Abstract
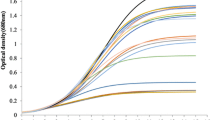
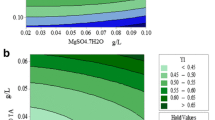
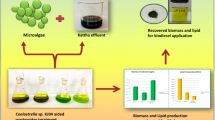
This experimental study was framed with an objective to optimize the nutrients ((dairy industry wastewater (DIWW), nitrate (NO3−), and phosphate (PO4−3)) using response surface methodology (RSM) with Chlorella to investigate biomass and lipid with FAME content. Quality of obtained bio-oil was also indexed on scale of fuel quality parameters. Three variables were tested with 20 interactions between these variables, and their affects were statistically studied using central composite design. Biomass and lipid concentration with FAME content varied between 0.59 to 1.54 g L−1, 19.81 to 34.54%, and 69.32 to 82.78%, respectively, for different combinations of operating variables. Interactive effects of combined nutrients were also studied in combination, and the best biomass and lipid productivity was noticed with NO3− + PO4−3 in comparison to others, whereas the best FAME content (85.23%) was observed with DIWW + NO3. FTIR analysis showed variation in the spectra with different selected combinations with 1.7-fold increase in the lipid content in respect to control. Qualitative assessment of parameters ranges in between 0.74 and 1.01 mg KOH/g for acid value (AV), 152.34–187.24 mg KOH/g for saponification value (SV), 135.39–143.95-mg I2/100 g oil for iodine value (IN), whereas cetane value (CN) was found in between 43.06 and 50.31 and higher heating values (HHVs)) almost 45 MJ kg−1. Crude bio-oil’s quality was compared with the fuel quality index (FQI), where S4 quality sample (59.12) was found significantly compatible with the commercial biodiesel.








Similar content being viewed by others
References
Khanra A, Vasistha S, Rai MP (2017) Glycerol on lipid enhancement and fame characterization in algae for raw material of biodiesel. International Renewable Energy Res (IJRER) 7(4):1970–1978. https://doi.org/10.1016/j.rser.2014.08.031
Sergeeva YE, Mostova EB, Gorin KV, Komova AV, Konova IA, Pojidaev VM, Sineoky SP (2017) Calculation of biodiesel fuel characteristics based on the fatty acid composition of the lipids of some biotechnologically important microorganisms. Appl Biochem Microbiol 53(8):807–813. https://doi.org/10.1134/s0003683817080063
Chandra R, Rohit MV, Swamy YV, Mohan SV (2014) Regulatory function of organic carbon supplementation on biodiesel production during growth and nutrient stress phases of mixotrophic microalgae cultivation. Bioresour Technol 165:279–287. https://doi.org/10.1016/jbiortech201402102
DuongV T, Ahmed F, Thomas-Hall SR, Quigley S, Nowak E, Schenk PM (2015) High protein-and high lipid-producing microalgae from northern Australia as potential feedstock for animal feed and biodiesel. Front Bioeng Biotechnol 3:353. https://doi.org/10.3389/fbioe201500053
Kim H, Jo BY, Kim HS (2017) Effect of different concentrations and ratios of ammonium, nitrate, and phosphate on growth of the blue-green alga (cyanobacterium) Microcystis aeruginosa isolated from the Nakdong River, Korea. Algae 32(4):275–284. https://doi.org/10.4490/algae2017321023
Yodsuwan N, Sawayama S, Sirisansaneeyakul S (2017) Effect of nitrogen concentration on growth, lipid production and fatty acid profiles of the marine diatom Phaeodactylum tricornutum. Agric Nat Resour 51(3):190–197. https://doi.org/10.1016/janres201702004
Kirrolia A, Bishnoi NR, Singh R (2014) Response surface methodology as a decision-making tool for optimization of culture conditions of green microalgae Chlorella spp for biodiesel production. Ann Microbiol 64(3):1133–1147. https://doi.org/10.1007/s13213-013-0752-4
Ahmad S, Pathak VV, Kothari R, Kumar A, Krishna SBN (2018) Optimization of nutrient stress using C. pyrenoidosa for lipid and biodiesel production in integration with remediation in dairy industry wastewater using response surface methodology. 3 Biotech 8(8):326. https://doi.org/10.1007/s13205-018-1342-8
