2012;20(1):30–9. https://doi.org/10.1038/oby.2011.211.
Haun CT, Vann CG, Roberts BM, Vigotsky AD, Schoenfeld BJ, Roberts MD. A critical evaluation of the biological construct skeletal muscle hypertrophy: Size matters but so does the measurement. Front Physiol. 2019. https://doi.org/10.3389/fphys.2019.00247.
Nana A, Slater GJ, Stewart AD, Burke LM. Methodology review: Using dual-energy X-ray absorptiometry (dxa) for the assessment of body composition in athletes and active people. Int J Sport Nutr Exerc Metab. 2015;25(2):198–215. https://doi.org/10.1123/ijsnem.2013-0228.
Tavoian D, Ampomah K, Amano S, Law TD, Clark BC. Changes in dxa-derived lean mass and mri-derived cross-sectional area of the thigh are modestly associated. Sci Rep. 2019;9(1):10028. https://doi.org/10.1038/s41598-019-46428-w.
Mitsiopoulos N, Baumgartner RN, Heymsfield SB, Lyons W, Gallagher D, Ross R. Cadaver validation of skeletal muscle measurement by magnetic resonance imaging and computerized tomography. J Appl Physiol (1985). 1998;85(1):115–22. https://doi.org/10.1152/jappl.1998.85.1.115.
Estes RR, Malinowski A, Piacentini M, Thrush D, Salley E, Losey C, et al. The effect of high intensity interval run training on cross-sectional area of the vastus lateralis in untrained college students. Int J Exerc Sci. 2017;10(1):137–45.
Hawley JA, Burke LM, Phillips SM, Spriet LL. Nutritional modulation of training-induced skeletal muscle adaptations. J Appl Physiol (1985). 2011;110(3):834–45. https://doi.org/10.1152/japplphysiol.00949.2010.
Moore DR, Churchward-Venne TA, Witard O. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J Gerontol A Biol Sci Med Sci. 2015. https://doi.org/10.1093/gerona/glu103.
Wilkinson DJ, Hossain T, Hill DS, Phillips BE, Crossland H, Williams J, et al. Effects of leucine and its metabolite beta-hydroxy-beta-methylbutyrate on human skeletal muscle protein metabolism. J Physiol. 2013;591(11):2911–23. https://doi.org/10.1113/jphysiol.2013.253203.
Dai JM, Yu MX, Shen ZY, Guo CY, Zhuang SQ, Qiu XS. Leucine promotes proliferation and differentiation of primary preterm rat satellite cells in part through mtorc1 signaling pathway. Nutrients. 2015;7(5):3387–400. https://doi.org/10.3390/nu7053387.
Biolo G, Tipton KD, Klein S, Wolfe RR. An abundant supply of amino acids enhances the metabolic effect of exercise on muscle protein. Am J Physiol. 1997;273(1 Pt 1):E122–9.
Vliet SV, Beals JW, Martinez IG, Skinner SK, Burd NA. Achieving optimal post-exercise muscle protein remodeling in physically active adults through whole food consumption. Nutrients. 2018. https://doi.org/10.3390/nu10020224.
Phillips SM. The impact of protein quality on the promotion of resistance exercise-induced changes in muscle mass. Nutr Metab (Lond). 2016;13:64. https://doi.org/10.1186/s12986-016-0124-8.
Morton RW, Murphy KT, McKellar SR, Schoenfeld BJ, Henselmans M, Helms E, et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br J Sports Med. 2017. https://doi.org/10.1136/bjsports-2017-097608.
Cermak NM, Res PT, de Groot LC, Saris WH, van Loon LJ. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: a meta-analysis. Am J Clin Nutr. 2012;96(6):1454–64. https://doi.org/10.3945/ajcn.112.037556.
Timmerman KL, Dhanani S, Glynn EL, Fry CS, Drummond MJ, Jennings K, et al. A moderate acute increase in physical activity enhances nutritive flow and the muscle protein anabolic response to mixed nutrient intake in older adults. Am J Clin Nutr. 2012;95(6):1403–12. https://doi.org/10.3945/ajcn.111.020800.
