Abstract
Introduction
Heart failure is the leading cause of death in patients with diabetes. No treatment currently exists to specifically protect these patients at risk of developing cardiovascular complications. Accelerated oxidative stress-induced tissue damage due to persistent hyperglycemia is one of the major factors implicated in deteriorated cardiac function within a diabetic state. N-acetyl cysteine (NAC), through its enhanced capacity to endogenously synthesize glutathione, a potent antioxidant, has displayed abundant health-promoting properties and has a favorable safety profile.
Objective
An increasing number of experimental studies have reported on the strong ameliorative properties of NAC. We systematically reviewed the data on the cardioprotective potential of this compound to provide an informative summary.
Methods
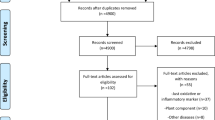
Two independent reviewers systematically searched major databases, including PubMed, Cochrane Library, Google scholar, and Embase for available studies reporting on the ameliorative effects of NAC as a monotherapy or in combination with other therapies against diabetes-associated cardiovascular complications. We used the ARRIVE and JBI appraisal guidelines to assess the quality of individual studies included in the review. A meta-analysis could not be performed because the included studies were heterogeneous and data from randomized clinical trials were unavailable.
Results
Most studies support the ameliorative potential of NAC against a number of diabetes-associated complications, including oxidative stress. We discuss future prospects, such as identification of additional molecular mechanisms implicated in diabetes-induced cardiac damage, and highlight limitations, such as insufficient studies reporting on the comparative effect of NAC with common glucose-lowering therapies. Information on the comparative analysis of NAC, in terms of dose selection, administration mode, and its effect on different cardiovascular-related markers is important for translation into clinical studies.
Conclusions
NAC exhibits strong potential for the protection of the diabetic heart at risk of myocardial infarction through inhibition of oxidative stress. The effect of NAC in preventing both ischemia and non-ischemic-associated cardiac damage is also of interest. Consistency in dose selection in most studies reported remains important in dose translation for clinical relevance.




Similar content being viewed by others
Abbreviations
- AKT:
-
Protein kinase B
- AMPK:
-
5′ AMP-activated protein kinase
- Bak:
-
Bcl-2 antagonist/killer 1
- Bax:
-
Bcl-2-like protein 4
- Bcl-2:
-
B-cell lymphoma 2
- Brg1:
-
Brahma-related gene
- COX2:
-
Cyclooxygenase 2
- CTGF:
-
Connective tissue growth factor
- eNOS:
-
Endothelial nitric oxide synthase
- ERK1/2:
-
Extracellular signal-regulated kinase 1 and 2
- FoxO1:
-
Forkhead box protein O1
- GSH:
-
Glutathione
- HO-1:
-
Heme oxygenase 1
- IL-6:
-
Interleukin 6
- iNOS:
-
Inducible nitric oxide synthase
- JBI:
-
Joanna Briggs Institute
- JNK:
-
c-Jun N-terminal kinases
- L-NAME:
-
N-ω-nitro-L-arginine methyl ester
- NAC:
-
n-acetyl cysteine
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NF-kB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- p38 MAPK:
-
p38 mitogen-activated protein kinases
- p53:
-
Tumor protein p53
- PERK:
-
Protein kinase RNA-like endoplasmic reticulum kinase
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- STAT3:
-
Signal transducer and activator of transcription 3
- STZ:
-
Streptozotocin
- UCF-101:
-
5-[5-(2-Nitrophenyl) furfuryliodine]-1,3-diphenyl-2-thiobarbituric acid
References
Islam SM, Purnat TD, Phuong NT, Mwingira U, Schacht K, Froschl G. Non-communicable diseases (NCDs) in developing countries: a symposium report. Glob. Health. 2014;10:81.
WHO. World health statistics 2012. World Health Organization (WHO), 2012. http://apps.who.int/iris/bitstream/10665/44844/1/9789241564441_eng.pdf?ua=1. Accessed 20 Oct 2017.
WHO. The top 10 causes of death. 2017. http://www.who.int/mediacentre/factsheets/fs310/en/. Accessed 20 Oct 2017.
American Heart Association. Cardiovascular disease and diabetes. http://www.heart.org/HEARTORG/Conditions/More/Diabetes/WhyDiabetesMatters/Cardiovascular-Disease-Diabetes_UCM_313865_Article.jsp/#.WhVgWEqWaUk. Accessed 20 Oct 2017.
