Abstract
In recent years, there has been developing interests in microbial fuel cell (MFC) technology as a hopeful approach to overcome worldwide energy crisis that will come into account by restricting fossil fuels consumption in the future. In addition, many drugs around the world are accessible to some populations easily and remain unused annually. This paper discusses bioelectricity generation with whey degradation in a dual chambered MFC in the presence of some expired medicinal drug-feeding cathodes. Consequently, a two-chamber MFC applied using Escherichia coli as biocatalyst, humic acid as electron mediator and Nafion 117 with a 5 nm in size as nano-membrane. The results showed that the open-circuit potential was 0.751 V at ambient condition. Stability of the voltage was exceeded 20 h. Acetaminophen codeine and Bismuth—as two expired medicinal drugs—applied as possible catholyte. In conclusion, Bismuth revealed more opportunity for power deriving in comparison with Acetaminophen. The best values were close to 2.9 × 10−5 W and 1.75 × 10−4 A, referring to power generation and current production, respectively.
Similar content being viewed by others
Introduction
By confronting environmental pollution issues and fossil fuels overconsumption, it is manifest for humanity to explore a utile, trusty, and clean energy system to replace the current energy production systems. A great volume of expired and outdated drugs as well as generated wastewater determined by chemical and biological oxygen demand (COD and BOD) concentrations [1,2,3,4] are other important issue imposing lots of expense on governments around the world. In addition, Whey is a well-known product of cheese processing industry [5, 6] and could be used as suggested biochemical analyte for generation of bio-derived source electricity. The possibility of using MFC for power generation has studied before [7,8,9,10]. Some researchers studied the principles of MFC especially in selecting microorganisms of interest [11], economical substrate [12], pilot-plant study for power production [13], modeling of biofilm constitution with engineering aspect [14], and role of electron acceptors in MFCs [30], respectively. The other groups verified the importance of this technology for power production [15,16,17,18,19,20,21]. Several kinds of mediators had applied in such system to augment the electron transfer rate, but they are generally expensive to be used in pilot or industrial scale program of MFC manufacture and showed toxicity to secreted biocatalysts for long-period operation [22]. In addition, several microbial systems with various substrates and mediators have been reported [23,24,25]. To the best of our knowledge, this is the first survey presenting MFCs for generating electricity using expired drugs as electron shuttle. The main novelty was investigation and introduction of electricity harvesting from whey wastewater in the anode compartment, especially applying two expired medical drugs in cathode compartment of MFC as a new suggested trend and their functions as a light at the end of the tunnel in terms of power generation by MFC.
Materials and methods
Chemicals and materials
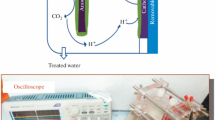
All chemicals used as our previous work as follows [3], unless otherwise stated. Gela dairy product industry (Amol, Iran) was the provider of substrate. Excess proteins in substrate were precipitated by acid solution (HCl, 2 N). The autoclaving of solution was carried out at 15 psig, 121 °C for 15 min. After passing of cooling down process to mild temperature, it underwent centrifuging at 7000×g-values of RCF in units of times gravity for unwanted solids removal. Then, the supernatant transferred to refrigerator for 12 h and stored. Rohani Hospital (Babol, Iran) was the provider of microbial catalyst (Escherichia coli) as a close cooperation. Microorganisms have grown in an anaerobic condition. The anode mediator and cathodic electron shuttles was supplied by Merck (Darmstadt, Germany). Figure 1 depicts the schematic diagram of the MFC and its auxiliary equipment [3, 4], made of plexy glass material. The total volume of cells was 910 ml, which 800 ml out of expressed primary volume was used as working volume. Graphite was the selected electrode which has been provided in the size of 40 mm × 90 mm × 3 mm. The proton exchange membrane (PEM, Nafion 117 with a 5 nm pore size, Sigma-Aldrich) provided to separate two chambers of MFC. Ordinarily, all materials used in the experiments were analytical grade and supplied by Merck (Dortmund, Germany) except Acetaminophen codeine (325 mg) and Bismuth (200 mg) bought from local pharmacy. In all solutions, digital Metrohm pH meter (model 744) equipped with a combined glass–calomel electrode was used to adjust the pH. Dinitrosalicylic acid method applied to measure whey consumption using colorimetric method [26].
