Abstract
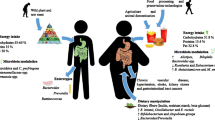
A possible causal role of the gut microbiota in human obesity is capturing interest. Recent experimental evidence and mechanistic hypotheses suggest that a ‘dysbiotic’ large bowel microbiota, induced mainly by poor diet, increases dietary energy bioavailability and storage in the host. However, research findings in both animals and humans are inconsistent and whether an altered gut microbiota meaningfully impacts host energetics remains an open question. Future intervention studies must control diet and other lifestyle factors that profoundly influence the composition and activity of the intestinal microbiota to define its potential role in and contribution to the human obesity problem.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–81.
Huang T, Hu FB. Gene-environment interactions and obesity: recent developments and future directions. Medical Genet. 2015;8 Suppl 1:S2–7.
Goodrich JK, Waters JL, Poole AC, Sutter JL, Koren O, Blekhman R. Human genetics shape the gut microbiome. Cell. 2014;159:789–99.
Parks BW, Nam E, Org E, Kostem E, Norheim F, Hui ST, et al. Genetic control of obesity and gut microbiota composition in response to high-fat, high-sucrose diet in mice. Cell Metab. 2013;17:141–52.
Friedman JM. Causes and control of excess body fat. Nature. 2009;459:340–2.
Turnbaugh PJ, Backhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3:213–23.
Ley RE, Backhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI. Obesity alters gut microbial ecology. Proc Natl Acad Sci U S A. 2005;102:11070–5.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–31.
Backhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A. 2004;101:15718–23.
Heinken A, Thiele I. Systems biology of host–microbe metabolomics. WIREs Syst Biol Med. 2015;7:195–219.
Burcelin R, Serino M, Chabo C, Garidou L, Pomie C, Courtney M, et al. Metagenome and metabolism: the tissue microbiota hypothesis. Diabetes Obes Metab. 2013;15 Suppl 3:61–70.
Romani L, Zelante T, Palmieri M, Napolioni V, Picciolini M, Velardi A. The cross-talk between opportunistic fungi and the mammalian host via microbiota’s metabolism. Semin Immunopathol. 2015;37:163–71.
Huttenhower C, Gevers D, Knight R, Abubucker S, Badger JH, Chinwalla AT, et al. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207–14.
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, et al. Diversity of the human intestinal microbial flora. Science. 2005;308:1635–8.
Huse SM, Ye YZ, Zhou YJ, Fodor AA. A core human microbiome as viewed through 16S rRNA sequence clusters. PLoS One. 2012;7:e34242.
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505:559–63. Intervention study showing the considerable capacity of the large bowel (fecal) microbiota to respond to dietary change.
Simoes CD, Virtanen KA, Maukonen J, Scott KP, Pietilainen KH, Saarela M. Impact of a very low-energy diet on the fecal microbiota of obese individuals. Eur J Nutr. 2014;53:1421–9.
Voreades N, Kozil A, Weir TL. Diet and the development of the human intestinal microbiome. Front Microbiol. 2014;5:494.
Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut. 2015;64(1):93–100.
Nielsen DS, Krych L, Buschard K, Hansen CHF, Hansen AK. Beyond genetics. Influence of dietary factors and gut microbiota on type 1 diabetes. FEBS Lett. 2015;588:4234–43.
Tai N, Wong FS, Wen L. The role of gut microbiota in the development of type 1, type 2 diabetes mellitus and obesity. Rev Endocr Metab Disord. 2015;16:55–65.
Ridaura VK, Jeremiah JF, Rey FE, Cheng J, Duncan AE, Kau AL, et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science. 2013;341:1241214.
Fei N, Zhao L. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germ free mice. ISME J. 2013;7:880–4.
Fleissner CK, Huebel N, Abd El-Bary MM, Loh G, Klaus S, Blaut M. Absence of intestinal microbiota does not protect mice from diet-induced obesity. Br J Nutr. 2010;104:919–29.
Zhao L. The gut microbiota and obesity: from correlation to causality. Nat Rev Microbiol. 2013;11:639–47.
Hildebrandt MA, Hoffmann C, Sherrill-Mix SA, Keilbaugh SA, Hamady M, Chen Y-Y, et al. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology. 2009;137:1716–24.
