Abstract
• Key message
Decomposition of forest herb species litter was not always completed in less than a year and was not always faster than decomposition of tree leaf litter in an oak-hornbeam forest in Western Poland. Litter decomposition of herbaceous plants is connected with their life strategy and functional traits of their leaves.
• Context
Forest understories are frequently ignored in ecological research on decomposition, although they play an important role in biomass and nutrient cycling in forest ecosystems.
• Aims
We hypothesized that the decomposition process of herbaceous species was completed in less than a year, as opposed to tree leaf litter. The second aim of our study was to determine if life strategy affects the rate of litter decomposition.
• Methods
We performed the decomposition experiment in the oak-hornbeam forest in Czmoń (Western Poland) using the litter bag method to determine decay constants (k) for all species studied. The influence of species identity, functional group, and functional traits of leaves and other effects on the decomposition process was assessed.
• Results
The decomposition process was significantly dependent on the functional group of plants, time of exposure in the field, species identity, and precipitation. We found a significant correlation between leaf traits and decay rates of the species studied.
• Conclusion
Litter decomposition of herbaceous plants is connected with their life strategy and functional traits of their leaves in an oak-hornbeam forests.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.1 Introduction
Forest understories are frequently ignored in ecological research on productivity, probably due to their relatively small (ca. 1–2%) contribution to total plant biomass of ecosystems. However, herbaceous species have great importance for forest nutrient retention (Bormann et al. 1968; Muller and Bormann 1976; Muller 2014). The herb layer can provide up to 16% of annual litter fall in forests (Gilliam 2007; Muller 2014). Moreover, in temperate deciduous forests herbaceous species supply litter to the litter horizon continuously during the growing season, thus not only in an autumnal pulse like tree leaf litter (Wise and Shaefer 1994). Additionally, foliar concentrations of some nutrients (N, P, K, Mg) are higher in herbaceous than in tree species (Gilliam 2007). It is particularly important that, on average, the herbaceous layer contains 80% of forest plant species biodiversity and rare herb species can be useful as indicators of biodiversity or site quality (Gilliam 2007; Spyreas and Matthews 2006). Moreover, by competition with natural woody plant species regeneration, the herb layer influences or even determines overstory composition (Baraloto et al. 2005). The biomass of herb species is also an important source of food for animals (Dzięciołowski 1970).
The decomposition of organic matter is a crucial process in ecosystem functioning because it is responsible for replenishing the pool of soil nutrients available to plants, returning huge amounts of carbon dioxide to the atmosphere, and creating long-term storage of carbon as soil organic matter (Berg and McClaugherty 2014). In recent decades, the number of studies describing organic matter decomposition has increased rapidly. An important subset of those studies has been experiments comparing decomposition rates of litter from different species in common garden conditions (e.g., Hobbie 1996; Hobbie et al. 2006). These kinds of studies are keys for estimating species effects on litter decomposition and to create ecosystem models that help to illuminate their inner organization (Hobbie et al. 2006). Moreover, they are crucial for understanding the consequences of changes in plant biodiversity for ecosystem functioning (Chapin 2003; Handa et al. 2014; Liang et al. 2016). The majority of decomposition experiments in forest ecosystems have focused on woody species foliage (e.g., Dziadowiec 1987, 1990; Hobbie et al. 2006; Horodecki and Jagodziński 2017, 2019; Horodecki et al. 2019; Jackson et al. 2013; Jurkšienė et al. 2017) and wood (e.g., Bantle et al. 2014; Harmon et al. 2000). The limited studies available on decomposition in deciduous forests have focused on herb species decomposition rates (Halabuk and Gerhátová 2011; Mayer 2008; Rodgers et al. 2008; Wise and Shaefer 1994) and interactions with soil organisms (Wise and Shaefer 1994). Most of those studies noted that herb species litter in temperate deciduous forests was fully decomposed within 6 months after senescence (Halabuk and Gerhátová 2011; Mayer 2008; Muller 2014; Rodgers et al. 2008). Only herb species biomass dominated by sedges, shrubs, and mosses (Hobbie 1996) or ferns and shrubs (MacLean and Wein 1978) required more than 1 year to decompose completely.
Decomposition rates depend on climate, litter quality, and communities of soil organisms (Berg and McClaugherty 2014; Cornwell et al. 2008; Kamczyc et al. 2019; Urbanowski et al. 2018). Microclimates in temperate deciduous forest understories vary during the growing season, due to changing solar zenith angle and canopy phenology (Noda et al. 2015). Seasonal changes in the availability of light in the understory of deciduous forests are strictly connected with changes in other microclimatic conditions, like temperature and moisture (Graves 1990). This seasonality results in the occurrence of different phenological strategies among herb species in deciduous forests, including spring ephemeral, summer-green, winter-green, and evergreen species (Neufeld and Young 2014; Uemura 1994). These strategies represent a kind of niche separation, which allows the species to utilize habitat resources efficiently and avoid competition (Díaz and Cabido 2001; Jagodziński et al. 2016; Scherer-Lorenzen 2008).
Plant adaptations to variation in the physical environment is reflected in plant biological traits and connected with their different functions within an ecosystem. They determine the potential of a given species to establish or persist under any given set of environmental conditions (Díaz and Cabido 2001). The connection between plant adaptation strategies and decomposability is crucial for understanding vegetation–soil feedbacks. There is a general concept that functional traits of leaves (Leaf Economic Spectrum; Wright et al. 2005) influence leaf litter decomposition and nutrient release (Cornelissen and Thompson 1997; Zukswert and Prescott 2017). According to this concept species of plants with conservative resource strategies (high leaf dry matter contents (LDMC)), low nutrient concentrations, and low specific leaf area (SLA), decompose slower than fast-growing, acquisitive species (Díaz et al. 2016; Freschett et al. 2010, 2012; Wright et al. 2005). Studies that conceptualize decomposition within the tradeoff between defense and photosynthetic production have been frequently conducted for leaves of trees (Makkonen et al. 2012; Melilo et al. 1982; Zukswert and Prescott 2017) or herbaceous species of grasslands (Cornelissen and Thompson 1997; Cornwell et al. 2008). Previous research indicated that spring ephemerals have typical short-lived, sun-type leaves. They have the greatest metabolic activity, the highest rates of photosynthesis, and leaf N contents among all phenological groups of herbs (Muller 2014; Rothstein and Zak 2001). Generally, foliar nutrient concentrations of herbs are higher than overstory species (Muller 2014). In many previous studies, it was found that decomposition rate was strongly positively correlated with leaf N concentration and negatively with leaf life span (Bakker et al. 2011; Cornwell et al. 2008; Wright et al. 2005). To our knowledge, there is no information about differences in decomposition rates among species representing different life strategies of forest understory plant species in a temperate deciduous forest. Although functional traits of plants are nowadays widely accepted as potentially powerful indicators of the ecology of species, only a few forest understory species have been included in studies (Ma et al. 2010; Poorter and De Jong 1999; Rawlik and Jagodziński 2020; Rawlik et al. 2018; Rothstein and Zak 2001; Wang et al. 2010), and most frequently these species were pooled with other herbaceous plants. Many studies have demonstrated that plant traits have afterlife effects via their impacts on decomposition rates, however, it is still not clear whether patterns found on a global scale are reproducible at local scales, in specific growth forms (Kleyer et al. 2018), or different organs (Hobbie 2015).
Many studies researching correlations among plant traits or their correlations with decomposition included only one organ (e.g., stems or leaves), avoiding a whole-plant perspective (Kleyer and Minden 2015). These studies concerned homogeneous components of plants, usually focused on leaves (Hobbie 1992), even though a large part of herbaceous litter comes from stems or roots. Differences in structural and physiological traits between organs, connected with their different biological functions, might cause differential decomposability (Freschet et al. 2012). In this study, we wanted to know the true biological rate of biomass decomposition of the species included; therefore, we used mixed aboveground biomass (including leaves and shoots). Moreover, we decided to compare decomposition rates of leaves and blooming shoots (material dominated by shoots) of one herb species (Aegopodium podagraria) to assess differences in decomposition rates of different organs.