Sharma MD, Farmer JA, Garber A. Type 2 diabetes and cardiovascular risk factors. Curr Med Res Opin. 2011;27(Suppl 3):1–5.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107(9):1058–70.
Dludla PV, Joubert E, Muller CJF, Louw J, Johnson R. Hyperglycemia-induced oxidative stress and heart disease-cardioprotective effects of rooibos flavonoids and phenylpyruvic acid-2-O-beta-d-glucoside. Nutr Metab (Lond). 2017;14:45.
Mannucci E, Dicembrini I, Lauria A, Pozzilli P. Is glucose control important for prevention of cardiovascular disease in diabetes? Diabetes Care. 2013;36(Suppl 2):S259–63.
Ansley DM, Wang B. Oxidative stress and myocardial injury in the diabetic heart. J Pathol. 2013;229(2):232–41.
Kobylecki CJ, Afzal S, Nordestgaard BG. Genetically low antioxidant protection and risk of cardiovascular disease and heart failure in diabetic subjects. EBioMedicine. 2015;2(12):2010–5.
Sun X, Chen RC, Yang ZH, Sun GB, Wang M, Ma XJ, et al. Taxifolin prevents diabetic cardiomyopathy in vivo and in vitro by inhibition of oxidative stress and cell apoptosis. Food Chem Toxicol. 2014;63:221–32.
Mohammadshahi M, Haidari F, Soufi FG. Chronic resveratrol administration improves diabetic cardiomyopathy in part by reducing oxidative stress. Cardiol J. 2014;21(1):39–46.
Bai Y, Cui W, Xin Y, Miao X, Barati MT, Zhang C, et al. Prevention by sulforaphane of diabetic cardiomyopathy is associated with up-regulation of Nrf2 expression and transcription activation. J Mol Cell Cardiol. 2013;57:82–95.
Huynh K, Bernardo BC, McMullen JR, Ritchie RH. Diabetic cardiomyopathy: mechanisms and new treatment strategies targeting antioxidant signaling pathways. Pharmacol Ther. 2014;142(3):375–415.
Yin M, van der Horst IC, van Melle JP, Qian C, van Gilst WH, Sillje HH, et al. Metformin improves cardiac function in a nondiabetic rat model of post-MI heart failure. Am J Physiol Heart Circ Physiol. 2011;301(2):H459–68.
Zhang E, Guo Q, Gao H, Xu R, Teng S, Wu Y. Metformin and resveratrol inhibited high glucose-induced metabolic memory of endothelial senescence through SIRT1/p300/p53/p21 pathway. PloS One. 2015;10(12):e0143814.
Camuglia AC, Maeder MT, Starr J, Farrington C, Kaye DM. Impact of N-acetylcysteine on endothelial function, B-type natriuretic peptide and renal function in patients with the cardiorenal syndrome: a pilot cross over randomised controlled trial. Heart Lung Circ. 2013;22(4):256–9.
Giam B, Chu PY, Kuruppu S, Smith AI, Horlock D, Kiriazis H, et al. N-acetylcysteine attenuates the development of cardiac fibrosis and remodeling in a mouse model of heart failure. Physiol Rep. 2016;4(7):e12757.
Suddarth SB. Acetylcysteine, a new and effective mucolytic agent. Bull Geisinger. 1963;15:65–9.
Asevedo E, Mendes AC, Berk M, Brietzke E. Systematic review of N-acetylcysteine in the treatment of addictions. Rev Bras Psiquiatr. 2014;36(2):168–75.
Heard KJ. Acetylcysteine for acetaminophen poisoning. N Engl J Med. 2008;359(3):285–92.
Rasi Hashemi S, Noshad H, Tabrizi A, Mobasseri M, Tayebi Khosroshahi H, Heydarnejad M, et al. Angiotensin receptor blocker and N-acetyl cysteine for reduction of proteinuria in patients with type 2 diabetes mellitus. Iran J Kidney Dis. 2012;6(1):39–43.
Schmitt B, Vicenzi M, Garrel C, Denis FM. Effects of N-acetylcysteine, oral glutathione (GSH) and a novel sublingual form of GSH on oxidative stress markers: a comparative crossover study. Redox Biol. 2015;6:198–205.
Dludla PV, Muller CJ, Louw J, Joubert E, Salie R, Opoku AR, et al. The cardioprotective effect of an aqueous extract of fermented rooibos (Aspalathus linearis) on cultured cardiomyocytes derived from diabetic rats. Phytomedicine. 2014;21(5):595–601.