Zhang CH, Zhang MH, Pang XY, Zhao YF. Structural resilience of the gut microbiota in adult mice under high-fat dietary perturbations. ISME J. 2012;6:1848–57.
Krych L, Hansen CHF, Hansen AK, van den Berg FWJ, Nielsen DS. Quantitatively different, yet qualitatively alike: a meta-analysis of the mouse core gut microbiome with a view towards the human gut microbiome. PLoS One. 2013;8:e62578.
Le Leu RK, Conlon MA, Bird AR, Clarke JM. Housing experimental rats in solid-based cages with digestible bedding may confound outcomes of nutritional studies. J Sci Food Agric. 2015;95:2155–8. Simple feeding trial demonstrating unequivocally that housing arrangements for experimental animals may compromise study outcomes.
Hariri N, Thibault L. High-fat diet-induced obesity in animal models. Nutr Res Rev. 2010;23:270–99.
Buettner R, Scholmerich J, Bollheimer LC. High-fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity. 2007;15:798–808.
Commerford SR, Pagliassotti MJ, Melby CL, Wei Y, Hill J. Inherent capacity for lipogenesis or dietary fat retention is not increased in obesity-prone rats. Am J Physiol Regul Integr Comp Physiol. 2001;280:R1680–7.
Million M, Angelakis E, Paul M, Armougom F, Leibovici L, Raoult D. Comparative meta-analysis of the effect of Lactobacillus species on weight gain in humans and animals. Microb Pathog. 2012;53:100–18.
Santacruz A, Collado MC, Garcıa-Valdes L, Segura MT, Martın-Lagos JA, Anjos T, et al. Gut microbiota composition is associated with body weight, weight gain and biochemical parameters in pregnant women. Br J Nutr. 2010;104:83–92.
Hu HJ, Park SJ, Jang HN, Choi MG, Park KH, Kang JH, et al. Obesity alters the microbial community profile in Korean adolescents. PLoS One. 2015;10:e0134333.
Schwiertz A, Taras D, Schafer K, Beijer S, Bos NA, Donus C, et al. Microbiota and SCFA in lean and overweight healthy subjects. Obesity. 2010;18:190–5.
Kasai C, Sugimoto K, Moritani I, Tanak J, Oya Y, Inoue H, et al. Comparison of the gut microbiota composition between obese and non-obese individuals in a Japanese population, as analysed by terminal restriction fragment length polymorphism and next-generation sequencing. BMC Gastroenterol. 2015;15:100–10.
Armougom F, Henry M, Vialettes B, Raccah D, Raoult D. Monitoring bacterial community of human gut microbiota reveals an increase in Lactobacillus in obese patients and methanogens in anorexic patients. PLoS One. 2009;4:e7125.
Angelakis E, Armougom F, Amougom F, Million M, Raoult D. The relationship between gut microbiota and weight gain in humans. Future Microbiol. 2012;7:91–109.
Walters WA, Xu Z, Knight R. Meta-analyses of human gut microbes associated with obesity and IBD. FEBS Lett. 2014;588:4223–33. Meta-analysis of a small number of studies that used comparable analytical methodologies. The analysis reveals that fecal microbiota differences between lean and obese are inconsistent across studies, indicative of a small effect size for obesity.
Balamurugan R, George G, Kabeerdoss J, Hepsiba J, Chandragunasekaran AM, Ramakrishna BS. Quantitative differences in intestinal Faecalibacterium prausnitzii in obese Indian children. Br J Nutr. 2010;103:335–8.
Nadal I, Santacruz A, Marcos A, Warnberg J, Garagorri M, Moreno LA. Shifts in clostridia, bacteroides and immunoglobulin-coating fecal bacteria associated with weight loss in obese adolescents. Int J Obes. 2009;33:758–67.
Duncan SH, Belenguer A, Holtrop G, Johnstone AM, Flint HJ, Loblet GE. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl Environ Microbiol. 2007;73:1073–8.
Salonen A, Lahti L, Salojarvi J, Holtrop G, Korpela K, Duncan S. Impact of diet and individual variation on intestinal microbiota composition and fermentation products in obese men. ISME J. 2014;8:2218–30.