Our primary objective was to compare decomposition rates of oak-hornbeam forest herb species with different ecological requirements, phenology, and life-history traits. The second aim of our study was to compare the decomposition rates of these plants with leaf litter of tree species occurring in the overstory. We hypothesized that (1) biomass of herbaceous plants in a temperate deciduous forest decomposes completely in less than a year (decomposition constants k > 1) (Muller 2014 and literature cited therein). We also hypothesized that (2) spring ephemerals decompose faster than summer and autumn species (Jagodziński et al. 2016; Neufeld and Young 2014), and (3) herb species biomass decomposes faster than that of tree leaves (Mayer 2008; Muller 2014).
2 Materials and methods
2.1 Study area
This study was conducted in the Czmoń Forest (Babki Forest District, W Poland; 52° 09′ 05.76″ N, 17° 03′ 00.68″ E; 76 m a.s.l.), in the temperate climatic zone. Mean annual temperature in this area was 8.7 °C, and mean annual precipitation was 514 mm in 1971–2010, and 9.2 °C and 535 mm in 2001–2010 (Central Statistical Office 2020). According to meteorological data from a nearby meteorological station (Institute of Dendrology, Polish Academy of Sciences, Kórnik; 52° 14′ 41″ N, 17° 06′ 03″ E; 10.5 km from the study area) the mean annual temperature in 2011–2013 was 9.1 °C (Fig. 6a in the Appendix) and mean annual precipitation was 573 mm (Fig. 7 in the Appendix). In the year preceding the experiment (2011), the mean annual temperature was 9.5 °C, which was higher than during a typical year in the study area. More specifically, during the 12 months preceding the experiment (June 2011–May 2012), the average monthly temperature was as follows: 19.0 °C in June, 18.2 °C in July, 19.7 °C in August, 14.7 °C in September, 9.1 °C in October, 3.2 °C in November, 3.3 °C in December, 0.4 °C in January, − 4.6 °C in February, 5.8 °C in March, 9.0 °C in April, 15.2 °C in May. In the year preceding the experiment (2011), the annual precipitation was 431 mm, which was less than during a typical year in the study area. In the 12 months preceding the experiment (June 2011–May 2012), the monthly sum of precipitation was as follows: 59.3 mm in June, 108.1 mm in July, 78.8 mm in August, 24.7 in September, 26.8 mm in October, 0.7 mm in November, 48.0 mm in December, 74.5 mm in January, 44.2 mm in February, 10.3 mm in March, 30.5 mm in April, 40.8 mm in May. Air temperature at the meteorological station was measured at the level of 2 m. During the experiment (28 May 2012–26 October 2013), we also measured temperatures at the ground level every hour using four data loggers evenly distributed within the stand (HOBO U23-001 Pro v2 Temperature/Relative Humidity, Onset Computer Corporation, Bourne, Massachusetts, USA) (Fig. 6b in the Appendix). Since we compared the decomposition rates of the species studied after ca. 6 months from the beginning of the experiment, we show detailed temperature and precipitation conditions for this period (Table 4 in the Appendix). There were differences in average air temperatures, ground temperatures, and total precipitation among the five dates of the field experiment.
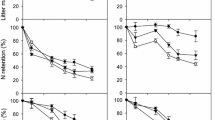
Decomposition (percentage of mass loss) for all plant species studied during the experiment by date during the years 2012 and 2013. Explanations of abbreviations: AcePse - Acer pseudoplatanus; AdoMos - Adoxa moschatellina; AegPodL - Aegopodium podagraria leaves; AegPodS - Aegopodium podagraria shoots; AllPet - Alliaria petiolata; AneNem - Anemone nemorosa; AneRan - Anemone ranunculoides; AsaEur - Asarum europaeum; CarBet - Carpinus betulus; CorAve - Corylus avellana; CorCav - Corydalis cava; FicVer - Ficaria verna; FraExc - Fraxinus excelsior; GalLut - Galeobdolon luteum; MaiBif - Maianthemum bifolium; MerPer - Mercurialis perennis; ParQua - Paris quadrifolia; QueRob - Quercus robur; StaSyl - Stachys sylvatica; UrtDio - Urtica dioica
Decomposition (mean percentage of initial mass loss) of all functional groups of plants studied during the experiment. *Time of the exposition in the field = time from the start of the experiment, which varied in time of the year for different functional groups as shown in Fig. 1
The study area was located in a deciduous forest complex, covered by a 97-year-old oak-hornbeam stand (Table 5 in the Appendix). Detailed descriptions of the study area were given by Horodecki et al. (2014), Rawlik et al. (2015), and Wiczyńska et al. (2013).
We determined soil particle-size distribution, soil pH in H2O and in 1 M KCl, physicochemical soil characteristic (Table 6 in the Appendix). These properties were measured in two soil samples collected in October 2013.
The number of sample plots and growing seasons included in the study was limited by the high labor demand for sampling senescent herbaceous plants and preparing the litter bag experiment. We are aware that chances to generalize results from our study are limited by the low replicability (one sample plot and one growing season). However, despite the lack of replications, our assessments of the effects of life-history traits on biomass decomposition, inclusion of stem and leaf biomass to estimate ecologically relevant decomposition of aboveground herbaceous biomass, and comparisons of herbaceous biomass with tree leaf litter decomposition, gives unique insight into the complexity of decomposition at this site. In addition, our study provides novel data that could be used in designing further studies.
2.2 Species studied
We chose 14 vascular plant species, which are the most abundant in the understories of fertile deciduous forests in Central Europe and present within the research site (Ellenberg 1988), i.e., Adoxa moschatellina L., Aegopodium podagraria L., Alliaria petiolata (Bieb.) Cav. et Grande, Anemone nemorosa L., Anemone ranunculoides L., Asarum europaeum L., Corydalis cava L. (Schweigger et Koerte), Ficaria verna Huds., Galeobdolon luteum Hudson, Maianthemum bifolium L. (F.W. Schmidt), Mercurialis perennis L., Paris quadrifolia L., Stachys sylvatica L., and Urtica dioica L. Additionally, we studied the five most abundant tree and shrub species in the overstory and undergrowth of the forest stand, i.e., Acer pseudoplatanus L., Carpinus betulus L., Corylus avellana L., Fraxinus excelsior L., and Quercus robur L. We chose these herb and woody plant species because they have a strong influence on ecosystem functioning, due to the fact that abundance of species is correlated with their importance to ecosystem function (Grime 2001), and because these species differ in their ecological requirements, phenology, and life-history traits (Table 1).
2.3 Methods
We harvested herbaceous plants during one growing season (2012) at the time when most of the plants within each population began senescing. In most cases, we collected senescent aboveground biomass (mixed leaves and stems). In the case of A. podagraria, we collected leaves and blooming shoots separately. During autumn 2012, we collected freshly fallen leaves of the mentioned tree species from stands in the vicinity of the sample plot. After collection material was dried in the laboratory at 65 °C to a constant weight in a dryer with forced air circulation (UFE 600, Memmert GmbH+Co.KG, Germany). Dried litter was weighed using BP 210 S (http://www.sartorius.dataweigh.com) and Mettler Toledo PG 1003-S (http://www.mt.com) scales with an accuracy of 0.001 g and placed into “litter bags” made of fiberglass netting (15 cm × 15 cm) with a mesh size of 1 mm.
Those bags were filled with 3.9–4.2 g (U. dioica), 1.9–3.2 (the remaining herb species), or 8.0–8.3 g (tree leaves) of litter and labeled. In total, 2658 litter bags were placed in the forest at five dates, according to the time of senescence of most plants of the particular taxon (Table 1). The decision on mesh size for our experiment took into account that it can modify activities of mesofauna and macrofauna, microclimatic conditions, and material leaching out of litter bags. Results of choosing different mesh sizes were shown by many previous methodological studies (Bradford et al. 2002; Harmon et al. 1999; Slade and Riutta 2012; Wise and Shaefer 1994). Thus, our choice was a compromise. The masses put in litter bags differed for particular species to avoid excessive compaction of the material. Moreover, the amount of material was adjusted to each material type, to standardize litter densities and textures inside the bags. For all herbaceous species, we placed a mix of leaves and shoots in litter bags, in proportions similar to what occurs in specimens in the field.
