Abstract
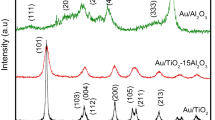
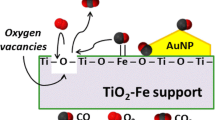
Fe-doped TiO2 supported gold nanoparticles as high-performance CO oxidation catalysts were prepared. XRD data revealed that TiO2 support was in an anatase phase. After calcination at 300 °C, the sample showed nanotube structure, and the size of gold nanoparticles was 3.1 nm. When calcined at 500 °C, most nanotubes broke, and gold nanoparticles grew up to 5.9 nm. XPS spectrum indicated the presence of Fe in the +3 oxidation state. Au/Fe-TiO2(Au: 1.44%, Fe: 1.35%) calcined at 300 °C possessed the best catalytic activity, and it could completely convert CO at 25 °C. The temperature of 100% CO conversion(T100%) of Fe-free catalyst was 40 °C. After the catalysts were stored at room temperature for 7 d, T100% of Au/Fe-TiO2 increased from 25 °C to 30 °C, while T100% of Fe-free catalyst increased from 40 °C to 80 °C. The catalytic activity and storage stability of Au/TiO2 could be improved by Fe-doping. The increase of specific surface area, generation of oxygen vacancies and new adsorption sites, depression of the growth of gold nanoparticles, and strong metal-support interaction were responsible for the promoting effect of iron on the catalytic performance of Au/TiO2 for CO oxidation.
Similar content being viewed by others
References
Ma Z., Dai S., Nano Res., 2011, 4, 3
Lakshmanan P., Park J. E., Park E. D., Catal. Surv. Asia, 2014, 18, 75
Roy P., Berger S., Schmuki P., Angew. Chem. Int. Ed., 2011, 50, 2904
Méndez-Cruz M., Ramírez-Solísa J., Zanella R., Catal. Today, 2011, 166, 172
Zhao H., Zhang P., Wang Y. D., Huang W. P., Zhang S. M., J. Sol-Gel Sci. Technol., 2014, 71, 406
Shou M., Takekawa H., Ju D., Hagiwara T., Lu D., Tanaka K., Catal. Lett., 2006, 108, 119
Moreau F., Bond G. C., Catal. Today, 2006, 114, 362
Yang Y. F., Sangeetha P., Chen Y. W., Ind. Eng. Chem. Res., 2009, 48, 10402
Parida K. M., Sahua N., Mohapatra P., Scurrell M. S., J. Mol. Catal. A: Chem., 2010, 319, 92
Yu S., Yun H. J., Lee D. M., Yi J., J. Mater. Chem., 2012, 22, 12629
Mariana H. R., Roberto C. S., Rodolfo Z., Vicente R. G., Facundo R., Catal. Lett., 2018 148, 383
Zhang P., Guo J. L., Zhao P., Zhu B. L., Huang W. P., Zhang S. M., RSC Adv., 2015, 5, 11989
Tong T. Z., Zhang J. L., Tian B. Z., Chen F., He D. N., J. Hazard Mater., 2008, 155, 572
Wu Y. M., Zhang J. L., Xiao L., Chen F., Appl. Catal. B: Envir., 2009, 88, 525
Xu Z. H., Yu J. G., Nanoscale, 2011, 3, 3138
Deng L. X., Wang S. R., Liu D. Y., Zhu B. L., Huang W. P., Wu S. H., Zhang S. M., Catal. Lett., 2009, 129, 513
Gao Y., Fan X. B., Zhang W. F., Zhao Q. S., Zhang G. L., Zhang F. B., Li Y., Mater. Lett., 2014, 130, 1
Li X., Zheng J. M., Yang X. L., Dai W. L., Fan K. N., Chin. J. Catal., 2013, 34, 1013
Zwijnenburg A, Goossens A., Sloof W. G., Crajé M. W. J., van der Kraan A. M., Jos de Jongh L., Makkee M., Moulijn J. A., J. Phys. Chem. B., 2002, 106, 9853
Boccuzzi F., Chiorino A., Manzoli M., Lu P., Akita T., Ichikawa S., Haruta M., J. Catal., 2001, 202, 256
Daté M., Ichihashi Y., Yamashita T., Chiorino A., Boccuzzi F., Haruta M., Catal. Today, 2002, 72, 89
Zanella R., Louis C., Catal. Today, 2005, 107/108, 768
Hussain I., Graham S., Wang Z. X., Tan B., Sherrington D. C., Ran-nard S. P., Cooper A. I., Brust M., J. Am. Chem. Soc., 2005, 127, 16398
Westcott S. L., Oldenburg S. J., Lee T. R., Halas N. J., Langmuir, 1998, 14, 5396
Jain P. K., Huang X. H., El-Sayed I. H., El-Sayed M. A., Plasmonics, 2007, 2, 107
Driskell J. D., Lipert R. J., Porter M. D., J. Phys. Chem. B, 2006, 110, 17444
Carrot G., Valmalette J. C., Plummer C. J. G., Scholz S. M., Dutta J., Hofmann H., Hilborn J. G., Colloid Polym. Sci., 1998, 276, 853
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21271110, 21373120 and 21301098), the Applied Basic Research Programs of Science and Technology Commission Foundation of Tianjin, China(Nos.12JCYBJC13100 and 13JCQNJC02000) and the MOE Innovation Team of China(No.IRT13022).
Rights and permissions
About this article
Cite this article
Zhong, S., Han, Q., Zhu, B. et al. Promoting Effects of Iron on CO Oxidation over Au/TiO2 Supported Au Nanoparticles. Chem. Res. Chin. Univ. 34, 965–970 (2018). https://doi.org/10.1007/s40242-018-8141-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-018-8141-y