Duncan SH, Lobley GE, Holtrop G, Ince J, Johnstone AM, Louis P, et al. Human colonic microbiota associated with diet, obesity and weight loss. Int J Obes. 2008;32:1720–4.
Brinkworth GD, Noakes M, Clifton PM, Bird AR. Comparative effects of very low-carbohydrate, high-fat and high-carbohydrate, low-fat weight-loss diets on bowel habit and faecal short-chain fatty acids and bacterial populations. Br J Nutr. 2009;101:1493–502.
Prideaux L, Kang S, Wagner J, Buckley M, Mahar JE, De Cruz P, et al. Impact of ethnicity, geography, and disease on the microbiota in health and inflammatory bowel disease. Inflamm Bowel Dis. 2013;19:2906–18.
Dominianni C, Sinha R, Goedert JJ, Pei ZH, Yang LY, Hayes RB, et al. Sex, body mass index, and dietary fiber intake influence the human gut microbiome. PLoS One. 2015;10:e0124599.
Maurice CF, Haiser HJ, Turnbaugh PJ. Xenobiotics shape the physiology and gene expression of the active gut microbiome. Cell. 2013;152:39–50.
Cani PD, Everard A. Keeping gut lining at bay: impact of emulsifiers. Trends Endocrinol Metab. 2015;26:273–4.
Thaiss CA, Zeevi D, Levy M, Zilverman-Shapira G, Suez J, Tengeler AC, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159:514–29. Comprehensive study in mice and humans showing that gut microbiota composition and function fluctuates largely in response to daily feeding rhythms, patterns and behaviours. Disrupted circadian rhythmicity impacts the microbiota (dysbiosis) which in turn may have adverse metabolic consequences for the host.
Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2224–60.
Ebbert JO, Elrashidi MY, Jensen MD. Managing overweight and obesity in adults to reduce cardiovascular disease risk. Curr Atheroscler Rep. 2014;16:445.
Rogers CJ, Prabhu KS, Vijay-Kumar M. The microbiome and obesity—an established risk for certain types of cancer. Cancer J. 2014;20(3):176–80.
Kostic AD, Gevers D, Siljander H, Vatanen T, Hyotylainen T, Hamalainen A-M, et al. The dynamics of the human infant gut microbiome in development and in progression toward type 1 diabetes. Cell Host Microbe. 2015;17:260–73.
Zhang XY, Shen DQ, Fang ZW, Jie ZY, Qiu XM, Zhang CF, et al. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS One. 2013;8(8):e71108.
Palmer BF, Clegg DJ. The sexual dimorphism of obesity. Mol Cell Endocinol. 2015;402:113–9.
Ringel Y, Maharshak N, Ringel-Kulka T, Wolber EA, Sartor RB, Carroll IM. High throughput sequencing reveals distinct microbial populations within the mucosal and luminal niches in healthy individuals. Gut Microbes. 2015;6(3):173–81. Observational study showing that the large bowel luminal (fecal) and mucosa-associated microbiota are essentially two distinct ecosystems that differ in microbial diversity and composition.
Faith JJ, McNulty NP, Rey FE, McNulty NP, Rey FE, Gordon JI. Predicting a human gut microbiota’s response to diet in gnotobiotic mice. Science. 2011;333:101–4.
Zhang CH, Zhang MH, Wang SY, Han RJ, Cao YF, Hua WY. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J. 2010;4(2):232–41.
Backhed F, Roswall J, Peng YQ, Feng Q, Jia HJ, Kovatcheva-Datchary P, et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe. 2015;17(5):690–703.
Pacheco AR, Barile D, Underwood MA, Mills DA. The impact of the milk glycobiome on the neonate gut microbiota. Annu Rev Anim Biosci. 2015;3:419–45.
Jost T, Lacroix C, Braegger C, Chassard C. Impact of human milk bacteria and oligosaccharides on neonatal gut microbiota establishment and gut health. Nutr Rev. 2015;7 3(7):426–37.
Wang M, Li M, Wu S, Lebrilla CB, Chapkin RS, Ivanov I, et al. Fecal microbiota composition of breast-fed infants is correlated with human milk oligosaccharides consumed. J Pediatr Gastroenterol Nutr. 2015;60(6):825–33.
