Abstract
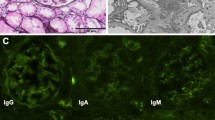
Pregnancy and membranous nephropathy (MN) can occur concurrently with nephrotic syndrome. However, the pathophysiology of MN associated with pregnancy remains unclear, including the involvement of anti-M-type phospholipase A2 receptor (PLA2R) antibody, the major antigen of idiopathic MN (iMN). A treatment for the condition is also not established. We present the case of a 43-year-old pregnant female with incidental proteinuria and hypoalbuminemia. We made a diagnosis of nephrotic syndrome at 11 week gestation. Renal biopsy revealed iMN using predominant granular staining of IgG4 along the glomerular basement membrane. No secondary cause was identified. Oral glucocorticoid therapy was started from 17 week gestation and induced complete remission at 28 week gestation. A healthy infant was born at 38 week gestation. Glucocorticoid therapy was stopped postpartum without MN relapse. Anti-PLA2R antibody was later found to be positive using serum reserved from before treatment. In conclusion, we presented the case of a pregnant woman with iMN and anti-PLA2R antibodies, whose nephrotic syndrome was successfully controlled with oral glucocorticoids to reach complete remission, even after tapering off the medication. Pregnancy per se might be associated with iMN onset.


Similar content being viewed by others
References
Umans JG. Obstetric nephrology: preeclampsia—the nephrologist’s perspective. Clin J Am Soc Nephrol. 2012;7(12):2107–13.
Nevis IF, Reitsma A, Dominic A, McDonald S, Thabane L, Akl EA, et al. Pregnancy outcomes in women with chronic kidney disease: a systematic review. Clin J Am Soc Nephrol. 2011;6(11):2587–98.
Piccoli GB, Cabiddu G, Attini R, Vigotti FN, Maxia S, Lepori N, et al. Risk of Adverse Pregnancy Outcomes in Women with CKD. J Am Soc Nephrol. 2015;26(8):2011–22.
Yokoyama H, Taguchi T, Sugiyama H, Sato H. Membranous nephropathy in Japan: analysis of the Japan Renal Biopsy Registry (J-RBR). Clin Exp Nephrol. 2012;16(4):557 – 63.
Ronco P, Debiec H. Pathophysiological advances in membranous nephropathy: time for a shift in patient’s care. Lancet. 2015;385(9981):1983–92.
Beck LH Jr, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, et al. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med. 2009;361(1):11–21.
Al-Rabadi L, Ayalon R, Bonegio RG, Ballard JE, Fujii AM, Henderson JM, et al. Pregnancy in a patient with primary membranous nephropathy and circulating anti-PLA2R antibodies: a case report. Am J Kidney Dis. 2016;67(5):775–8.
Timmermans SA, Damoiseaux JG, Heerings-Rewinkel PT, Ayalon R, Beck LH Jr, Schlumberger W, et al. Evaluation of anti-PLA2R1 as measured by a novel ELISA in patients with idiopathic membranous nephropathy: a cohort study. Am J Clin Pathol. 2014;142(1):29–34.
Akiyama S, Akiyama M, Imai E, Ozaki T, Matsuo S, Maruyama S. Prevalence of anti-phospholipase A2 receptor antibodies in Japanese patients with membranous nephropathy. Clin Exp Nephrol. 2015;19(4):653–60.
Ope-Adenuga S, Moretti M, Lakhi N. Management of membranous glomerulonephritis in pregnancy: a multidisciplinary challenge. Case Rep Obstet Gynecol. 2015;2015:839376.
Hofstra JM, Beck LH Jr, Beck DM, Wetzels JF, Salant DJ. Anti-phospholipase A2 receptor antibodies correlate with clinical status in idiopathic membranous nephropathy. Clin J Am Soc Nephrol. 2011;6(6):1286–91.
Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A, et al. Proteomics. Tissue-based map of the human proteome. Science. 2015;347(6220):1260419.
Stojan G, Baer AN. Flares of systemic lupus erythematosus during pregnancy and the puerperium: prevention, diagnosis and management. Expert Rev Clin Immunol. 2012;8(5):439 – 53.
Fervenza FC, Abraham RS, Erickson SB, Irazabal MV, Eirin A, Specks U, et al. Rituximab therapy in idiopathic membranous nephropathy: a 2-year study. Clin J Am Soc Nephrol. 2010;5(12):2188–98.
Piccoli GB, Daidola G, Attini R, Parisi S, Fassio F, Naretto C, et al. Kidney biopsy in pregnancy: evidence for counselling? A systematic narrative review. BJOG. 2013;120(4):412 – 27.
Packham DK, North RA, Fairley KF, Whitworth JA, Kincaid-Smith P. Membranous glomerulonephritis and pregnancy. Clin Nephrol. 1987;28(2):56–64.
Lindheimer MD, Katz AI. Gestation in women with kidney disease: prognosis and management. Bailliere’s Clin Obstet Gynaecol. 1994;8(2):387–404.
Shiiki H, Saito T, Nishitani Y, Mitarai T, Yorioka N, Yoshimura A, et al. Prognosis and risk factors for idiopathic membranous nephropathy with nephrotic syndrome in Japan. Kidney Int. 2004;65(4):1400–7.
Mitchell K, Kaul M, Clowse MEB. The management of rheumatic diseases in pregnancy. Scand J Rheumatol. 2010;39(2):99–108.
Paziana K, Del Monaco M, Cardonick E, Moritz M, Keller M, Smith B, et al. Ciclosporin use during pregnancy. Drug Saf. 2013;36(5):279 – 94.
Friedrichs B, Tiemann M, Salwender H, Verpoort K, Wenger MK, Schmitz N. The effects of rituximab treatment during pregnancy on a neonate. Haematologica. 2006;91(10):1426.
Acknowledgements
We thank Dr. Shinichi Akiyama (Department of Nephrology, Graduate School of Medicine, Nagoya University) for measuring anti-PLA2R antibody, and Dr. Sachiko Minamiguchi (Department of Diagnostic Pathology, Kyoto University Hospital) for histological diagnosis. We also thank Mr. Chris Rowthorn for the English language review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Yanagita is on the advisory board of Astellas and receives research grants from Astellas, Chugai, Daiichi Sankyo, Fujiyakuhin, Kyowa Hakko Kirin, Mitsubishi Tanabe Pharma Corporation, MSD, Nippon Boehringer Ingelheim, and Torii. The other authors declare no conflicts of interest.
Ethical approval
This study was exempted from institutional review board approval, because it was a case review. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Written informed consent was obtained from the patient for publication of this case and any accompanying images.
About this article
Cite this article
Uchino, E., Takada, D., Mogami, H. et al. Membranous nephropathy associated with pregnancy: an anti-phospholipase A2 receptor antibody-positive case report. CEN Case Rep 7, 101–106 (2018). https://doi.org/10.1007/s13730-018-0304-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-018-0304-7