Girouard H, Denault C, Chulak C, de Champlain J. Treatment by N-acetylcysteine and melatonin increases cardiac baroreflex and improves antioxidant reserve. Am J Hypertens. 2004;17(10):947–54.
Haleagrahara N, Julian V, Chakravarthi S. N-acetylcysteine offers cardioprotection by decreasing cardiac lipid hydroperoxides and 8-isoprostane level in isoproterenol-induced cardiotoxicity in rats. Cardiovasc Toxicol. 2011;11(4):373–81.
Liu C, Lu XZ, Shen MZ, Xing CY, Ma J, Duan YY, et al. N-acetyl cysteine improves the diabetic cardiac function: possible role of fibrosis inhibition. BMC Cardiovasc Disord. 2015;15:84.
Dludla PV, Nkambule BB, Dias SC, Johnson R. Cardioprotective potential of N-acetyl cysteine against hyperglycaemia-induced oxidative damage: a protocol for a systematic review. Syst Rev. 2017;6(1):96.
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8(6):e1000412.
Institute The Joanna Briggs. Joanna Briggs Institute reviewers’ manual. 2016th ed. Australia: The Joanna Briggs Institute; 2016.
Matsuzaki T, Sakanashi M. Comparison of the development of tolerance to nitroglycerin in aortic preparations isolated from non-diabetic and diabetic rats. Heart Vessels. 1992;7(1):1–7.
Fiordaliso F, Bianchi R, Staszewsky L, Cuccovillo I, Doni M, Laragione T, et al. Antioxidant treatment attenuates hyperglycemia-induced cardiomyocyte death in rats. J Mol Cell Cardiol. 2004;37(5):959–68.
Cheng X, Xia Z, Leo JM, Pang CC. The effect of N-acetylcysteine on cardiac contractility to dobutamine in rats with streptozotocin-induced diabetes. Eur J Pharmacol. 2005;519(1–2):118–26.
Nagareddy PR, Xia Z, MacLeod KM, McNeill JH. N-acetylcysteine prevents nitrosative stress-associated depression of blood pressure and heart rate in streptozotocin diabetic rats. J Cardiovasc Pharmacol. 2006;47(4):513–20.
Xia Z, Guo Z, Nagareddy PR, Yuen V, Yeung E, McNeill JH. Antioxidant N-acetylcysteine restores myocardial Mn-SOD activity and attenuates myocardial dysfunction in diabetic rats. Eur J Pharmacol. 2006;544(1–3):118–25.
Xia Z, Nagareddy PR, Guo Z, Zhang W, McNeill JH. Antioxidant N-acetylcysteine restores systemic nitric oxide availability and corrects depressions in arterial blood pressure and heart rate in diabetic rats. Free Radic Res. 2006;40(2):175–84.
Fauconnier J, Andersson DC, Zhang SJ, Lanner JT, Wibom R, Katz A, et al. Effects of palmitate on Ca(2+) handling in adult control and ob/ob cardiomyocytes: impact of mitochondrial reactive oxygen species. Diabetes. 2007;56(4):1136–42.
Li S, Li X, Li YL, Shao CH, Bidasee KR, Rozanski GJ. Insulin regulation of glutathione and contractile phenotype in diabetic rat ventricular myocytes. Am J Physiol Heart Circ Physiol. 2007;292(3):H1619–29.
Guo Z, Xia Z, Jiang J, McNeill JH. Downregulation of NADPH oxidase, antioxidant enzymes, and inflammatory markers in the heart of streptozotocin-induced diabetic rats by N-acetyl-l-cysteine. Am J Physiol Heart Circ Physiol. 2007;292(4):H1728–36.
Iwata K, Nishinaka T, Matsuno K, Kakehi T, Katsuyama M, Ibi M, et al. The activity of aldose reductase is elevated in diabetic mouse heart. J Pharmacol Sci. 2007;103(4):408–16.
Pelletier A, Coderre L. Ketone bodies alter dinitrophenol-induced glucose uptake through AMPK inhibition and oxidative stress generation in adult cardiomyocytes. Am J Physiol Endocrinol Metab. 2007;292(5):E1325–32.
Xia Z, Kuo KH, Nagareddy PR, Wang F, Guo Z, Guo T, et al. N-acetylcysteine attenuates PKCbeta2 overexpression and myocardial hypertrophy in streptozotocin-induced diabetic rats. Cardiovasc Res. 2007;73(4):770–82.
Matejíková J, Kucharska J, Pancza D, Ravingerova T. The effect of antioxidant treatment and NOS inhibition on the incidence of ischemia-induced arrhythmias in the diabetic rat heart. Physiol Res. 2008;57(Suppl 2):S55–60.