For A. europaeum, we decided to start the experiment according to the time of senescence of most of last-year’s leaves. For G. luteum, we decided to start the experiment according to the time of senescence of most of the current year’s leaves. Our main criterion (time of senescence of most plants of the particular taxon) was used to separate spring ephemerals and summer-green plants. Summer-green plants were further separated into two groups, mid-summer, and autumn-senescing plants. We decided to use different starting times of the experiment for particular groups of plants because the aim of our studies was to find real, biological rates of decomposition of the species studied. We established one research plot (ca. 0.25 ha in total). Litter bags were randomly placed on this research plot in six sets of samples. Distance between every set of samples was ca. 10 m. On the research plot we established samples of all species harvested in each term of collection (six samples per collection term).
Six randomly selected litter bags of each species were collected every week for herb species or every 2 weeks for tree species (Rawlik et al. 2020). The time of exposition in the field was generally about one half year (175 or 182 days) for herb species and 364 days for leaves of trees. After drying at 65 °C to a constant weight in a dryer with forced air circulation (UFE 600, Memmert GmbH+Co.KG, Germany), litter was removed from bags, and cleaned to remove sand, fungi and roots, and then weighed. The mass loss of the plant material was determined systematically during the experiment. For some species, we noticed that the decomposition rate was lower than previously assumed, and thus, we decided to extend the period of litter collection (see Table 7 in the Appendix). Thus, we decided to continue the experiment for longer durations for blooming shoots of A. podagraria (406 days) and M. bifolium (238 days), and leaves of A. podagraria (203 days), M. perennis (203 days), S. sylvatica (203 days), and U. dioica (203 days).
We are aware of the limitations of the litter bag method, due to artifacts the method has compared to real biological decomposition rates. Mainly, drying plant material can slow the decomposition rate because the chemical composition of samples may be changed, making dried material less attractive for consumers. Secondly, putting material into litter bags influences decomposition. In this context chosen mesh size is important. Mesh size is important because of the exclusion of macrofauna and different impacts of microclimate, and thus biological activity and control on handling effects and increased exposure to abiotic factors (Bradford et al. 2002). The litter bag mesh size chosen for our experiment was a compromise between the smallest mesh, which inhibits meso- and macrofauna from entering litter bags, and the largest mesh, which leads to material leaching out of the bags. Moreover, in our studies, it was important to treat all samples in the same way, to enable making comparisons among them.
2.4 Data analysis
For each litter bag, we determined the proportion of initial litter mass remaining. We analyzed decay constants (k) by fitting the data for each species (the proportion of initial mass remaining was calculated by dividing the mass at each harvest date by the initial mass) with a negative exponential decay model. We used linear regressions of log-transformed proportions of initial mass remaining against time (Berg and McClaugherty 2014; Hobbie 1996; Olson 1963) using the following formula:
where X is the proportion of remaining biomass at time t and k is the decay rate.
Differences in the rates of decomposition among the species studied were assessed using a one-way analysis of variance (ANOVA), followed by Tukey’s test. After that, we used Bonferroni correction, meaning that we tested hypotheses at α = 0.000877. We used the Bonferroni correction to control the family-wise error rate (FWER). The FWER is the probability of rejecting at least one true Hi, that means making at least one type I error. The Bonferroni correction rejects the null hypothesis for each pi = α/m, thereby controlling the FWER at ≤ α. The influence of the studied factors (species, time of exposure in the field, their interaction) on litter decomposition rates was assessed using two-way ANOVA. We checked the normality and homogeneity of the distribution of variables in each group compared by ANOVA. The assumptions of normality and homogeneity were not always valid; however, we decided to assume a normal distribution of data, as due to high sample size we may assume that with increasing sample size distribution of a variable in the whole population tends to a normal distribution, according to the central limit theorem.
Functional trait data were obtained from BiolFlor (Klotz et al. 2002), the LEDA trait database (Kleyer et al. 2008), and the TRY database (Kattge et al. 2011). We focused on morphological and chemical traits of living leaves known to affect components of the carbon and/or nitrogen cycles at the leaf, whole-plant, and ecosystem levels (Cornelissen et al. 1999; Reich et al. 1999): specific leaf area (SLA), leaf nitrogen (N) content per leaf dry mass (LNC), and leaf dry matter content (LDMC). The traits involved in this study were chosen to represent the trade-off between fast acquisition and conservation of resources. Moreover, these traits are correlated with leaf litter traits, traits of other organs, as well as with decomposition rates of leaves and decomposition rates of other organs (Freschet et al. 2012). Four species (A. moschatellina, A. ranunculoides, C. cava, P. quadrifolia) were excluded from the analysis of LNC impact on the decay process because of a lack of data. We evaluated simple linear regression models of species-specific k and the above-mentioned plant traits as independent variables. Moreover, we used principal components analysis (PCA) to assess the correlations between plant traits and decomposition rates. We performed an analysis of variance of a mixed-effects linear model, describing differences in mass loss as a function of functional group, exposition time, temperature, and precipitation. To account for species-dependence of samples representing particular species we treated species as random factors with random slopes (we expected different trajectories of decomposition rates for each species).
All analyses were conducted in JMP Pro 14.0 (SAS Institute Inc. Cary, NC. USA; http://www.sas.com).
3 Results
3.1 Litter decomposition of understory herb species
We found statistically significant effects of herb species (p < 0.0001, df = 14, F = 350.6065), time (p < 0.0001, df = 1, F = 1707.408) and interaction of species × time (p < 0.0001, df = 14, F = 36.503) on litter decomposition. After ca. 2 months of incubation, spring ephemeral species (A. moschatellina and C. cava) reached 95% biomass losses. After the same time of decomposition in the forest, losses of biomass were the highest for the second group of spring ephemerals, i.e. A. moschatellina, P. quadrifolia; on average 95 % of litter decayed. At the same time, ca. 92 % of the first group of spring ephemerals (C. cava, A. ranunculoides, F. verna, and A.nemorosa), 69% of winter-green plants (A. europaeum and G. luteum), 35% of mid-summer senescing plants (M. bifolium, A. podagraria (blooming shoots), A. petiolata) and 30.5% of autumn-senescing plants (A. podagraria (leaves), M. perennis, U. dioica, and S. sylvatica) decomposed. Later, after ca. 6 months of incubation, rates of decomposition of litter for most of the herb species were considerably slower than at the start of the process. Six-month decay constants of spring ephemerals decreased, with the lowest value for A. moschatellina (k = 4.7) and the highest for A. ranunculoides (k = 6.9). Mid-summer senescing plants decomposed the slowest (k range 0.2–3.2). During 182 days (ca. 6 months) of the experiment 95% of the biomass of all spring ephemeral species decomposed (Table 2). Plants that senesced during the mid-summer (M. bifolium, A. petiolata, A. podagraria (blooming shoots)) decomposed the slowest (Figs. 1 and 2). Biomass losses of A. moschatellina and C. cava were the most rapid—96% and 95% of the initial litter mass decayed during 63 days, respectively. After the same time of decomposition, 94% of P. quadrifolia and F. verna, 91% of A. ranunculoides, 90% of A. europaeum, 88% of A. nemorosa, 54% of M. bifolium, 48% of G. luteum, 34% of A. podagraria (leaves), 33% of A. podagraria (blooming shoots), 33% of M. perennis, 30% of U. dioica, 25% of S. sylvatica, and 18% of A. petiolata litter decomposed (Fig. 1). During ca. 6 months of the experiment, 95% of the biomass of C. cava, 94% of A. ranunculoides, 93% of P. quadrifolia, 91% of F. verna, and A. moschatellina decomposed (Table 2). After the same time of decomposition, 89% of the biomass of A. nemorosa, 87% of leaves of A. podagraria, 81% of A. europaeum, and 73% of M. perennis decomposed. Biomass of M. bifolium, U. dioica, S. sylvatica, and A. petiolata were 68, 50, 42, and 41% decomposed, respectively, whereas 28% of blooming shoots of A. podagraria decomposed.
