Abstract
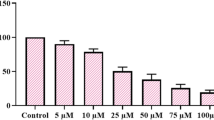
Fourteen diphenyl imidazoline derivatives were designed, synthesized, and identified using NMR spectroscopy and high-resolution mass spectrometry. Their cytotoxicities in HCT 116 colorectal cancer cell lines were measured using a clonogenic long-term survival assay and the half-maximal cell growth inhibitory concentration (GI50) values were in the range 3.1–58.4 μM. As the anticancer effects of diphenyl imidazolines were reported to be caused by the inhibition of mouse double minute 2 homolog (MDM2), the inhibitory effects of the most potent derivative on MDM2 were assessed through Western blotting analysis. In silico docking experiments revealed the binding mode between this derivative and MDM2.









Similar content being viewed by others
References
Ueno M, Imaizumi K, Sugita T, Takata I, Takeshita M (1995) Effect of a novel anti-rheumatic drug, TA-383, on type II collagen-induced arthritis. Int J Immunopharmacol 17(7):597–603
Sugita T, Ueno M, Furukawa O, Murakami T, Takata I, Tosa T (1993) Effect of a novel anti-rheumatic drug, TA-383, on type II collagen-induced arthritis—suppressive effect of TA-383 on interleukin 6 production. Int J Immunopharmacol 15(4):515–519
Li YF, Gong ZH, Cao JB, Wang HL, Luo ZP, Li J (2003) Antidepressant-like effect of agmatine and its possible mechanism. Eur J Pharmacol 469(1–3):81–88
Rodriguez F, Rozas I, Kaiser M, Brun R, Nguyen B, Wilson WD, Garcia RN, Dardonville C (2008) New bis(2-aminoimidazoline) and bisguanidine DNA minor groove binders with potent in vivo antitrypanosomal and antiplasmodial activity. J Med Chem 51(4):909–923
Browne N, Hackenberg F, Streciwilk W, Tacke M, Kavanagh K (2014) Assessment of in vivo antimicrobial activity of the carbene silver(I) acetate derivative SBC3 using Galleria mellonella larvae. Biometals 27(4):745–752
Ueno M, Sugita T, Murakami T, Takata I (1997) The novel anti-rheumatic drug TA-383 has a macrophage migration enhancing activity. Jpn J Pharmacol 74(2):221–224
Shukla S, Bhalla M, Misra U, Mukerjee D, Saxsena AK, Sinha JN, Shanker K (1998) Cardiovascular effects of novel imidazoline congeners. Boll Chim Farm 137(7):229–232
Dardonville C, Goya P, Rozas I, Alsasua A, Martin MI, Borrego MJ (2000) New aromatic iminoimidazolidine derivatives as alpha1-adrenoceptor antagonists: a novel synthetic approach and pharmacological activity. Bioorg Med Chem 8(7):1567–1577
Marson CM, Matthews CJ, Atkinson SJ, Lamadema N, Thomas NS (2015) Potent and selective inhibitors of histone deacetylase-3 containing chiral oxazoline capping groups and a N-(2-aminophenyl)-benzamide binding unit. J Med Chem 58(17):6803–6818
Koga K, Honda K, Ando S, Harasawa I, Kamiya HO, Takano Y (2004) Intrathecal clonidine inhibits mechanical allodynia via activation of the spinal muscarinic M1 receptor in streptozotocin-induced diabetic mice. Eur J Pharmacol 505(1–3):75–82
Cheng YF, Hu YZ, He QJ (2005) Synthesis and antitumor activity of arylsubstituted imidazolin-2-one derivatives. Yao Xue Xue Bao 40(8):711–716
American Cancer Society. www.cancer.org
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63
Cory AH, Owen TC, Barltrop JA, Cory JG (1991) Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun 3(7):207–212
Berridge MV, Tan AS (1993) Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch Biochem Biophys 303(2):474–482
Franken NA, Rodermond HM, Stap J, Haveman J, van Bree C (2006) Clonogenic assay of cells in vitro. Nat Protoc 1(5):2315–2319
Zhang Z, Chu XJ, Liu JJ, Ding Q, Zhang J, Bartkovitz D, Jiang N, Karnachi P, So SS, Tovar C, Filipovic ZM, Higgins B, Glenn K, Packman K, Vassilev L, Graves B (2014) Discovery of potent and orally active p53-MDM2 inhibitors RO5353 and RO2468 for potential clinical development. ACS Med Chem Lett 5(2):124–127
Carry JC, Garcia-Echeverria C (2013) Inhibitors of the p53/hdm2 protein-protein interaction-path to the clinic. Bioorg Med Chem Lett 23(9):2480–2485
Ghosh P, Zhang J, Shi ZZ, Li K (2013) Synthesis and evaluation of an imidazole derivative-fluorescein conjugate. Bioorg Med Chem 21(8):2418–2425
Zhang Z, Ding Q, Liu JJ, Zhang J, Jiang N, Chu XJ, Bartkovitz D, Luk KC, Janson C, Tovar C, Filipovic ZM, Higgins B, Glenn K, Packman K, Vassilev LT, Graves B (2014) Discovery of potent and selective spiroindolinone MDM2 inhibitor, RO8994, for cancer therapy. Bioorg Med Chem 22(15):4001–4009