Wu GD, Chen J, Hoffmann C, Bittinger K, Chen Y-Y, Keilbaugh SA, et al. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334:105–8.
Bervoets L, Van Hoorenbeeck K, Kortleven I, Van Noten C, Hens N, Vael C. Differences in gut microbiota composition between obese and lean children: a cross-sectional study. Gut Pathog. 2013;5:10.
Conlon MA, Bird AR. The impact of diet and lifestyle on gut microbiota and human health. Nutrients. 2015;7(1):17–44.
Walker AW, Ince J, Duncan SH, Webster LM, Holtrop G, Ze XL. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011;5(2):220–30.
Korpela K, Flint HJ, Johnstone AM, Lappi J, Poutanen K, Dewulf E. Gut microbiota signatures predict host and microbiota responses to dietary interventions in obese individuals. PLoS One. 2014;9(3):e90702.
Le Leu RK, Winter JM, Christophersen C, Young GP, Humphreys KJ, Hu Y, et al. Butyrylated starch intake can prevent red meat induced O6-methyl-2-deoxyguanosine adducts in human rectal tissue: a randomised clinical trial. Br J Nutr. 2015;114(2):220–30.
Bird AR, Usher S, May B, Topping DL, Morell MK. 2012. Resistant starch—measurement, intakes and dietary targets. In: Dietary Fiber and Health. Eds Susan Cho and Nelson Almeida. pp 41–56.
Payne AN, Chassard C, Lacroix C. Gut microbial adaptation to dietary consumption of fructose, artificial sweeteners and sugar alcohols: implications for host-microbe interactions contributing to obesity. Obes Rev. 2012;13(9):799–809.
Gibson GR, Probert HM, Van Loo J, Rastall RA, Roberfroid MB. Dietary modulation of the human colonic microbiota: updating the concept of prebiotics. Nutr Res Rev. 2004;17(2):259–75.
Russell WR, Gratz SW, Duncan SH, Holtrop G, Ince J, Scobbie L, et al. High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am J Clin Nutr. 2011;93(5):1062–72.
Silvester KR, Bingham SA, Pollock JRA, Cummings JH, ONeill IK. Effect of meat and resistant starch on fecal excretion of apparent N-nitroso compounds and ammonia from the human large bowel. Nutr Cancer. 1997;29(1):13–23.
O’Callaghan NJ, Toden S, Bird AR, Topping DL, Fenech M, Conlon MA. Colonocyte telomere shortening is greater with dietary red meat than white meat and is attenuated by resistant starch. Clin Nutr. 2012;31:60–4.
Winter J, Nyskohus L, Young GP, Hu Y, Conlon MA, Bird AR, et al. Inhibition by resistant starch of red meat-induced promutagenic adducts in mouse colon. Cancer Prev Res. 2011;4:1920–8.
Toden S, Bird AR, Topping DL, Conlon MA. Differential effects of dietary whey, casein and soya on colonic DNA damage and large bowel SCFA in rats fed diets low and high in resistant starch. Br J Nutr. 2007;97:535–43.
Maukonen J, Saarela M. Human gut microbiota: does diet matter? Proc Nutr Soc. 2015;74:23–36.
Belobrajdic DP, Bird AR. The potential role of phytochemicals in wholegrain cereals for the prevention of type-2 diabetes. Nutr J. 2013;12:62.
Tuohy KM, Conterno L, Gasperotti M, Viola R. Up-regulating the human intestinal microbiome using whole plant foods, polyphenols, and/or fiber. J Agric Food Chem. 2012;60(36):8776–82.
Martin FPJ, Montoliu I, Nagy K, Moco S, Collino S, Guy P. Specific dietary preferences are linked to differing gut microbial metabolic activity in response to dark chocolate intake. J Proteome Res. 2012;11(12):6252–63.
Muir JG, Yeow EGW, Keogh J, Pizzey C, Bird AR, Sharpe K, et al. Combining wheat bran with resistant starch raised faecal butyrate and lowered phenols in humans. Am J Clin Nutr. 2004;79:1020–8.