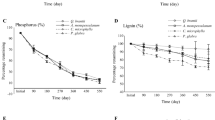
Results of regressions of the decay rate (decay constant after 6 months of decay) of each litter species with functional traits: a SLA vs. decay rates; b LDMC vs. decay rates; c LNC vs. decay rates. The gray area shows 95% confidence intervals for the predicted values. Explanations of abbreviations: AcePse - Acer pseudoplatanus; AdoMos - Adoxa moschatellina; AegPodL - Aegopodium podagraria leaves; AegPodS - Aegopodium podagraria shoots; AllPet - Alliaria petiolata; AneNem - Anemone nemorosa; AneRan - Anemone ranunculoides; AsaEur - Asarum europaeum; CarBet - Carpinus betulus; CorAve - Corylus avellana; CorCav - Corydalis cava; FicVer - Ficaria verna; FraExc - Fraxinus excelsior; GalLut - Galeobdolon luteum; MaiBif - Maianthemum bifolium; MerPer - Mercurialis perennis; ParQua - Paris quadrifolia; QueRob - Quercus robur; StaSyl - Stachys sylvatica; UrtDio - Urtica dioica
Results of principal components analysis. Explanations of abbreviations: AcePse - Acer pseudoplatanus; AdoMos - Adoxa moschatellina; AegPodL - Aegopodium podagraria leaves; AegPodS - Aegopodium podagraria shoots; AllPet - Alliaria petiolata; AneNem - Anemone nemorosa; AneRan - Anemone ranunculoides; AsaEur - Asarum europaeum; CarBet - Carpinus betulus; CorAve - Corylus avellana; CorCav - Corydalis cava; FicVer - Ficaria verna; FraExc - Fraxinus excelsior; GalLut - Galeobdolon luteum; MaiBif - Maianthemum bifolium; MerPer - Mercurialis perennis; ParQua - Paris quadrifolia; QueRob - Quercus robur; StaSyl - Stachys sylvatica; UrtDio - Urtica dioica
3.2 Litter decomposition of the overstory species
We found statistically significant influence of woody plant species (p < 0.0001, df = 4, F = 616.6597), time (p < 0.0001, df = 1, F = 916.3222), and interaction of species × time (p < 0.0001, df = 4, F = 70.108) on litter decomposition. After ca. 6 months in the field decay rates of decomposition of tree foliage litter ranged from 0.2 to 0.8. Q. robur litter had the lowest decomposition rate (12%). At the same time, 21% of C. avellana leaf biomass, 24% of C. betulus, 25% of A. pseudoplatanus, and 43% of F. excelsior decomposed (Fig. 1). After 1 year of exposure in the field, 20% of Q. robur, 30% of C. avellana, 38% of A. pseudoplatanus, 43% of C. betulus, and 86% of F. excelsior decomposed. The foliage of trees had decay rates ranging from 0.2 to 1.9 during the first year of decomposition. F. excelsior leaf biomass decomposed significantly faster than leaves of other tree species (Table 7 in the Appendix).
3.3 Herbs vs. deciduous tree species litter decomposition
In general, litter decomposition for woody plant species was distinctly lower than for herbaceous species. The decay rates calculated for 6 months ranged from 0.2 to 0.8 and 0.2 to 6.9, respectively. F. excelsior leaf biomass did not decompose significantly slower than A. podagraria (both leaves and blooming shoots), A. petiolata, M. perennis, U. dioica, or S. sylvatica litter during ca. 2 months and A. petiolata, A. podagraria (blooming shoots), U. dioica, or S. sylvatica litter during 6 months of exposure in the field (Figs. 1 and 2; Table 7 in the Appendix). The value of mass remaining after ca. 6 months of the study reached by this tree species foliage was more similar to values reached by A. petiolata, S. sylvatica, and U. dioica than to the other woody species studied.
The species studied may be arranged from the fastest to slowest decomposition rate determined after ca. 6 months of the study: A. ranunculoides, C. cava, A. nemorosa, F. verna, P. quadrifolia, A. moschatellina, A. europaeum, A. podagraria leaves, M. perennis, M. bifolium, U. dioica, S. sylvatica, A. petiolata, F. excelsior, C. betulus, A. pseudoplatanus, C. avellana, Q. robur, and A. podagraria blooming shoots (Table 2).
3.4 Predictors of decay
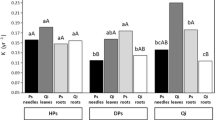
We found significant regressions between morphological (SLA and LDMC) or chemical (LNC) leaf traits and decay rates of the species studied after six months of the experiment (Fig. 3, Table 3). Moreover, LDMC was a stronger predictor of decay rates than SLA and LNC. The regressions between k‐values and traits are visualized in Fig. 3. Variation among traits studied was effectively captured by PCA. The principal ordination axis (PC1) accounted for 65.2% of the total trait variation, and together with the first two principal axes, accounted for 85.8% (Fig. 4). K decay and SLA contributed to the first axis, as well as LDMC, but in opposite directions, whereas LNC was correlated with the second axis. The decomposition process depended on fixed effects (time of exposition in the field, the functional group of plants, and mean total daily precipitation during 6 months of the experiment), as well as on species identity (random intercept) (Table 3). Impact of the other fixed effect (mean temperature during 6 months of the experiment) on the decay process was not statistically significant (Table 3). We found statistically significant differences in the rate of litter decomposition among all functional groups studied (Fig. 5).
4 Discussion
Our hypotheses were only partially supported by the results of the experiment. We found statistically significant differences in litter decomposition among the plant species studied. Our study revealed that: (1) decomposition of herbaceous plants (including stems) was not always completed in less than a year, (2) spring ephemerals had higher decomposition rates than species which dominate the understory during summer and autumn, and (3) decomposition rates of herb species biomass were not always higher than that of tree leaves.
Few published papers on biomass and nutrient cycling of forests have addressed the dynamics of herbaceous material decomposition. Most of them suggest that decay rates (k) for herbaceous plants are higher than one, being in some cases considerably higher (Muller 2014). Exceptions are tundra vegetation dominated by mosses, sedges, or woody understory species (Hobbie 1996), and bulked herbaceous litter, dominated by ferns and woody understory species in a mixed hardwood stand in Canada (MacLean and Wein 1978). This is not in accordance with our study. In most cases, decomposition of herb litter biomass was completed much faster than within 1 year. During our experiment (ca. 6 months) decomposition rates of six species included in the spring ephemeral group reached k values in the range of 4.7–6.9, while mid-summer senescing summer-green plants (three species) ranged from 0.2–1.4, autumn-senescing summer-green plants (four species) ranged from 0.9 to 3.2, and winter-green A. europaeum reached 4.1. According to Muller (2014), the decomposition rates of herbaceous species during 12 months of field exposure reached values in the range of 0.61–3.31. This was likely related to the species taken into account in Muller’s (2014) review, as only two species are in common between that study and our study, namely, A. nemorosa and M. perennis. Data about these species were obtained from Wise and Shaefer (1994), who found statistically significant differences between decomposition rates of these species connected with three litter bag mesh sizes used in their experiment. In the treatment similar to ours (1 mm mesh size), they found 12-month decomposition constants k of 13.99 and 10.58 for A. nemorosa and M. perennis, respectively. Our study had 6-month decomposition constants of 6.2 and 2.1 for A. nemorosa and M. perennis, respectively. However, the results cited were obtained in beech forests on mull soils, where mean annual temperatures during the period of the study, were 6, 6.7, and 6 °C, and mean annual precipitation was 706, 726, and 717 mm. We conducted our studies in drier and warmer conditions, which may have significantly influenced k constants since mean temperature and precipitation are key factors determining decomposition rate (Aerts 1997; Hobbie 1996; Trofymow et al. 2002). In contrast, Halabuk and Gerhátová (2011) in SW Slovakia found results similar to our study, in similar environmental and climatic conditions (an ecotone of the hornbeam-oak forest situated in a region with a mean annual temperature of 9.3 °C and total annual precipitation of 580 mm). They found decomposition rates of k = 2.41 for M. perennis and k = 2.44 for A. petiolata during a 324-day experiment. In an experiment conducted in North America by Rodgers et al. (2008), green rosettes of A. petiolata decomposed in about 6 months, and senesced litter of A. petiolata decomposed in about one year. These results are very close to our 0.9 decomposition rate for A. petiolata, although those studies were conducted at five different sites including deciduous, coniferous, and mixed forests. In our study, decomposition constants of A. petiolata and blooming shoots of A. podagraria were lower than one. These species (especially A. podagraria blooming shoots) decomposed as slowly as tree leaves during our experiment. These two plant species had the highest contributions of stems to aboveground biomass among the species studied (Paź-Dyderska et al. 2020). For these two species, the decomposition rate visibly decreased during decomposition. In our opinion, this is connected with the differences in organ-specific decomposition rate k. Every sample is a mix of leaves and shoots in proportions similar to what occurs in specimens in the field. After the first stage of decomposition, when leaves have decomposed, the process slows down, and rates of mass losses between dates of sample collection are stable or even decreasing. Biomass allocation to organs that differ in the way that they decompose (e.g., stems and leaves) affects decomposition itself (Hobbie 1996). However, this has not been confirmed by studies of Bumb et al. (2018), which showed the same leaf and shoot decomposability of sixteen Mediterranean species. More in-depth studies are needed to understand how the decomposability of different plant organs is correlated and if these processes are controlled by the same functional traits (Freschet et al. 2012; Hobbie 2015). The duration of our experiment was only half of a year for most of the species. In the case of spring ephemerals, this was enough time to complete decomposition, but in other cases, we considered only part of the process that included decay of the labile part of the plant material.