Breen L, Philp A, Witard OC, Jackman SR, Selby A, Smith K, et al. The influence of carbohydrate-protein co-ingestion following endurance exercise on myofibrillar and mitochondrial protein synthesis. J Physiol. 2011;589(Pt 16):4011–25. https://doi.org/10.1113/jphysiol.2011.211888.
Rowlands DS, Nelson AR, Phillips SM, Faulkner JA, Clarke J, Burd NA, et al. Protein-leucine fed dose effects on muscle protein synthesis after endurance exercise. Med Sci Sports Exerc. 2015;47(3):547–55. https://doi.org/10.1249/mss.0000000000000447.
Harber MP, Konopka AR, Jemiolo B, Trappe SW, Trappe TA, Reidy PT. Muscle protein synthesis and gene expression during recovery from aerobic exercise in the fasted and fed states. Am J Physiol Regul Integr Comp Physiol. 2010;299(5):R1254–62. https://doi.org/10.1152/ajpregu.00348.2010.
Coffey VG, Moore DR, Burd NA, Rerecich T, Stellingwerff T, Garnham AP, et al. Nutrient provision increases signalling and protein synthesis in human skeletal muscle after repeated sprints. Eur J Appl Physiol. 2011;111(7):1473–83. https://doi.org/10.1007/s00421-010-1768-0.
Mitchell CJ, Churchward-Venne TA, Cameron-Smith D, Phillips SM. What is the relationship between the acute muscle protein synthesis response and changes in muscle mass? J Appl Physiol (1985). 2015;118(4):495–7. https://doi.org/10.1152/japplphysiol.00609.2014.
Rundqvist HC, Esbjornsson M, Rooyackers O, Osterlund T, Moberg M, Apro W, et al. Influence of nutrient ingestion on amino acid transporters and protein synthesis in human skeletal muscle after sprint exercise. J Appl Physiol (1985). 2017. https://doi.org/10.1152/japplphysiol.00244.2017.
Leuchtmann AB, Mueller SM, Aguayo D, Petersen JA, Ligon-Auer M, Flück M, et al. Resistance training preserves high-intensity interval training induced improvements in skeletal muscle capillarization of healthy old men: a randomized controlled trial. Sci Rep. 2020;10(1):6578. https://doi.org/10.1038/s41598-020-63490-x.
Gaffney KA, Lucero A, Stoner L, Faulkner J, Whitfield P, Krebs J, et al. Nil whey protein effect on glycemic control after intense mixed-mode training in type 2 diabetes. Med Sci Sports Exerc. 2018;50(1):11–7. https://doi.org/10.1249/mss.0000000000001404.
Hickson RC. Interference of strength development by simultaneously training for strength and endurance. Eur J Appl Physiol Occup Physiol. 1980;45(2–3):255–63.
Esbjörnsson M, Rundqvist HC, Mascher H, Österlund T, Rooyackers O, Blomstrand E, et al. Sprint exercise enhances skeletal muscle p70s6k phosphorylation and more so in women than in men. Acta Physiol (Oxf). 2012;205(3):411–22. https://doi.org/10.1111/j.1748-1716.2012.02404.x.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article.
Conflict of interest
Marcus Callahan, Evelyn Parr, John Hawley and Donny Camera declare that they have no conflicts of interest relevant to the content of this review.
Authorship contributions
MC wrote the first draft of the manuscript. MC, EBP, JH and DMC revised the original manuscript. All authors read and approved the final manuscript.
Rights and permissions
About this article
Cite this article
Callahan, M.J., Parr, E.B., Hawley, J.A. et al. Can High-Intensity Interval Training Promote Skeletal Muscle Anabolism?. Sports Med 51, 405–421 (2021). https://doi.org/10.1007/s40279-020-01397-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-020-01397-3