Abdel Baky NA, Mohamed AM, Faddah LM. Protective effect of N-acetyl cysteine and/or provitamin A against monosodium glutamate-induced cardiopathy in rats. JPT. 2009;4(5):178–93.
Li Q, Hueckstaedt LK, Ren J. The protease inhibitor UCF-101 ameliorates streptozotocin-induced mouse cardiomyocyte contractile dysfunction in vitro: role of AMP-activated protein kinase. Exp Physiol. 2009;94(9):984–94.
Zou P, Wu LL, Wu D, Fan D, Cui XB, Zhou Y, et al. High glucose increases periostin expression and the related signal pathway in adult rat cardiac fibroblasts. Sheng Li Xue Bao. 2010;62(3):247–54.
Gao S, Yuan K, Shah A, Kim JS, Park WH, Kim SH. Suppression of high pacing-induced ANP secretion by antioxidants in isolated rat atria. Peptides. 2011;32(12):2467–73.
Okazaki T, Otani H, Shimazu T, Yoshioka K, Fujita M, Iwasaka T. Ascorbic acid and N-acetyl cysteine prevent uncoupling of nitric oxide synthase and increase tolerance to ischemia/reperfusion injury in diabetic rat heart. Free Radic Res. 2011;45(10):1173–83.
Ungvari Z, Bailey-Downs L, Gautam T, Jimenez R, Losonczy G, Zhang C, et al. Adaptive induction of NF-E2-related factor-2-driven antioxidant genes in endothelial cells in response to hyperglycemia. Am J Physiol Heart Circ Physiol. 2011;300(4):H1133–40.
Wang J, Wang H, Hao P, Xue L, Wei S, Zhang Y, et al. Inhibition of aldehyde dehydrogenase 2 by oxidative stress is associated with cardiac dysfunction in diabetic rats. Mol Med. 2011;17(3–4):172–9.
Wang T, Qiao S, Lei S, Liu Y, Ng KF, Xu A, et al. N-acetylcysteine and allopurinol synergistically enhance cardiac adiponectin content and reduce myocardial reperfusion injury in diabetic rats. PloS One. 2011;6(8):e23967.
Lei S, Liu Y, Liu H, Yu H, Wang H, Xia Z. Effects of N-acetylcysteine on nicotinamide dinucleotide phosphate oxidase activation and antioxidant status in heart, lung, liver and kidney in streptozotocin-induced diabetic rats. Yonsei Med J. 2012;53(2):294–303.
Liu Y, Lei S, Gao X, Mao X, Wang T, Wong GT, et al. PKCbeta inhibition with ruboxistaurin reduces oxidative stress and attenuates left ventricular hypertrophy and dysfunction in rats with streptozotocin-induced diabetes. Clin Sci (Lond). 2012;122(4):161–73.
Kuo WW, Wang WJ, Tsai CY, Way CL, Hsu HH, Chen LM. Diallyl trisufide (DATS) suppresses high glucose-induced cardiomyocyte apoptosis by inhibiting JNK/NFkappaB signaling via attenuating ROS generation. Int J Cardiol. 2013;168(1):270–80.
Wang T, Mao X, Li H, Qiao S, Xu A, Wang J, et al. N-acetylcysteine and allopurinol up-regulated the Jak/STAT3 and PI3K/Akt pathways via adiponectin and attenuated myocardial postischemic injury in diabetes. Free Radic Biol Med. 2013;63:291–303.
Cicek FA, Toy A, Tuncay E, Can B, Turan B. Beta-blocker timolol alleviates hyperglycemia-induced cardiac damage via inhibition of endoplasmic reticulum stress. J Bioenerg Biomembr. 2014;46(5):377–87.
Neves KB, Nguyen Dinh Cat A, Lopes RA, Rios FJ, Anagnostopoulou A, Lobato NS, et al. Chemerin regulates crosstalk between adipocytes and vascular cells through Nox. Hypertension. 2015;66(3):657–66.
Suzuki H, Kayama Y, Sakamoto M, Iuchi H, Shimizu I, Yoshino T, et al. Arachidonate 12/15-lipoxygenase-induced inflammation and oxidative stress are involved in the development of diabetic cardiomyopathy. Diabetes. 2015;64(2):618–30.
Lin J, Wang T, Li Y, Wang M, Li H. N-acetylcysteine restores sevoflurane postconditioning cardioprotection against myocardial ischemia-reperfusion injury in diabetic rats. J Diabetes Res. 2016;2016:9213034.