Cornwell et al. (2008) found that decomposition and nutrient cycling processes across biomes were affected by the functional group of plant species. This has been described for a wide range of temperate plant species (Cornelissen and Thompson 1997), in Alaskan tundra (Hobbie 1996), lowland tropical forest (Santiago 2010), Mediterranean forests (De la Riva et al. 2019), rainforests (Jackson et al. 2013), experimental grasslands (Scherer-Lorenzen 2008), old-fields (Kazakou et al. 2006), alpine meadow (Jiang et al. 2013), alpine snow beds (Carbognani et al. 2014), and post-mining sites (Rawlik et al. 2019). There is little information available about tradeoffs between plant traits (Kleyer et al. 2018), and between plant functional traits and decomposition, in forest herb species. Plant functional groups were also connected with similar SLA, LDMC, and LNC (Cornwell et al. 2008). It was previously reported that these traits have an “after-life effect” on decomposition (Freschet et al. 2010, 2012; Jackrel and Wootton 2015; Santiago 2010). Freschet et al. (2012) reported that structural (lignin, DMC) and chemical (N) traits together were better predictors for decomposition rates of several high-turnover organs (leaves, fine stems, and reproductive parts) than structural traits alone, whereas leaf nitrogen content influenced leaf decomposition, but this relationship was not apparent in any other organs. Results of our studies focused on the decomposition of aboveground biomass of herbaceous plants, confirmed patterns reported by Freschet (2012). The best predictor for decomposition rates in our studies was LDMC. The results obtained were similar to those reported in previous studies, although we decided to use values obtained from global databases of functional traits. This method has limitations, as it is known that measurements of traits and decay rates on the same species, conducted at different sites, can potentially add noise to the analyses (Zanne et al. 2015).
In temperate deciduous forests, light availability is a major factor that determines leaf structure, with spring ephemeral species exhibiting nutrient-rich leaves with higher SLA relative to other herb species groups or late-successional canopy tree species (Jagodziński et al. 2016; Muller 2014; Rothstein and Zak 2001). Therefore, leaf traits associated with canopy openness determine litter decomposition rates. This is in accordance with our results showing decreasing decay rates in this order: spring ephemerals (high N concentration, high SLA), mid-summer, and autumn-senescing summer-green herbaceous species. Our study further confirmed that fast decomposition rates were related to fast growth and can be generally predicted from functional plant group. Higher decomposition rates for spring ephemerals are supposedly connected with their traits as well as with ecological function in forest ecosystems. Results of previously published papers suggest that the spring ephemeral group of plants act as a “vernal dam” for nutrients. According to this theory spring ephemerals, by nutrient uptake and storage before canopy leaf-out, prevent nutrient losses by spring flow of soil water (Muller and Bormann 1976; Muller 2014; Rothstein and Zak 2001). Fast decomposition of the litter of this phenological group of herbaceous species may be the main part of their function in nutrient cycling, because it allows a huge pool of nutrients to be returned to the soil, making them available for trees at the time of their most intensive physiological activity. The remaining herb species with lower decomposition rates can be very important for nutrient storage in plant biomass. However, a broader study is needed to understand the function of herbaceous species in nutrient cycling in deciduous forest systems.
Most recent research results suggest that decomposition of tree leaf litter requires longer than one year for completion. Moreover, for leaves of woody species, decomposition rates have most often been reported in the range from 0.3 to 0.8 (see Muller 2014 and literature cited therein). This is generally in accordance with our studies showing that ca. 6-month decomposition rates of four oak-hornbeam tree species were in the range cited above (F. excelsior k = 0.8, C. betulus k = 0.5, A. pseudoplatanus k = 0.4, C. avellana k = 0.3, and Q. robur k = 0.2).
Our results are similar to previous estimates of tree foliage decomposition in areas of similar climatic and environmental conditions, although the variability of values is high because decay processes are sensitive to even small microclimatic and climatic fluctuations. In the experiment provided by Dziadowiec (1987) in an oak-linden-hornbeam forest in the Białowieża Forest, 65% of the initial biomass of A. pseudoplatanus and 36.5% of Q. robur leaves decomposed after 1 year in the field. This author also reported similar results from an oak-hornbeam forest near Toruń in Poland (Dziadowiec 1990). After 1 year of the experiment, 46% of the initial biomass of C. betulus leaf litter and 36% of Q. robur leaves decomposed. For those species, we found 43.3% and 20.2% of biomass decomposed, respectively, at the same time. Faster decomposition in Białowieża can be connected with bigger litter bag mesh size (2 mm) and more humid climatic conditions (Dziadowiec 1987). Hobbie et al. (2006) reported decomposition results for Q. robur leaves in central Poland similar to our results (k range after 1 year of exposure in the field of 0.21–0.24 vs. 0.23). However, in the case of the experiment cited above, decomposition constants were lower than in our experiment for A. pseudoplatanus (0.29 vs. 0.47) and C. betulus (0.30 vs. 0.54). Although several studies described tree leaf litter decomposition rates much higher than for herbaceous litter, research focused on oak-hornbeam forest tree species from other parts of those species geographical distributions are still limited (Jacob et al. 2009; Slade and Riutta 2012).
One of the key certainties in decomposition in forest ecosystems is the faster decomposition of herb litter than tree leaf biomass (Halabuk and Gerhátová 2011; Mayer 2008; Muller 2014), as most reported tree foliage has decomposition rates of k < 1 and most herbaceous litter of k > 1. We did not find significant differences between the decomposition of tree leaves and summer-green herb species after six months of the study. This is particularly important because it shows that not all herb species litter is part of the labile litter fraction. Some parts of those plant materials should be categorized as more resistant to decomposition—this seems to be a fact for species with a higher allocation of biomass to stems or with more lignified stems. In contrast, our study indicated that F. excelsior leaf litter can be included in the labile pool of litter which decomposes and returns nutrients to available nutrient pools quickly. Unfortunately, our studies focused on the first stages of the decomposition process, and differences between decomposition of herb and tree species foliage may be more visible in the later stages of decomposition.
Previous studies recognized that climatic factors such as mean annual temperature, mean annual precipitation and annual actual evapotranspiration regulate litter decomposition rate (Berg and McClaugherty 2014; Zhang et al. 2008). Generally, higher temperatures and precipitation stimulate mass loss during decomposition (Hobbie 1996; Mueller et al. 2016; Trofymow et al. 2002). In this context, reported differences in decomposition rates of the species we studied can be partly explained by changes in microclimatic conditions, and by their life history traits. At the same time, climate conditions have impacts on both litter quality (Coûteaux et al. 1995) and plant traits. Moreover, it is difficult to separate the influence of climate from the influence of litter quality on decomposition processes (Berg and McClaugherty 2014). As summarized by Aerts (1997), climate, litter chemistry, and litter decomposition are connected by a triangular relationship. As we mentioned above (introduction), we found differences between average air temperatures, ground temperatures, and total precipitation during the field incubation of litter for different groups of plants, e.g., during the 6-month experiment for the 1st group of plants mean ground temperature was more than 10 °C higher than during the 6-month experiment for tree leaves. Total precipitation also differed for the periods of field experiments for different groups of plants. Our results indicated that microclimatic conditions stimulated decomposition when spring ephemeral litter decays in forest ecosystems and precipitation had a statistically significant impact on the decay process. Our study seemed to suffer because different times of incubation, which made it impossible to disentangle the influences of different climatic conditions and litter quality on the decomposition process. However, our studies aimed to find real, biological rates of decomposition of the species studied. Generally, we conducted our study with weather conditions similar to average conditions of the last decade (mean annual temperature: 9.1 °C in 2011–2013 vs. 9.2 in 2001–2010 and mean annual precipitation: 573 mm vs. 535 in the same periods of time, respectively). Moreover, we included microclimatic conditions as fixed effects into linear mixed models, to assess the impact of technical limitations of our studies on the results obtained.
