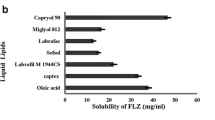
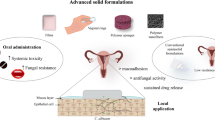
Abstract
The main objective of the present work was to develop and evaluate a mucoadhesive in-situ based gel for the vaginal delivery of Fluconazole (FLZ). The formulation was developed using the cold method and evaluated for various physicodynamic and aesthetic properties. The gelling agents used were evaluated for their potential to gel FLZ without affecting its structure. The bioadhesive potential and antifungal activity of the FLZ in-situ gel was determined in comparison to the marketed antifungal topical cream, FLUCOS®. The drug content varied from 95.3 to 98.01% and pH values were found to be slightly acidic (4.2–4.5), as normal for the vaginal milieu. The FLZ in-situ gel showed significantly higher in-vitro bioadhesion of 0.277 and antifungal activity as compared to the marketed formulation with a bioadhesion of 0.281, and did not show any signs of vaginal irritation. Thus, it can be concluded that the developed intravaginal in-situ gel formulation can be used for effective targeted treatment of vulvovaginal candidiasis (VVC) in comparison to FLUCOS® with better mucoadhesion, drug release, permeability and drug retention characteristics with more acceptable patient compliance.






Similar content being viewed by others
References
Baloglu E, Karavana SY et al (2011) Rheological and mechanical properties of poloxamer mixtures as a mucoadhesive gel base. Pharm Dev Technol 16:627–636
Bernkop-Schnürch A, Hornof M (2003) Intravaginal drug delivery systems. Am J Drug Deliv 1(4):241–254
Caramella CM, Rossi S et al (2015) Mucoadhesive and thermogelling systems for vaginal drug delivery. Adv Drug Deliv Rev 92:39–52
Chang JY, Oh YK et al (2002) Rheological evaluation of thermosensitive and mucoadhesive vaginal gels in physiological conditions. Int J Pharm 24(1):155–163
Choi H, Jung JH et al (1998) Development of in-situ gelling and muco-adhesive acetaminophen liquid suppository. Int J Pharm 165(4):33–44
Curran RM, Donnelly L et al (2009) Vaginal delivery of the recombinant HIV-1 clade-C trimeric gp140 envelope protein CN54gp140 within novel rheologically structured vehicles elicits specific immune responses. Vaccine 27:6791–6798
Horowitz BJ, Giaquinta D et al (1992) Evolving pathogens in vulvovaginal candidiasis: implications for patient care. J Clin Pharmacol 32(3):248–255
Ibrahim ESA, Ismail S et al (2012) Development and characterization of thermosensitive pluronic-based metronidazole in-situ gelling formulations for vaginal application. Acta Pharm Sci 62:59–70
Jain N, Aswal V et al (1998) Micellar structure of an ethylene oxide-propylene oxide block copolymer: a small-angle neutron scattering study. J Phys Chem 102:8452–8460
Johal HS, Garg T et al (2016) Advanced topical drug delivery system for the management of vaginal candidiasis. Drug Deliv 23(2):550–563
Kabanov AV, Batrakova EV et al (2002) Pluronic® block copolymers as novel polymer therapeutics for drug and gene delivery. J Controll Release 82(2):189–212
Kant A, Reddy S et al (2011) In situ gelling system—an overview. Pharmacol Online 2:28–44
Majid Z, Ali ZM (2009) Invasive candidiasis; a review article. Jundishapur J Microbiol 2009(1, Winter):1–6
Mohandas V, Ballal M (2011) Distribution of Candida species in different clinical samples and their virulence: biofilm formation, proteinase and phospholipase production: a study on hospitalized patients in southern India. J Glob Infect Dis 3(1):4
Patil PR, Shaikh SS et al (2014) In-situ gel: a novel drug delivery system. Indo Am J Pharm Res 4(11):5406–5414
Rendo-Lopez V, Lynch M et al (1990) Torulopsis glabrata vaginitis: clinical aspects and susceptibility to antifungal agents. Obstetr Gynecol 76(4):651–655
Sakthivel M, Kannan K et al (2012) Formulation and in vitro evaluation of niosomes containing oxcarbazepine. Int J Pharm Sci 4:563–567
Schippera NGM, Varumb KM et al (1999) Chitosans as absorption enhancers of poorly absorbable drugs 3: influence of mucus on absorption enhancement. Eur J Pharm Sci 8:335–343
Shankar GN, Alt C (2014) Prophylactic treatment with a novel bioadhesive gel formulation containing aciclovir and tenofovir protects from HSV-2 infection. J Antimicrob Chemother 69:3282–3293
Sobel JD (1993) Candidal vulvovaginitis. Clin Obstet Gynecol 36(1):153–165
Tett S, Moore S et al (1995) Pharmacokinetics and bioavailability of fluconazole in two groups of males with human immunodeficiency virus (HIV) infection compared with those in a group of males without HIV infection. Antimicrob Agents Chemother 39(8):1835–1841
Woolfson AD, Malcolm RK et al (2000) Drug delivery by the intravaginal route. Crit Rev Therap Drug Carrier Syst 17:509–555
Yamsani V, Gannu Y et al (2007) Development an in-vitro evaluation of buccoadhesive carvedilol tablets. Acta Pharm 57:185–197
Acknowledgements
The authors are thankful to University Institute of Pharmaceutical Sciences, Panjab University, Chandigarh for their skillfull assistance and for providing study facility in this study. Authors would like to acknowledge Microbiology Department, Panjab University, Chandigarh for their support rendered during this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest regarding publication of this paper.
Rights and permissions
About this article
Cite this article
Sarwal, A., Singh, G., Singh, S. et al. Novel and effectual delivery of an antifungal agent for the treatment of persistent vulvovaginal candidiasis. J. Pharm. Investig. 49, 135–147 (2019). https://doi.org/10.1007/s40005-018-0395-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-018-0395-3