The microorganism growth monitoring was accomplished by spectrophotometer (Unico, USA) established as optical density. SEM (Philips XL30 scanning microscope, Philips, The Netherlands) employed to determine the shape and surface morphology of electrode in the present MFC. The graphite was coated with gold before the SEM measurement with the selected magnification of 5000.
Arrangement of medium and inoculum
The medium is comprised of glucose, yeast extract, ammonium chloride, and peptone in grouping of 10, 1.0, 0.5, and 1.0 g/l, respectively. It was disinfected by autoclaving at 121 °C, 15 psig for 20 min. Initially, the pH of medium, inoculum, and other parameters had adjusted in 7.0. Thereafter, it was heated at 30 °C. The E. coli had completely developed for 24 h in 100 ml flux with no additional shaking. The sampling was done in every 4.0 h to investigate the substrate utilization on reduced sugar content by aforementioned technique [27].
Anodes and nano-membrane pretreatment
To keep up layer for good conductivity, the two compartments preserved in double-distilled water when the bioreactor was not being applied [28]. The anode compartment had introduced to serial pretreatment, which has been engrossing 100% ethanol for 45 min and in 1.0 M HCl for 1.0 h. After every usage, cathodes were immersed in 1.0 M HCl, 1.0 M NaOH, for 1.0 h, independently to clear any metallic and normal contamination by exposing indistilled water before use, respectively [28]. Nafion membrane was exposed to serial treatment to remove contaminations which include washing the film for 1.0 h, immersion in 3.0% H2O2, following washing with double-distilled water, re-immersing in acidic solution and eventually re-exposing to distilled water, respectively.
Depiction of the data procurement framework
Simple computerized information procurement had produced to record sampling information in every 6 s. The framework records measurements for variable resistances which were imposed to the MFC. By dividing the obtained voltage from characterized resistance, the output current recorded. Then, the system provides power calculation by multiplication of voltage and current. In addition, the microcontroller sends the essential information to a PC by analogue–digital converter. Moreover, unique capacity of MATLAB programming (7.4, 2007a) was utilized to store and synchronically show the received information.
Statistical analysis
All facts are presented in this text as mean result ± SD (standard deviation). Statistical analysis were assessed applying the t test and considered significant at P < 0.05 level. All figures shown in this article have been received from three unbiased experiments with similar results.
Results and discussion
MFC performance
Power deriving was accomplished applying the by-product, abundant, and easily available source of food industry. The effect of mediators as electron promoters had investigated. Polarization is the difference of electrical potential between two ends of an electrical tool when there is no external load connected. Hence, there is not external electric current flow between the ends. It is sometimes given the symbol Voc or OCV; but in network analysis, this voltage is also known as the Thévenin voltage. As resistance had reduced in such gadget, the voltage will lower. In the first moment of action, the voltage was in lower level (390 mV) and then gradually reached to a high value. To put in a nutshell, carbon source utilization and final product formation in the form of bioenergy were the outcome of this stage [3]. Open-circuit voltage is measured in the presence of humic acid in anode chamber with no catholyte involvement during about 4.0 days. Following 60 h of operation, the recorded Voc value was 0.751 V. It was thoroughly unchanged for more than 1 day as our previous applied protocol [3]. Finally, a decrease as instability was observed (Fig. 2). It can be attributed to decrease of lactose content in the anode chamber by bacteria depleting as substrate after 30 h. Generally talking, polarization curve is a main concept in electrochemical reactions. In other words, it is a law based on kinetic principals that is visualized as a plot of current density versus electrode potential or having three parameters as voltage, current, and power in a plot simultaneously. The achieved data rely on electrical parameters involved in the MFC. In Figs. 3 and 4, the polarization curve in the presence of Acetaminophen codeine and Bismuth as suitable catholyte is shown. The selected mediator in MFC was humic acid (0.2 mol/m3) based on our previous work due to highlighted potential [3]. Derived power and related current were close to 6.8 × 10−6 W and 3.5 × 10−5 A, respectively.