Bird AR, Vuaran M, Crittenden R, Hayakawa T, Playne M, Brown I, et al. Comparative effects of a high-amylose starch and a fructooligosaccharide on fecal bifidobacteria numbers and short-chain fatty acids in pigs fed Bifidobacterium animalis. Dig Dis Sci. 2009;54(5):947–54.
Baer DJ, Rumpler WV, Miles CW, Fahey GC. Dietary fiber decreases the metabolizable energy content and nutrient digestibility of mixed diets fed to humans. J Nutr. 1997;127(4):579–86.
Thornton JR, Dryden A, Kelleher J, Losowsky MS. Super-efficient starch absorption—a risk factor for colonic neoplasia. Dig Dis Sci. 1987;32(10):1088–91.
James SL, Christophersen CT, Bird AR, Rosella O, Muir JG, Gibson PR. Abnormal fibre usage in UC in remission. Gut. 2015;64(4):562–70. Randomised controlled trial showing that the gut microbiota of patients with quiescent ulcerative colitis has a reduced capacity to ferment dietary fibres such as resistant starch and nonstarch polysaccharides.
Backhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI. Host-bacterial mutualism in the human intestine. Science. 2005;307:1915–20.
den Besten G, van Eunen K, Groen AK, Venema K, Reijngoud D-J, Bakker BM. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res. 2013;54:2325–40.
Bach Knudsen KE. Microbial degradation of whole-grain complex carbohydrates and impact on short-chain fatty acids and health. Adv Nutr. 2015;6:206–13.
Topping DL, Clifton PM. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev. 2001;81:1031–64.
McOrist AL, Miller RB, Bird AR, Keogh JB, Noakes M, Topping DL, et al. Fecal butyrate levels vary widely among individuals but are usually increased by a diet high in resistant starch. J Nutr. 2011;141:883–9.
Rahat-Rozenbloom S, Fernandes J, Gloor GB, Wolever TMS. Evidence for greater production of colonic short-chain fatty acids in overweight than lean humans. Int J Obes. 2014;38(12):1525–31.
Cox LM, Blaser MJ. Pathways in microbe-induced obesity. Cell Metab. 2013;17(6):883–94.
Hooper B, Spiro A, Stanner S. 30 g of fibre a day: an achievable recommendation? Nutr Bull. 2015;40:118–29.
Leach JD. Evolutionary perspective on dietary intake of fibre and colorectal cancer. Eur J Clin Nutr. 2007;61:140–2.
Hill JO, Wyatt HR, Reed GW, Peters JC. Obesity and environment: where do we go from here? Science. 2003;299:854–5.
Graham C, Mullen A, Whelan K. Obesity and the gastrointestinal microbiota: a review of associations and mechanisms. Nutr Rev. 2015;73(6):376–85.
Yang Y, Zhao L-G, Wu Q-J, Ma X, Xiang Y-B. Association between dietary fibre and lower risk of all-cause mortality: a meta-analysis of cohort studies. Am J Epidemiol. 2015;181(2):83–91.
Higgins JA. Resistant starch and energy balance: impact on weight loss and maintenance. Crit Rev Food Sci Nutr. 2014;54:1158–66.
Keenan MJ, Zhou J, Hegsted M, Pelkman C, Durham HA, Coulon DB, et al. Role of resistant starch in improving gut health, adiposity, and insulin resistance. Adv Nutr. 2015;6:198–205.
Zhou J, Martin RJ, Tulley RT, Raggio AM, Shen L, Lissy E, et al. Failure to ferment dietary resistant starch in specific mouse models of obesity results in no body fat loss. J Agric Food Chem. 2009;57(19):8844–51.
Belobrajdic DP, King RS, Christophersen CT, Bird AR. Dietary resistant starch dose-dependently reduces adiposity in obesity-prone and obesity–resistant male rats. Nutr Metab. 2012;9:93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Anthony R. Bird and Michael A. Conlon declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Diabetes and Obesity
Rights and permissions
About this article
Cite this article
Bird, A.R., Conlon, M.A. Obesity, Diet and the Gut Microbiota. Curr Nutr Rep 4, 340–347 (2015). https://doi.org/10.1007/s13668-015-0146-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13668-015-0146-2