In our studies, we make an effort to find real biological rates of decomposition of the species studied. However, we are aware of the limitations of research methods used in decomposition research. Kurz-Besson et al. (2005) compared the results of litter bag and direct observation methods and found higher mass losses measured by litter bags than by direct observation. These results were in agreement with previous results (De Santo et al. 1993) and were probably connected with a higher and more stable moisture content of litter in litter bags than in surrounding litter. In light of these findings, our results could overestimate decomposition rates; however, the studies mentioned took place with different material (Scots pine needles) and habitat. On the other hand, taking into account the results obtained by Bradford et al. (2002), our results could underestimate decomposition rates. According to these results, using litter bags of 1 mm mesh size decreased decomposition by 20% at 35 days because of macrofauna exclusion. Despite these limitations, the litter bag method is highly repeatable, relatively inexpensive, and widely used in decomposition studies (Harmon et al. 1999).
5 Conclusions
-
1.
The decomposition process was dependent on the functional group of plants, time of exposure in the field, species identity, and precipitation.
-
2.
Spring ephemerals had higher decomposition rates than species that dominate the understory of oak-hornbeam forest during summer and autumn.
-
3.
The decomposition of herbaceous plants was not always completed in less than a year—not all litter of herbaceous species is part of the labile litter fraction.
-
4.
Functional traits of leaves of herbaceous plants were correlated with their decomposition. The best predictor for decomposition rates in our studies was leaf dry matter content (LDMC).
Data availability
The datasets generated and analyzed during the current study are available in the FigShare repository, https://doi.org/10.6084/m9.figshare.12987410.v3.
Change history
02 June 2021
ORCID information of the first author has been updated.
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449. https://doi.org/10.2307/3546886
Bakker MA, Carreño-Rocabado G, Poorter L (2011) Leaf economics traits predict litter decomposition of tropical plants and differ among land use types: leaf economics traits and decomposition. Funct Ecol 25:473–483. https://doi.org/10.1111/j.1365-2435.2010.01802.x
Bantle A, Borken W, Ellerbrock RH, Schulze ED, Weisser WW, Matzner E (2014) Quantity and quality of dissolved organic carbon released from coarse woody debris of different tree species in the early phase of decomposition. For Ecol Manage 329:287–294. https://doi.org/10.1016/j.foreco.2014.06.035
Baraloto C, Goldberg DE, Bonal D (2005) Performance trade-offs among tropical tree seedlings in contrasting microhabitats. Ecology 86:2461–2472. https://doi.org/10.1890/04-1956
Berg B, McClaugherty C (2014) Climatic environment. In: McClaugherty C (ed) Berg B. Plant Litter, Springer, pp 143–170
Bumb I, Garnier E, Coq S, Nahmani J, Del Rey GM, Gimenez O, Kazakou E (2018) Traits determining the digestibility–decomposability relationships in species from Mediterranean rangelands. Ann Bot 121:459–469. https://doi.org/10.1093/aob/mcx175
Bormann FH, Likens GE, Fisher DW, Pierce RS (1968) Nutrient loss accelerated by clear-cutting of a forest ecosystem. Science 159:882–884. https://doi.org/10.1126/science.159.3817.882
Bradford MA, Tordoff GM, Eggers T et al (2002) Microbiota, fauna, and mesh size interactions in litter decomposition. Oikos 99:317–323. https://doi.org/10.1034/j.1600-0706.2002.990212.x
Carbognani M, Tomaselli M, Petraglia A (2014) Current vegetation changes in an alpine late snowbed community in the south-eastern Alps (N-Italy). Alpine botany 124:105–113
Central Statistical Office (2020) Concise Statistical Yearbook of the Republic of Poland. Statistical Publishing Establishment, Warszawa. https://stat.gov.pl/obszary-tematyczne/roczniki-statystyczne/roczniki-statystyczne/maly-rocznik-statystyczny-polski-2020,1,22.html
Chapin FS (2003) Effects of plant traits on ecosystem and regional processes: a conceptual framework for predicting the consequences of global change. Ann Bot 91:455–463. https://doi.org/10.1093/aob/mcg041
Cornelissen JHC, Thompson K (1997) Functional leaf attributes predict litter decomposition rate in herbaceous plants. New Phytol 135:109–114. https://doi.org/10.1046/j.1469-8137.1997.00628.x10.1046/j.1469-8137.1997.00628.x
Cornelissen JHC, Perez-Harguindeguy N, Dı́az S, et al (1999) Leaf structure and defence control litter decomposition rate across species and life forms in regional floras on two continents. New Phytol 143:191–200. https://doi.org/10.1046/j.1469-8137.1999.00430.x
Cornwell WK, Cornelissen JHC, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O et al (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071. https://doi.org/10.1111/j.1461-0248.2008.01219.x
Coûteaux MM, Bottner P, Berg B (1995) Litter decomposition, climate and litter quality. Trees 10:63–66. https://doi.org/10.1016/S0169-5347(00)88978-8
De la Riva EG, Prieto I, Villar R (2019) The leaf economic spectrum drives leaf litter decomposition in Mediterranean forests. Plant Soil 435:353–366. https://doi.org/10.1007/s11104-018-3883-3
De Santo AV, Berg B, Rutigliano FA, Alfani A, Floretto A (1993) Factors regulating early-stage decomposition of needle litters in five different coniferous forests. Soil Biol Biochem 25:1423–1433. https://doi.org/10.1016/0038-0717(93)90057-I
Dı́az S, Cabido M, (2001) Vive la difference: plant functional diversity matters to ecosystem processes. Trends Ecol Evol 16:646–655. https://doi.org/10.1016/S0169-5347(01)02283-2
Díaz S, Kattge J, Cornelissen JHC, Wright IJ, Lavorel S, Dray S et al (2016) The global spectrum of plant form and function. Nature 529:167–171. https://doi.org/10.1038/nature16489
Dziadowiec H (1987) The decomposition of plant litterfall in an oak-linden-hornbeam forest and oak-pine mixed forest of the Bialowieza National Park. Acta Soc Bot Pol 56:169–185
Dziadowiec H (1990) Rozkład ściółek w wybranych ekosystemach leśnych (mineralizacja, uwalnianie składników pokarmowych, humifikacja). Rozprawy Uniwersytetu Mikołaja Kopernika, Toruń
Dzięciołowski R (1970) Biomass of herb layer and understory plants as potential food resources for herbivorous mammals. Folia Forestalia Polonica A 16:69–90
Ellenberg H (1988) Vegetation Ecology of Central Europe. Cambridge University Press, Cambridge
Freschet GT, Aerts R, Cornelissen JHC (2012) A plant economics spectrum of litter decomposability. Funct Ecol 26:56–65. https://doi.org/10.1111/j.1365-2435.2011.01913.x
Freschet GT, Cornelissen JHC, Logtestijn RSPV, Aerts R (2010) Evidence of the ‘plant economics spectrum’ in a subarctic flora. J Ecol 98:362–373. https://doi.org/10.1111/j.1365-2745.2009.01615.x
Gilliam FS (2007) The ecological significance of the herbaceous layer in temperate forest ecosystems. Bioscience 57:845–858. https://doi.org/10.1641/B571007
Graves JD (1990) A model of the seasonal pattern of carbon acquisition in two woodland herbs, Mercurialis perennis L. and Geum urbanum L. Oecologia 83:479–484
Grime JP (2001) Plant strategies, vegetation processes and ecosystem properties, 2nd edn. John Wiley & Sons Ltd, Chichester
Halabuk A, Gerhátová K (2011) Comparative study of leaf litter decomposition of exotic and native species in an ecotop of the hornbeam-oak forest near Báb village, SW Slovakia. Folia Oecologica 38:17–27
Handa IT, Aerts R, Berendse F, Berg MP, Bruder A, Butenschoen O et al (2014) Consequences of biodiversity loss for litter decomposition across biomes. Nature 509:218–221. https://doi.org/10.1038/nature13247