Initial open-circuit voltage (OCV) versus time following current switch-off of fabricated MFC [3]
The mediator in MFC was the main factor in increasing electron transfer interestingly achieves more power production and cell current efficiency. The drawbacks of MFC were mentioned in the previous scientific papers [25]. One of the expressed suggestions for overcoming proposed issue is identification of new mediators. Figures 3, 4 show power generation in the presence of two aforementioned expired medicinal drugs as hygienic organization wastes. The maximum power and current production in the presence of Acetaminophen and Bismuth as novel cathodic mediators was 2.55 × 10−5 W, 1.29 × 10−5 A, 2.9 × 10−5 W and 1.75 × 10−4 A, respectively. The maximum value of Voc is attributed to the presence of expired drugs as electron mediators. Humic acid acts as electron acceptor in the anode compartment and two expired medicines act as electron shuttle in the cathode chamber. These outputs are comparable to other researchers’ findings [15, 16], especially in a case without using nanomaterial except the selected membrane. In this work, a novel approach to bioelectricity generation and new limited medicinal waste management opportunity as a novelty was introduced and developed. To best of our knowledge, this is the first attitude as a light at the end of the tunnel upon applying and developing expired drugs as mediator in cathode compartment of a bioreactor like as MFC. Although the obtained quantities based on power and generation are not in high level, but the results open a new horizon for related industries and compartments to investigate more in such a new introduced concept.
Characterization of graphite electrode
As our previous work, the same electrodes were applied for power generation and transformation in designed electrical circuit. Figure 5a, b shows surface of the electrode before and after the operation. In addition, SEM image of the surface electrode with fixed regulated magnification of 5000 successfully shows characteristic of the graphite plate (Fig. 6). SEM imaging on the applied graphite electrode in the size of 1 cm × 1 cm was established to characterize the shape and surface morphology. Philips XL30 scanning microscope, (Philips, The Netherlands) was the features of applied SEM. Coating of membrane was achieved with gold before taking any action. The surface impurities have depicted on the electrode surface with black arrows embedded in Fig. 6. Based on obtained SEM, electrodes need retreating to achieve a better efficiency if designed to be reused.
Graphite electrode connected with wire before (a) and after (b) bioelectricity generation [3]
COD and BOD removal efficiencies
To assess the stable performance of the MFC (Fig. 7), COD removal efficiency and voltage output were considered as indicators. The MFC were operated continuously for 74 h. Approximately, after 60 h, a stable condition was achieved for the MFC. Maximum COD and BOD removal up to 89 and 90% was obtained for the selected wastewater with average initial concentration of 70,000 and 40,000 mg/l, respectively. The efficiency of COD removal was in a very good agreement with the maximum reported efficiency 91 and 82%, respectively [29,30,31].
Columbic efficiency
Columbic efficiency (CE) was calculated according to Logan and his co-workers [29]. The result is shown in Fig. 8. For whey wastewater, the CEs increased over time. After 60 h, the CEs reached its maximum value, indicating that microorganisms consumed whey as close to 89%. In addition, the electrons in the removed substrate were used for current production. The percentage of columbic efficiency reveals a good phenomenon based on data obtained from Fig. 8 [32].
Conclusion
Whey utilized as selected analyte in the MFC. The performance of MFC investigated using two expired medicinal drugs so-called Acetaminophen codeine and Bismuth implementing as mediatory agents in cathode compartment. The result of this study indicated that humic acid served as suitable mediator. Implemented Acetaminophen as expired drug was unsuitable choice as cathodic electron acceptor, but Bismuth opened a new horizon toward working on other expired therapeutic drugs in cathode compartment for ideal performance. The most interesting finding was that the produced power in the presence of Acetaminophen codeine was lower than OCV in the absence of catholyte. The reason for this is not clear, but it may have something to do with relative competent groups for electricity exists in Acetaminophen codeine. The maximum power and current was 2.87 × 10−5 W and 1.75 × 10−4 A, respectively, although further studies will be need on many aspects of this technology before these approaches could be commercialized successfully, but these results provided a new horizon to produce bioelectricity in a world where the energy crisis threatens its inhabitants. The future program will be focused on using different class of antibiotics and nano-materials to develop new introduced horizon.