Shi S, Guo Y, Lou Y, Li Q, Cai X, Zhong X, et al. Sulfiredoxin involved in the protection of peroxiredoxins against hyperoxidation in the early hyperglycaemia. Exp Cell Res. 2017;352(2):273–80.
Yao Y, Li R, Ma Y, Wang X, Li C, Zhang X, et al. Alpha-lipoic acid increases tolerance of cardiomyoblasts to glucose/glucose oxidase-induced injury via ROS-dependent ERK1/2 activation. Biochim Biophys Acta. 2012;1823(4):920–9.
De Bairros AV, Roehrs M, Ribeiro G, de Freitas F, Moreira AP, Tonello R, Mazzanti C, et al. Treatment with N-acetylcysteine does not alter blood glucose levels and the oxidative stress status in diabetic rats. J Pharm Negative Results. 2013;4(1):5.
Liu ZW, Zhu HT, Chen KL, Dong X, Wei J, Qiu C, et al. Protein kinase RNA-like endoplasmic reticulum kinase (PERK) signaling pathway plays a major role in reactive oxygen species (ROS)-mediated endoplasmic reticulum stress-induced apoptosis in diabetic cardiomyopathy. Cardiovasc Diabetol. 2013;12:158.
Xu J, Lei S, Liu Y, Gao X, Irwin MG, Xia ZY, et al. Antioxidant N-acetylcysteine attenuates the reduction of Brg1 protein expression in the myocardium of type 1 diabetic rats. J Diabetes Res. 2013;2013:716219.
Xu W, Wu W, Chen J, Guo R, Lin J, Liao X, et al. Exogenous hydrogen sulfide protects H9c2 cardiac cells against high glucose-induced injury by inhibiting the activities of the p38 MAPK and ERK1/2 pathways. Int J Mol Med. 2013;32(4):917–25.
Yildirim SS, Akman D, Catalucci D, Turan B. Relationship between downregulation of miRNAs and increase of oxidative stress in the development of diabetic cardiac dysfunction: junctin as a target protein of miR-1. Cell Biochem Biophys. 2013;67(3):1397–408.
Michaelson J, Hariharan V, Huang H. Hyperglycemic and hyperlipidemic conditions alter cardiac cell biomechanical properties. Biophys J. 2014;106(11):2322–9.
Moazzen H, Lu X, Ma NL, Velenosi TJ, Urquhart BL, Wisse LJ, et al. N-acetylcysteine prevents congenital heart defects induced by pregestational diabetes. Cardiovasc Diabetol. 2014;13:46.
Muravyeva M, Baotic I, Bienengraeber M, Lazar J, Bosnjak ZJ, Sedlic F, et al. Cardioprotection during diabetes: the role of mitochondrial DNA. Anesthesiology. 2014;120(4):870–9.
Tuncay E, Okatan EN, Toy A, Turan B. Enhancement of cellular antioxidant-defence preserves diastolic dysfunction via regulation of both diastolic Zn2+ and Ca2+ and prevention of RyR2-leak in hyperglycemic cardiomyocytes. Oxid Med Cell Longev. 2014;2014:290381.
Liu ZW, Wang JK, Qiu C, Guan GC, Liu XH, Li SJ, et al. Matrine pretreatment improves cardiac function in rats with diabetic cardiomyopathy via suppressing ROS/TLR-4 signaling pathway. Acta Pharmacol Sin. 2015;36(3):323–33.
Moazzen H, Lu X, Liu M, Feng Q. Pregestational diabetes induces fetal coronary artery malformation via reactive oxygen species signaling. Diabetes. 2015;64(4):1431–43.
Roussel J, Thireau J, Brenner C, Saint N, Scheuermann V, Lacampagne A, et al. Palmitoyl-carnitine increases RyR2 oxidation and sarcoplasmic reticulum Ca2+ leak in cardiomyocytes: role of adenine nucleotide translocase. Biochim Biophys Acta. 2015;1852(5):749–58.
Ali F, Khan M, Khan SN, Riazuddin S. N-acetyl cysteine protects diabetic mouse derived mesenchymal stem cells from hydrogen-peroxide-induced injury: a novel hypothesis for autologous stem cell transplantation. J Chin Med Assoc. 2016;79(3):122–9.
Su W, Zhang Y, Zhang Q, Xu J, Zhan L, Zhu Q, et al. N-acetylcysteine attenuates myocardial dysfunction and postischemic injury by restoring caveolin-3/eNOS signaling in diabetic rats. Cardiovasc Diabetol. 2016;15(1):146.