Harmon ME, Nadelhoffer KJ, Blair JM (1999) Measuring Decomposition. In: Robertson GP, Coleman DC, Bledsoe CS, Sollins P (eds) Standard Soil Methods for Long-Term Ecological Research. Oxford University Press, pp:202–240
Harmon ME, Krankina ON, Sexton J (2000) Decomposition vectors: a new approach to estimating woody detritus decomposition dynamics. Can J For Res 30:76–84. https://doi.org/10.1139/x99-187
Hobbie SE (1992) Effects of plant species on nutrient cycling. Trends Ecol Evol 7:336–339. https://doi.org/10.1016/0169-5347(92)90126-V
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–522. https://doi.org/10.2307/2963492
Hobbie SE, Reich PB, Oleksyn J, Ogdahl M, Zytkowiak R et al (2006) Tree species effects on decomposition and forest floor dynamics in a common garden. Ecology 87:2288–2297. https://doi.org/10.1890/0012-9658(2006)87[2288:TSEODA]2.0.CO;2
Hobbie SE (2015) Plant species effects on nutrient cycling: revisiting litter feedbacks. Trends Ecol Evol 30:357–363. https://doi.org/10.1016/j.tree.2015.03.015
Horodecki P, Wiczyńska K, Jagodziński AM (2014) Natural regeneration in the ‘Czmoń’ nature reserve (Wielkopolska Region). Forest Research Papers 75:61–75. https://doi.org/10.2478/frp-2014-0007
Horodecki P, Jagodziński AM (2017) Tree species effects on litter decomposition in pure stands on afforested post-mining sites. For Ecol Manage 406:1–11. https://doi.org/10.1016/j.foreco.2017.09.059
Horodecki P, Jagodziński AM (2019) Site type effect on litter decomposition rates: a three-year comparison of decomposition process between spoil heap and forest sites. Forests 10:353. https://doi.org/10.3390/f10040353
Horodecki P, Nowiński M, Jagodziński AM (2019) Advantages of mixed tree stands in restoration of upper soil layers on postmining sites: a five-year leaf litter decomposition experiment. Land Degrad Dev 30:3–13. https://doi.org/10.1002/ldr.3194
Jackrel SL, Wootton JT (2015) Cascading effects of induced terrestrial plant defences on aquatic and terrestrial ecosystem function. Proceedings of the Royal Society B: Biological Sciences 282:20142522–20142522. https://doi.org/10.1098/rspb.2014.2522
Jackson BG, Peltzer DA, Wardle DA (2013) Are functional traits and litter decomposability coordinated across leaves, twigs and wood? A test using temperate rainforest tree species. Oikos 122:1131–1142. https://doi.org/10.1111/j.1600-0706.2012.00056.x
Jacob M, Weland N, Platner C, Schaefer M, Leuschner C, Thomas FM (2009) Nutrient release from decomposing leaf litter of temperate deciduous forest trees along a gradient of increasing tree species diversity. Soil Biol Biochem 41:2122–2130. https://doi.org/10.1016/j.soilbio.2009.07.024
Jagodziński AM, Dyderski MK, Rawlik K, Kątna B (2016) Seasonal variability of biomass, total leaf area and specific leaf area of forest understory herbs reflects their life strategies. For Ecol Manage 374:71–81. https://doi.org/10.1016/j.foreco.2016.04.050
Jiang J, Li Y, Wang M et al (2013) Litter species traits, but not richness, contribute to carbon and nitrogen dynamics in an alpine meadow on the Tibetan Plateau. Plant Soil 373:931–941. https://doi.org/10.1007/s11104-013-1859-x
Jurkšienė G, Janušauskaitė D, Armolaitis K, Baliuckas V (2017) Leaf litterfall decomposition of pedunculate (Quercus robur L.) and sessile (Q. petraea [Matt.] Liebl.) oaks and their hybrids and its impact on soil microbiota. Dendrobiology 78:51–62. https://doi.org/10.12657/denbio.078.006
Kamczyc J, Dyderski MK, Horodecki P, Jagodziński AM (2019) Mite communities (Acari, Mesostigmata) in the initially decomposed ‘litter islands’ of 11 tree species in Scots pine (Pinus sylvestris L.) forest. Forests 10:#403. https://doi.org/10.3390/f10050403
Kattge J, Díaz S, Lavorel S, Prentice IC, Leadley P, Bonisch G et al (2011) TRY – a global database of plant traits. Glob Change Biol 17:2905–2935. https://doi.org/10.1111/j.1365-2486.2011.02451.x
Kazakou E, Vile D, Shipley B, Gallet C, Garnier E (2006) Co-variations in litter decomposition, leaf traits and plant growth in species from a Mediterranean old-field succession. Funct Ecol 20:21–30. https://doi.org/10.1111/j.1469-8137.2005.01349.x
Kleyer M, Bekker RM, Knevel IC, Bakker JP, Thompson K, Sonnenschein M et al (2008) The LEDA Traitbase: a database of life-history traits of the Northwest European flora. J Ecol 96:1266–1274. https://doi.org/10.1111/j.1365-2745.2008.01430.x
Kleyer M, Minden V (2015) Why functional ecology should consider all plant organs: an allocation-based perspective. Basic Appl Ecol 16:1–9. https://doi.org/10.1016/j.baae.2014.11.002
Kleyer M, Trinogga J, Cebrián-Piqueras MA et al (2018) Trait correlation network analysis identifies biomass allocation traits and stem specific length as hub traits in herbaceous perennial plants. J Ecol 2018:1–14. https://doi.org/10.1111/1365-2745.13066
Klotz S, Kühn I, Durka W (2002) BIOLFLOR–eine Datenbank zu biologisch-ökologischen Merkmalen der Gefäßpflanzen in Deutschland. Bundesamt für Naturschutz, Bonn. Schriftenreihe für Vegetationskunde 38
Kurz-Besson C, Coûteaux M-M, Thiéry JM, Berg B, Remacle J (2005) A comparison of litterbag and direct observation methods of Scots pine needle decomposition measurement. Soil Biol Biochem 37:2315–2318. https://doi.org/10.1016/j.soilbio.2005.03.022
Liang J, Crowther TW, Picard N, Wiser S, Zhou M, Alberti G, et al (2016) Positive biodiversity-productivity relationship predominant in global forests. Science 354:aaf8957. https://doi.org/10.1126/science.aaf8957
Ma W, Shi P, Li W et al (2010) Changes in individual plant traits and biomass allocation in alpine meadow with elevation variation on the Qinghai-Tibetan Plateau. Science China Life Sciences 53:1142–1151. https://doi.org/10.1007/s11427-010-4054-9
MacLean DA, Wein RW (1978) Weight loss and nutrient changes in decomposing litter and forest floor material in New Brunswick forest stands. Can J Bot 56:2730–2749. https://doi.org/10.1139/b78-326
Makkonen M, Berg MP, Handa IT, Hättenschwiler S, van Ruijven J, van Bodegom PM, Aerts R (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041. https://doi.org/10.1111/j.1461-0248.2012.01826.x
Mayer PM (2008) Ecosystem and decomposer effects on litter dynamics along an old field to old-growth forest successional gradient. Acta Oecologica 33:222–230. https://doi.org/10.1016/j.actao.2007.11.001
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626
Mueller KE, Eisenhauer N, Reich PB, Hobbie SE, Chadwick OA, Chorover J et al (2016) Light, earthworms, and soil resources as predictors of diversity of 10 soil invertebrate groups across monocultures of 14 tree species. Soil Biol Biochem 92:184–198. https://doi.org/10.1016/j.soilbio.2015.10.010
Muller RN, Bormann FH (1976) Role of Erythronium americanum Ker. in energy flow and nutrient dynamics of a northern hardwood forest ecosystem. Science 193:1126. https://doi.org/10.1126/science.193.4258.1126
Muller RN (2014) Nutrient relations of the herbaceous layer in deciduous forest ecosystems. In: Gilliam F (ed) The Herbaceous Layer in Forests of Eastern North America, 2nd edn. Oxford University Press, pp:12–34
Noda HM, Muraoka H, Nasahara KN, Saigusa N, Murayama S, Koizumi H (2015) Phenology of leaf morphological, photosynthetic, and nitrogen use characteristics of canopy trees in a cool-temperate deciduous broadleaf forest at Takayama, central Japan. Ecol Res 30:247–266. https://doi.org/10.1007/s11284-014-1222-6
Neufeld HS, Young DR (2014) Ecophysiology of the herbaceous layer in temperate deciduous forests. In: Gilliam F (ed) The Herbaceous Layer in Forests of Eastern North America, 2nd ed. Oxford University Press, pp:35–95