Abbreviations
- A:
-
Ampere
- BOD:
-
Biological oxygen demand
- CE:
-
Columbic efficiency
- cm:
-
Centimeter
- COD:
-
Chemical oxygen demand
- DNS:
-
Dinitrosalicylic Acid
- g:
-
Gram
- h:
-
Hour
- L:
-
Liter
- M:
-
Molarity
- MFC:
-
Microbial fuel cell
- mg:
-
Milligram
- min:
-
Minute
- ml:
-
Milliliter
- N:
-
Normality
- OCV:
-
Open-circuit voltage
- PC:
-
Personal computer
- PEM:
-
Proton exchange membrane
- SD:
-
Standard deviation
- SEM:
-
Scanning electron microscope
- W:
-
Watt
References
Akbari-adergani, B., Attaran, A.M., Veiskarami, M.: Optimization of parameters in concentration technology for treatment and reduction of organic load in distillery’s vinasse. J. Tolooebehdasht. 4, 21–31 (2013)
Akbari-adergani, B., Attaran, A.M., Taghimolla, Z., Shoeibi, Sh: Modification of designing system in chemical oxygen demand test with silver nano-particles for determination of organic load in effluent of pharmaceutical industries. J. Tolooebehdasht 3, 69–82 (2012)
Nasirahmadi, S., Safekordi, A.A.: Enhanced electricity generation from whey wastewater using combinational cathodic electron acceptor in a two-chamber microbial fuel cell. Int. J. Environ. Sci. Technol. 9(3), 473–478 (2012)
Nasirahmadi, S., Safekordi, A.A.: Whey as a substrate for generation of bioelectricity in microbial fuel cell using E. coli. Int. J. Environ. Sci. Technol. 8(4), 823–830 (2011)
Ferchichi, M., Crabbe, E., Gil, G.H., Hintz, W., Almadidy, A.: Influence of initial pH on hydrogen production from cheese whey. J. Biotechnol. 120, 402–409 (2005)
Kisaalita, W.S., Pinder, K.L., Lo, K.V.: Acidogenic fermentation of lactose. Biotechnol. Bioeng. 30, 88–95 (1987)
Lini, K.M.: Microbial fuel cells: a promising tool for power generation. Int. J. Sci. Res. 4(7), 2530–2532. ISSN 2319-7064 (2013)
Bavasso, I., Di Palma, L., Petrucci, E.: Treatment of wastewater in H-Type MFC with protonic exchange membrane: experimental study of organic carbon and ammonium reduction with electrochemical characterization. Chem. Eng. Trans. 47, 1–6 (2016)
Vinicius, F.P., Sidney, A.N., Adalgisa, R.A., Valeria, R.: Energy generation in a microbial fuel cell using anaerobic sludge from a wastewater treatment plant. Sci. Agric. 7(35), 424–428 (2015)
Weihua, H., Xiaoyuan, Z., Jia, L., Xiuping, Z., Yujie, F., Bruce, E.L.: Microbial fuel cells with an integrated spacer and separate anode and cathode modules. Environ. Sci. Water Res. Technol. 2, 186–195 (2015)
Mao, L., Verwoerd, W.S.: Selection of organisms for systems biology study of microbial electricity generation: a review. Int. J. Energy Environ. Eng. 4(17), 1–18 (2013)
Sirinutsomboon, : Modeling of a membrane less single-chamber microbial fuel cell with molasses as an energy source. Int. J. Energy Environ. Eng. 5(93), 1–9 (2014)
Kiran, T.M., Parneet, P.: Performance of pilot-scale microbial fuel cells treating wastewater with associated bioenergy production in the Caribbean context. Int. J. Energy. Environ Eng. 6, 213–220 (2015)
Gatti, M.N., Milocco, R.H.: A biofilm model of microbial fuel cells for engineering applications. Int. J. Energy Environ. Eng. 8, 303–315 (2017)
Entchev, E., Yang, L., Chorab, M., Rosato, A., Sibilio, S.: (2016) Energy, economic and environmental performance simulation of a hybrid renewable microgeneration system with neural network predictive control. Alex. Eng. J. 1–19. https://doi.org/10.1016/j.aej.2016.09.001
Gajda, I., Greenman, J., Melhuish, C., Ieropoulos, I.: Self-sustainable electricity production from algae grown in a microbial fuel cell system. Biomass Bioenergy 82, 87–93 (2015)