Dauwe C, Buddrus J (1995) Synthesis of enantiopure C-2-chiral amidines. Synthesis-Stuttgart 2:171–172
Ishihara M, Togo H (2006) An efficient preparation of 2-imidazolines and imidazoles from aldehydes with molecular iodine and (diacetoxyiodo)benzene. Synlett 2:227–230
Fujioka H, Murai K, Ohba Y, Hiramatsu A, Kita Y (2005) A mild and efficient one-pot synthesis of 2-dihydroimidazoles from aldehydes. Tetrahedron Lett 46(13):2197–2199
Yong Y, Ahn S, Hwang D, Yoon H, Jo G, Kim YH, Kim SH, Koh D, Lim Y (2013) 1H and 13C NMR spectral assignments of 2′-hydroxychalcones. Magn Reson Chem 51(6):364–370
Ahn S, Shin SY, Jung Y, Jung H, Kim BS, Koh D, Lim Y (2016) (1) H and (13) C NMR spectral assignments of novel flavonoids bearing benzothiazepine. Magn Reson Chem 54(5):382–390
Shin SY, Jung H, Ahn S, Hwang D, Yoon H, Hyun J, Yong Y, Cho HJ, Koh D, Lee YH, Lim Y (2014) Polyphenols bearing cinnamaldehyde scaffold showing cell growth inhibitory effects on the cisplatin-resistant A2780/Cis ovarian cancer cells. Bioorg Med Chem 22(6):1809–1820
Yoon H, Kim TW, Shin SY, Park MJ, Yong Y, Kim DW, Islam T, Lee YH, Jung KY, Lim Y (2013) Design, synthesis and inhibitory activities of naringenin derivatives on human colon cancer cells. Bioorg Med Chem Lett 23(1):232–238
Jung Y, Shin SY, Yong Y, Jung H, Ahn S, Lee YH, Lim Y (2015) Plant-derived flavones as inhibitors of aurora B kinase and their quantitative structure-activity relationships. Chem Biol Drug Des 85(5):574–585
Shin SY, Ahn S, Yoon H, Jung H, Jung Y, Koh D, Lee YH, Lim Y (2016) Colorectal anticancer activities of polymethoxylated 3-naphthyl-5-phenylpyrazoline-carbothioamides. Bioorg Med Chem Lett 26(17):4301–4309
Jung H, Shin SY, Jung Y, Tran TA, Lee HO, Jung KY, Koh D, Cho SK, Lim Y (2015) Quantitative relationships between the cytotoxicity of flavonoids on the human breast cancer stem-like cells MCF7-SC and their structural properties. Chem Biol Drug Des 86(4):496–508
Fry DC, Emerson SD, Palme S, Vu BT, Liu CM, Podlaski F (2004) NMR structure of a complex between MDM2 and a small molecule inhibitor. J Biomol NMR 30(2):163–173
Gonzalez AZ, Li Z, Beck HP, Canon J, Chen A, Chow D, Duquette J, Eksterowicz J, Fox BM, Fu J, Huang X, Houze J, Jin L, Li Y, Ling Y, Lo MC, Long AM, McGee LR, McIntosh J, Oliner JD, Osgood T, Rew Y, Saiki AY, Shaffer P, Wortman S, Yakowec P, Yan X, Ye Q, Yu D, Zhao X, Zhou J, Olson SH, Sun D, Medina JC (2014) Novel inhibitors of the MDM2-p53 interaction featuring hydrogen bond acceptors as carboxylic acid isosteres. J Med Chem 57(7):2963–2988
Shin SY, Yoon H, Hwang D, Ahn S, Kim DW, Koh D, Lee YH, Lim Y (2013) Benzochalcones bearing pyrazoline moieties show anti-colorectal cancer activities and selective inhibitory effects on aurora kinases. Bioorg Med Chem 21(22):7018–7024
Wallace AC, Laskowski RA, Thornton JM (1995) LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions. Protein Eng 8(2):127–134
Fridman JS, Lowe SW (2003) Control of apoptosis by p53. Oncogene 22(56):9030–9040
Yin Y, Tainsky MA, Bischoff FZ, Strong LC, Wahl GM (1992) Wild-type p53 restores cell cycle control and inhibits gene amplification in cells with mutant p53 alleles. Cell 70(6):937–948
Zhan Q (2005) Gadd45a, a p53- and BRCA1-regulated stress protein, in cellular response to DNA damage. Mutat Res 569(1–2):133–143
Kubbutat MH, Jones SN, Vousden KH (1997) Regulation of p53 stability by Mdm2. Nature 387(6630):299–303
Malmlof M, Roudier E, Hogberg J, Stenius U (2007) MEK-ERK-mediated phosphorylation of Mdm2 at Ser-166 in hepatocytes. Mdm2 is activated in response to inhibited Akt signaling. J Biol Chem 282(4):2288–2296
Zhou BP, Liao Y, Xia W, Zou Y, Spohn B, Hung MC (2001) HER-2/neu induces p53 ubiquitination via Akt-mediated MDM2 phosphorylation. Nat Cell Biol 3(11):973–982
Acknowledgments
This work was supported by the Priority Research Centers Program [NRF, 2009-0093824]. SY Shin was supported by the KU Research Professor Program of Konkuk University.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, Y., Koh, D., Ahn, S. et al. Clonogenic long-term survival assay of HCT 116 colorectal cancer cells after treatment with the synthesized diphenyl imidazoline derivatives. Appl Biol Chem 61, 303–312 (2018). https://doi.org/10.1007/s13765-018-0355-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-018-0355-7