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331. https://doi.org/10.2307/1932179
Paź-Dyderska S, Dyderski MK, Szwaczka P, Brzezicha M, Bigos K, Jagodziński AM (2020) Leaf traits and aboveground biomass variability of forest understory herbaceous plant species. Ecosystems 23:555–569. https://doi.org/10.1007/s10021-019-00421-6
Poorter H, De Jong R (1999) A comparison of specific leaf area, chemical composition and leaf construction costs of field plants from 15 habitats differing in productivity. The New Phytologist 143:163–176. https://doi.org/10.1046/j.1469-8137.1999.00428.x
Rawlik K, Horodecki P, Jagodziński AM (2015) Odnowienie naturalne drzew i krzewów w płatach dobrze zachowanego oraz spinetyzowanego grądu środkowoeuropejskiego Galio sylvatici-Carpinetum (R. Tx. 1937) Oberd. 1957. Studia i Materiały CEPL w Rogowie 17:112–124
Rawlik K, Nowiński M, Jagodziński AM (2020) Decomposition rates of woody plants leaf- and herb-litter. Figshare repository. V3 [dataset] https://doi.org/10.6084/m9.figshare.12987410.v3
Rawlik M, Jagodziński AM (2020) Seasonal dynamics of shoot biomass of dominant clonal herb species in an oak–hornbeam forest herb layer. Plant Ecol 221:1133-1142. https://doi.org/10.1007/s11258-020-01067-4
Rawlik M, Kasprowicz M, Jagodziński AM, Kaźmierowski C, Łukowiak R, Grzebisz W (2018) Canopy tree species determine herb layer biomass and species composition on a reclaimed mine spoil heap. Sci Total Environ 635:1205–1214. https://doi.org/10.1016/j.scitotenv.2018.04.133
Rawlik M, Kasprowicz M, Jagodziński AM, Rawlik K, Kaźmierowski C (2019) Slope exposure and forest stand type as crucial factors determining the decomposition rate of herbaceous litter on a reclaimed spoil heap. Catena 175:219–227. https://doi.org/10.1016/j.catena.2018.12.008
Reich PB, Ellsworth DS, Walters MB et al (1999) Generality of Leaf Trait Relationships: A Test Across Six Biomes. Ecology 80:1955–1969. https://doi.org/10.1890/0012-9658(1999)080[1955:GOLTRA]2.0.CO;2
Rodgers VL, Wolfe BE, Werden LK, Finzi AC (2008) The invasive species Alliaria petiolata (garlic mustard) increases soil nutrient availability in northern hardwood-conifer forests. Oecologia 157:459–471. https://doi.org/10.1007/s00442-008-1089-8
Rothstein DE, Zak DR (2001) Photosynthetic adaptation and acclimation to exploit seasonal periods of direct irradiance in three temperate, deciduous-forest herbs. Funct Ecol 15:722–731. https://doi.org/10.1046/j.0269-8463.2001.00584.x
Santiago LS (2010) Can growth form classification predict litter nutrient dynamics and decomposition rates in lowland wet forest?: Decomposition in wet tropical forest. Biotropica 42:72–79. https://doi.org/10.1111/j.1744-7429.2009.00604.x
Scherer-Lorenzen M (2008) Functional diversity affects decomposition processes in experimental grasslands. Funct Ecol 22:547–555. https://doi.org/10.1111/j.1365-2435.2008.01389.x
Slade EM, Riutta T (2012) Interacting effects of leaf litter species and macrofauna on decomposition in different litter environments. Basic Appl Ecol 13:423–431. https://doi.org/10.1016/j.baae.2012.06.008
Spyreas G, Matthews JW (2006) Floristic conservation value, nested understory floras, and the development of second-growth forest. Ecol Appl 16:1351–1366. https://doi.org/10.1890/1051-0761(2006)016[1351:FCVNUF]2.0.CO;2
Trofymow JA, Moore TR, Titus B, Prescott C, Morrison I, Siltanen M et al (2002) Rates of litter decomposition over 6 years in Canadian forests: influence of litter quality and climate. Can J For Res 32:789–804. https://doi.org/10.1139/x01-117
Uemura S (1994) Patterns of leaf phenology in forest understory. Can J For Res 72:409–414. https://doi.org/10.1139/b94-055
Urbanowski CK, Horodecki P, Kamczyc J, Skorupski M, Jagodziński AM (2018) Succession of mite assemblages (Acari, Mesostigmata) during decomposition of tree leaves in forest stands growing on reclaimed post-mining spoil heap and adjacent forest habitats. Forests 9:718. https://doi.org/10.3390/f9110718
Wang L, Niu K, Yang Y, Zhou P (2010) Patterns of above- and belowground biomass allocation in China’s grasslands: Evidence from individual-level observations. Science China Life Sciences 53:851–857. https://doi.org/10.1007/s11427-010-4027-z
Wiczyńska K, Horodecki P, Jagodziński AM (2013) Stand structure and species composition in the ‘Czmoń’ nature reserve. Science, Nature, Technologies/Nauka, Przyroda, Technologie 7:69
Wise DH, Schaefer M (1994) Decomposition of leaf litter in a mull beech forest: comparison between canopy and herbaceous species. Pedobiologia 38:269–288
Wright IJ, Reich PB, Cornelissen JHC, Falster DS, Garnier E, Hikosaka K et al (2005) Assessing the generality of global leaf trait relationships. New Phytol 166:485–496. https://doi.org/10.1111/j.1469-8137.2005.01349.x
Zanne AE, Oberle B, Dunham KM et al (2015) A deteriorating state of affairs: How endogenous and exogenous factors determine plant decay rates. J Ecol 103:1421–1431. https://doi.org/10.1111/1365-2745.12474
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. Journal of Plant Ecology 1:85–93. https://doi.org/10.1093/jpe/rtn002
Zukswert JM, Prescott CE (2017) Relationships among leaf functional traits, litter traits, and mass loss during early phases of leaf litter decomposition in 12 woody plant species. Oecologia 185:305–316. https://doi.org/10.1007/s00442-017-3951-z
Acknowledgments
We kindly thank Dr. Lee E. Frelich (The University of Minnesota Center for Forest Ecology, USA) for valuable comments on the early draft of the manuscript and linguistic support. We also thank Dr. Marcin K. Dyderski and Dr. Paweł Horodecki (Institute of Dendrology, Polish Academy of Sciences, Kórnik, Poland) for valuable comments on the early draft of the manuscript.
Funding
The study was supported by the General Directorate of State Forests, Warsaw, Poland (research project ‘Environmental and genetic factors affecting productivity of forest ecosystems on forest and post-industrial habitats’) and by the Institute of Dendrology, Polish Academy of Sciences, Kórnik, Poland (research project for young scientists, 2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Andreas Bolte
Contribution of the co-authors Katarzyna Rawlik: designed the research, formulated research problem and developed the methodology; collected the data; analyzed the data and wrote the first draft of the manuscript; contributed critically to the drafts and gave final approval for publication. Mirosław Nowiński: collected the data; analyzed the data; contributed critically to the drafts and gave final approval for publication. Andrzej M. Jagodziński: designed the research, formulated a research problem and developed the methodology; analyzed the data and wrote the first draft of the manuscript; contributed critically to the drafts and gave final approval for publication
Appendix
Appendix
Monthly precipitation during the course of the research. Data was from the meteorological station in Kórnik including the year prior to start of the research and the 2 years during the research project: 1- beginning of the experiment for the first group of plants; 2- beginning of the experiment for the second group of plants; 3- beginning of the experiment for the third group of plants; 4- beginning of the experiment for the fourth group of plants; 5- beginning of the experiment for the fifth group of plants
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rawlik, K., Nowiński, M. & Jagodziński, A.M. Short life–fast death: decomposition rates of woody plants leaf- and herb-litter. Annals of Forest Science 78, 6 (2021). https://doi.org/10.1007/s13595-020-01019-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13595-020-01019-y