Huawei, J., Azahar Ali, Md., Zhen, X., Larry, J.H., Liang, D.: Integrated microfluidic flow-through microbial fuel cells. Sci. Rep. 7, 41208 (2017). https://doi.org/10.1038/srep41208
Olga, T., Lihong, L., Aijie, W.: Electricity generation by Enterobacter sp. of single-chamber microbial fuel cells at different temperatures. J. Clean Energy Technol. 4(1), 36–42 (2016)
Izadi, P., Rahimnejad, M., Ghoreyshi, A.: Power production and wastewater treatment simultaneously by dual-chamber microbial fuel cell technique. Biotechnol. Appl. Biochem. (2015). https://doi.org/10.1002/bab.1345
Noori, P., Najafpour-Darzi, G.H.: Enhanced power generation in annular single chamber microbial fuel cell via optimization of electrode spacing using chocolate industry wastewater. Biotechnol. Appl. Biochem. (2015). https://doi.org/10.1002/bab.1374
Sarika, A.L., Vibhavari, D.P., Deelip, B.P.: Role of mediators in microbial fuel cell for generation of electricity and waste water treatment. Int. J. Chem. Sci. Appl. 6(1), 6–11 (2015)
Swamini, P., Kris, S., Abhilash, B., Froila, D., Yash, M., Vikram, S., Ritika, R., Joice, M., Radhika, B., Aruna, K.: Bioremediation of dye effluent waste through an optimized microbial fuel cell. Int. J. Adv. Res. Biol. Sci. 3, 214–226 (2016)
Venkatesh, C., Pradeep, V.: Microbial fuel cell: a green approach for the utilization of waste for the generation of bioelectricity. Bioresour. Bioprocess. 3(38), 1–14 (2016)
Thomas, L.C., Chamberlin, G.J., Ltd, Tintometer: Colorimetric chemical analytical methods, p. 31. Tintometer Ltd, Salisbury (1980)
Rodrigo, M.A., Canizares, P., Lobato, J., Paz, R., Saez, C., Linares, J.J.: Production of electricity from the treatment of urban waste water using a microbial fuel cell. J. Power. Sources 169, 198–204 (2007)
Chae, K.J., Choi, M., Ajayi, F.F., Park, W., Chang, I.S., Kim, I.S.: Mass transport through a proton exchange membrane (Nafion) in microbial fuel cell. Energy Fuels 22, 169–176 (2008)
Elakkiya, E., Matheswaran, M.: Comparison of anodic metabolisms in bioelectricity production during treatment of dairy wastewater in microbial fuel cell. Bioresour. Technol. 136, 407–412 (2013)
Mathuriya, A.S., Sharma, V.N.: Bioelectricity production from various wastewaters through microbial fuel cell technology. J. Biochem. Technol. 2, 133–137 (2010)
Logan, B.E., Hamelers, B., Rozendal, R., Schrorder, U., Keller, J., Freguia, S., Aelterman, P., Verstraete, W., Rabaey, K.: Microbial fuel cells: methodology and technology. Environ. Sci. Technol. 40, 5181–5192 (2006)
Ucar, D., Yifeng Zhang, Y., Angelidaki, I.: An overview of electron acceptors in microbial fuel cells. Front. Microbiol. 8(643), 1–14 (2017)
Du, Z., Li, H., Gu, T.: A state of the art review on microbial fuel cells: a promising technology for wastewater treatment and bioenergy. Biotechnol. Adv. 25, 464–482 (2007)
Timmers, R.A., Strik, D., Hamelers, H.V.M., Buisman, C.J.N.: Electricity generation by a novel design tubular plant microbial fuel cell. Biomass Bioenergy 51, 60–67 (2013)
Acknowledgements
The food and drug control reference laboratories and the water safety research center due to their laboratory facilities support in this work are acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to urisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nasirahmadi, S., Akbari-adergani, B. Microbial fuel cell with a nano-membrane and two expired medicinal drug-feeding cathode: a novel strategy. Int J Energy Environ Eng 9, 249–256 (2018). https://doi.org/10.1007/s40095-018-0262-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40095-018-0262-z