Wang Y, Li H, Huang H, Liu S, Mao X, Wang S, et al. Cardioprotection from emulsified isoflurane postconditioning is lost in rats with streptozotocin-induced diabetes due to the impairment of Brg1/Nrf2/STAT3 signalling. Clin Sci (Lond). 2016;130(10):801–12.
Wang S, Wang C. N-acetylcysteine attenuates diabetic myocardial ischemia reperfusion injury through inhibiting excessive autophagy. Mediat Inflamm. 2017;2017:9257291.
Prescott LF, Park J, Ballantyne A, Adriaenssens P, Proudfoot AT. Treatment of paracetamol (acetaminophen) poisoning with N-acetylcysteine. Lancet. 1977;2(8035):432–4.
Kelly GS. Clinical applications of N-acetylcysteine. Altern Med Rev. 1998;3(2):114–27.
Ercal N, Treeratphan P, Hammond TC, Matthews RH, Grannemann NH, Spitz DR. In vivo indices of oxidative stress in lead-exposed C57BL/6 mice are reduced by treatment with meso-2,3-dimercaptosuccinic acid or N-acetylcysteine. Free Radic Biol Med. 1996;21(2):157–61.
Aruoma OI, Halliwell B, Hoey BM, Butler J. The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid. Free Radic Biol Med. 1989;6(6):593–7.
Kerksick C, Willoughby D. The antioxidant role of glutathione and N-acetyl-cysteine supplements and exercise-induced oxidative stress. J Int Soc Sports Nutr. 2005;2:38–44.
Rubler S, Dlugash J, Yuceoglu YZ, Kumral T, Branwood AW, Grishman A. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol. 1972;30(6):595–602.
Aronson D, Edelman ER. Coronary artery disease and diabetes mellitus. Cardiol Clin. 2014;32(3):439–55.
Dandamudi S, Slusser J, Mahoney DW, Redfield MM, Rodeheffer RJ, Chen HH. The prevalence of diabetic cardiomyopathy: a population-based study in Olmsted County, Minnesota. J Card Fail. 2014;20(5):304–9.
Boudina S, Abel ED. Diabetic cardiomyopathy revisited. Circulation. 2007;115(25):3213–23.
Falah RRTW, Shbailat SJ. Combination of metformin and curcumin targets breast cancer in mice by angiogenesis inhibition, immune system modulation and induction of p53 independent apoptosis. Ther Adv Med Oncol. 2017;9(4):235–52.
Johnson R, Dludla P, Joubert E, February F, Mazibuko S, Ghoor S, et al. Aspalathin, a dihydrochalcone C-glucoside, protects H9c2 cardiomyocytes against high glucose induced shifts in substrate preference and apoptosis. Mol Nutr Food Res. 2016;60(4):922–34.
Loscalzo J. N-acetylcysteine potentiates inhibition of platelet aggregation by nitroglycerin. J Clin Invest. 1985;76(2):703–8.
Ungvari Z, Bagi Z, Feher A, Recchia FA, Sonntag WE, Pearson K, et al. Resveratrol confers endothelial protection via activation of the antioxidant transcription factor Nrf2. Am J Physiol Heart Circ Physiol. 2010;299(1):H18–24.
Dludla PV, Muller CJ, Joubert E, Louw J, Essop MF, Gabuza KB, et al. Aspalathin protects the heart against hyperglycemia-induced oxidative damage by up-regulating Nrf2 expression. Molecules. 2017;22(1):129.
Acknowledgements
The authors are indebted to Prof. Charles Shey Wiysonge of Cochrane South Africa for assistance with the development of this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Biomedical Research and Innovation Platform of the South Africa Medical Research Council (SAMRC). P.V. Dludla was partially supported as a Post-Doctoral Fellow by funding from the SAMRC. The SAMRC did not directly participate in the literature search, determination of study eligibility, data analysis, or interpretation or preparation of the manuscript for publication.
Conflict of interest
PVD, SCD, NO, RJ, JL, and BBN have no conflicts of interest that might be relevant to the contents of this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dludla, P.V., Dias, S.C., Obonye, N. et al. A Systematic Review on the Protective Effect of N-Acetyl Cysteine Against Diabetes-Associated Cardiovascular Complications. Am J Cardiovasc Drugs 18, 283–298 (2018). https://doi.org/10.1007/s40256-018-0275-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40256-018-0275-2