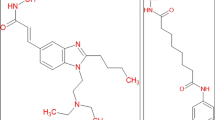
Abstract
Dihydropyrimidine derivatives possess many biological activities due to presence of pyrimidine ring structure in various nucleic acids, vitamins, coenzymes, uric acid and their derivatives. They have possessed broad spectrum actions like antibacterial, antifungal, antiviral, anticancer and antihypertensive etc. Before synthesis of compounds, it is good to predict biological activity using in silico methods. Here, we have selected some of N (3a–f) and O (4a–f) mannich bases of dihydro pyrimidine derivatives emphasized on histone deacetylase 4 (HDAC-4) inhibitions activity. We have used the different software tools like Lipinski’s rule of five; pass online; osiris property explorer and docking studies to predict anti cancer activity. All the selected compounds exhibited potential drug like molecule with anti cancer activity. Among all compound the substitution with methoxy group (3c) exhibited more drugs like property and substation with hydrogens (4a) showed high anti neoplastic activity; whereas substitution with dichloro groups (4e) showed more drug docking scores. These were compared with standard drugs tamoxifen and 5-flourouracil. The approach of predicting anticancer activity using in silico method may be more useful to select and synthesis novel compounds in research as well as in industry.






Similar content being viewed by others
References
Abadi AH, Ibrahim TM, Abouzid KM, Lehmann J, Tinsley HN, Gary BD et al (2009) Design, synthesis and biological evaluation of novel pyridine derivatives as anticancer agents and phosphodiesterase 3 inhibitors. Bioorg Med Chem 17:5974–5982
Atwal KS, Swanson BN, Unger SE, Floyd DM, Moreland S, Hedberg A et al (1991) Dihydropyrimidine calcium channel blockers. 3. 3-carbamoyl-4-aryl-1, 2, 3, 4-tetrahydro-6-methyl-5-pyrimidinecarboxylic acid esters as orally effective antihypertensive agents. J Med Chem 34:806–811
Bahekar SS, Shinde DB (2003) Synthesis and anti-inflammatory activity of some [2-amino-6-(4-substituted aryl)-4-(4-substituted phenyl)-1, 6-dihydropyrimidine-5-yl]-acetic acid derivatives. Acta Pharm 53:223–229
Bondensgaard K, Ankersen M, Thogersen H, Hansen BS, Wulff BS, Bywater RP (2004) Recognition of privileged structures by G-protein coupled receptors. J Med Chem 47:888–899
Congreve M, Carr R, Murray C, Jhoti H (2003) A ‘rule of three’ for fragment-based lead discovery? Drug Discov Today 8:876–877
DeSimone RW, Currie KS, Mitchell SA, Darrow JW, Pippin DA (2004) Privileged structures: applications in drug discovery. Comb Chem High Throughput Screen 7:473–494
Di L, Kerns EH, Carter GT (2009) Drug-like property concepts in pharmaceutical design. Curr Pharm Des 15:2184–2194
Dutta S, Sutradhar S, Sachan K (2010) Computer-aided drug design-a new approach in drug design and discovery. Computer 4:025
El-Sayed WA, Nassar IF, Adel A-H (2009) C-Furyl glycosides, II: synthesis and antimicrobial evaluation of C-furyl glycosides bearing pyrazolines, isoxazolines, and 5, 6-dihydropyrimidine-2 (1H)-thiones. Monatshefte für Chem Chem Mon 140:365–370
Filimonov DA, Lagunin AA, Gloriozova TA, Rudik AV, Druzhilovskii DS, Pogodin PV et al (2014) Prediction of the biological activity spectra of organic compounds using the pass online web resource. Chem Heterocycl Compd 50:444–457
Giffin MJ, Heaslet H, Brik A, Lin YC, Cauvi G, Wong CH et al (2008) A copper(I)-catalyzed 1,2,3-triazole azide-alkyne click compound is a potent inhibitor of a multidrug-resistant HIV-1 protease variant. J Med Chem 51:6263–6270
Hajduk PJ, Bures M, Praestgaard J, Fesik SW (2000) Privileged molecules for protein binding identified from NMR-based screening. J Med Chem 43:3443–3447
