Abstract
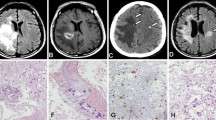
Retinal vasculopathy with cerebral leukodystrophy (RVCL) is a rare autosomal dominant systemic microvascular disease. Neurological disorders and visual disturbance are highlighted as manifestations of RVCL; however, there are few reports focused on nephropathy. Herein, we describe detailed renal histopathological findings in a daughter and father with RVCL, proven by TREX1 genetic analysis. A kidney biopsy of the daughter, 35-year-old with asymptomatic proteinuria, revealed unique and various glomerular changes. Atypical double contour (not tram track-like) of the capillary wall was widely found, an apparent characteristic finding. Glomerular findings were varied due to a combination of new and old segmental mesangial proliferative changes, mesangiolysis, and segmental glomerulosclerosis-like lesions; these changes may be related to endothelial cell damage. Collapsed tufts were also found and thought to be the result of ischemia due to arterial changes. Glomerular findings in a kidney biopsy of the father revealed similarity to the daughter’s glomerulus at a relatively advanced stage, but the degree of variety in the glomerular findings was much less. Kidney biopsy findings suggesting endothelial cell damage of unknown etiology need to be considered for possible RVCL.



Similar content being viewed by others
References
Richards A, van den Maagdenberg AM, Jen JC, et al. C-terminal truncations in human 3′-5′ DNA exonuclease TREX1 cause autosomal dominant retinal vasculopathy with cerebral leukodystrophy. Nat Genet. 2007;39:1068–70.
Stam AH, Kothari PH, Shaikh A, et al. Retinal vasculopathy with cerebral leukoencephalopathy and systemic manifestations. Brain. 2016;139:2909–22.
Fagerberg L, Hallström BM, Oksvold P, et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol Cell Proteomics. 2014;13:397–406.
Grand MG, Kaine J, Fulling K, et al. Cerebroretinal vasculopathy. A new hereditary syndrome. Ophthalmology. 1988;95:649–59.
Terwindt GM, Haan J, Ophoff RA, et al. Clinical and genetic analysis of a large Dutch family with autosomal dominant vascular retinopathy, migraine and Raynaud’s phenomenon. Brain. 1998;121:303–16.
Jen J, Cohen AH, Yue Q, et al. Hereditary endotheliopathy with retinopathy, nephropathy, and stroke (HERNS). Neurology. 1997;49:1322–30.
Kolar GR, Kothari PH, Khanlou N, Jen JC, Schmidt RE, Vinters HV. Neuropathology and genetics of cerebroretinal vasculopathies. Brain Pathol. 2014;24:510–8.
Cohn AC, Kotschet K, Veitch A, Delatycki MB, McCombe MF. Novel ophthalmological features in hereditary endotheliopathy with retinopathy,nephropathy and stroke syndrome. Clin Exp Ophthalmol. 2005;33:181–3.
Rambaud JC, Galian A, Touchard G, et al. Digestive tract and renal small vessel hyalinosis, idiopathic nonarteriosclerotic intracerebral calcifications, retinal ischemic syndrome, and phenotypic abnormalities. A new familial syndrome. Gastroenterology. 1986;90:930–8.
Winkler DT, Lyrer P, Probst A, et al. Hereditary systemic angiopathy (HSA) with cerebral calcifications, retinopathy, progressive nephropathy, and hepatopathy. J Neurol. 2008;255:77–88.
Vodopivec I, Oakley DH, Perugino CA, Venna N, Hedley-Whyte ET, Stone JH. A 44-year-old man with eye, kidney, and brain dysfunction. Ann Neurol. 2016;79:507–19.
Acknowledgements
This study was partially supported by a Grant ‘Initiative on Rare and Undiagnosed Diseases’ (to I.N.) from Japan Agency for Medical Research and Development.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have declared no competing interests.
Ethical statement
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Informed consent was obtained.
About this article
Cite this article
Tsubata, Y., Morita, T., Morioka, T. et al. Renal histopathological findings of retinal vasculopathy with cerebral leukodystrophy. CEN Case Rep 7, 83–89 (2018). https://doi.org/10.1007/s13730-017-0300-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-017-0300-3