Hughes JP, Rees S, Kalindjian SB, Philpott KL (2011) Principles of early drug discovery. Br J Pharmacol 162:1239–1249
Kumarachari RK, Peta S, Surur AS, Mekonnen YT (2016) Synthesis, characterization and in silico biological activity of some 2-(N, N-dimethyl guanidinyl)-4,6-diaryl pyrimidines. J Pharm Bioallied Sci 8:181–187
Lipinski CA (2004) Lead- and drug-like compounds: the rule-of-five revolution. Drug Discov Today Technol 1:337–341
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 46:3–26
Mayer TU, Kapoor TM, Haggarty SJ, King RW, Schreiber SL, Mitchison TJ (1999) Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 286:971–974
Molecular Properties Prediction (2017) OSIRIS property explorer. http://www.organic-chemistry.org/prog/peo/. Accessed 14 Apr 2017
Nakashima S, Yamamoto K, Arai Y, Ikeda Y (2013) Impact of physicochemical profiling for rational approach on drug discovery. Chem Pharm Bull 61:1228–1238
Narayanaswamy VK, Nayak SK, Pillay M, Prasanna R, Coovadia YM, Odhav B (2013) Synthesis and antitubercular activity of 2-(substituted phenyl/benzyl-amino)-6-(4-chlorophenyl)-5-(methoxycarbonyl)-4-methyl-3, 6-dihydropyrimidin-1-ium chlorides. Chem Biol Drug Des 81:219–227
Narayanaswamy VK, Gleiser RM, Chalannavar RK, Odhav B (2014) Antimosquito properties of 2-substituted phenyl/benzylamino-6-(4-chlorophenyl)-5-methoxycarbonyl-4-methyl-3,6-dihydropyrim idin–ium chlorides against Anopheles arabiensis. Med Chem 10:211–219
Nofal ZM, Fahmy HH, Zarea ES, El-Eraky W (2011) Synthesis of new pyrimidine derivatives with evaluation of their anti-inflammatory and analgesic activities. Acta Pol Pharm 68:507–517
Ou-Yang SS, Lu JY, Kong XQ, Liang ZJ, Luo C, Jiang H (2012) Computational drug discovery. Acta Pharmacol Sin 33:1131–1140
Sahu M, Siddiqui N (2016) A review on biological importance of pyrimidines in the new era. Int J Pharm Pharm Sci 2016:14
Sangameswaran L, Fales HM, Friedrich P, De Blas AL (1986) Purification of a benzodiazepine from bovine brain and detection of benzodiazepine-like immunoreactivity in human brain. Proc Natl Acad Sci USA 83:9236–9240
Shah T, Gupte A, Patel M, Chaudhari V, Patel H, Patel V (2010) Synthesis and in vitro study of biological activity of heterocyclic N-Mannich bases of 3,4-dihydro-pyrimidine-2 (1H)-thiones. Indian J Chem Sect B Org Incl Med 49:578
Taj T, Kamble RR, Gireesh TM, Hunnur RK, Margankop SB (2011) One-pot synthesis of pyrazoline derivatised carbazoles as antitubercular, anticancer agents, their DNA cleavage and antioxidant activities. Eur J Med Chem 46:4366–4373
Thomsen R, Christensen MH (2006) MolDock: a new technique for high-accuracy molecular docking. J Med Chem 49:3315–3321
Wang R, Fang X, Lu Y, Wang S (2004) The PDBbind database: collection of binding affinities for protein–ligand complexes with known three-dimensional structures. J Med Chem 47:2977–2980
Weigelt J (2010) Structural genomics-impact on biomedicine and drug discovery. Exp Cell Res 316:1332–1338
Yusof I, Segall MD (2013) Considering the impact drug-like properties have on the chance of success. Drug Discov Today 18:659–666
Zou Y, Zhao Q, Liao J, Hu H, Yu S, Chai X et al (2012) New triazole derivatives as antifungal agents: synthesis via click reaction, in vitro evaluation and molecular docking studies. Bioorg Med Chem Lett 22:2959–2962
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thipparapu, G., Ajumeera, R. & Venkatesan, V. Novel dihydropyrimidine derivatives as potential HDAC inhibitors: in silico study. In Silico Pharmacol. 5, 10 (2017). https://doi.org/10.1007/s40203-017-0030-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40203-017-0030-4