Yang J, Astatkie T, He QS (2016) A comparative study on the effect of unsaturation degree of camelina and canola oils on the optimization of bio-diesel production. Energy Rep 2:211–217. https://doi.org/10.1016/jegyr201608003
Kothari R, Pathak VV, Pandey A, Ahmad S, Srivastava C, Tyagi VV (2017) A novel method to harvest Chlorella sp via low cost bioflocculant: influence of temperature with kinetic and thermodynamic functions. Bioresour Technol 225:84–89. https://doi.org/10.1016/jbiortech201611050
Ahmad S, Kothari R, Pathak VV, Pandey MK (2018) Fuel quality index: a novel experimental evaluation tool for biodiesel prepared from waste cooking oil. Waste Biomass Valoriz:1–11. https://doi.org/10.1007/s12649-018-0250-9
Kothari R, Ahmad S, Pathak VV, Pandey AK, Saidur R (2018) Fuel quality index of transesterified and nontransestried oil samples of mustard and ricebran oil. 5th IET International Conference on Clean Energy and Technology (CEAT2018), 6 pp. https://doi.org/10.1049/cp.2018.1333
APHA/AWWA/WEF Standard Methods for the Examination of Water and Wastewater (2012) Stand methods 541. ISBN 9780875532356 https://doi.org/10.5860/choice49-6910
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917
Pathak VV, Kothari R, Chopra AK, Ahmad S, Pandey AK, Rahim NA (2016) Effect of solvent extraction methods on oil yield and its parametric feasibility with C. pyrenoidosa. IET Digital Library 87(6). https://doi.org/10.1049/cp20161344
Higgins BT, Gennity I, Fitzgerald PS, Ceballos SJ, Fiehn O, Vander Gheynst JS (2018) Algal–bacterial synergy in treatment of winery wastewater. npj Clean Water 1(1):6. https://doi.org/10.1038/s41545-018-0005-y
Planavsky NJ (2014) The elements of marine life. Nat Geosci 7:855–856. https://doi.org/10.1038/ngeo2307
Saifuddin N, Raziah AZ, Farah H (2009) Production of biodiesel from high acid value waste cooking oil using an optimized lipase enzyme/acid-catalyzed hybrid process. Aust J Chem 6:485–495. https://doi.org/10.1155/2009/801756
Zhang Y, Dube MA, McLean DDL, Kates M (2003) Biodiesel production from waste cooking oil: 1. Process design and technological assessment. Bioresour Technol (89):1–16. https://doi.org/10.1016/s0960-8524(03)00150-0
Osundeko O, Davies H, Pittman JK (2013) Oxidative stress-tolerant microalgae strains are highly efficient for biofuel feedstock production on wastewater. Biomass Bioenergy 56:284–294. https://doi.org/10.1016/jbiombioe201305027
Oluwaniyi OO, Dosumu OO (2009) Preliminary studies on the effect of processing methods on the quality of three commonly consumed marine fishes in Nigeria. Biokemistri 21:1–7. https://doi.org/10.1016/jfoodchem201005051
Schober S, Mittelbach M (2007) Iodine value and biodiesel: is limitation still appropriate? Lipid Technol 19(12):281–284. https://doi.org/10.1002/lite200700091
Ramos MJ, Fernández CM, Casas A, Rodríguez L, Pérez Á (2009) Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour Technol 100:261–268. https://doi.org/10.1016/jbiortech200806039
Mandotra SK, Kumar P, Suseela MR, Ramteke PW (2014) Fresh water green microalga Scenedesmus abundans: a potential feedstock for high quality biodiesel production. Bioresour Technol 156:42–47. https://doi.org/10.1016/jbiortech201312127
ASTM D6751 (2012) Standard specification for biodiesel fuel (B100) blend stock for middle distillate fuels. https://doi.org/10.1520/d6751
Fuel standard (biodiesel) determination approved under section 21 of the Fuel Quality Standard Act 2002 by the Australian Minister for the Environment and Heritage (2003)
Yang F, Long L, Sun X, Wu H, Li T, Xiang W (2014) Optimization of medium using response surface methodology for lipid production by Scenedesmus sp. Mar Drugs 12(3):1245–1257. https://doi.org/10.3390/md12031245
Prias-Peñaranda MT, Cristiani-Urbina E, Montes-Horcasitas C, Esparza-Garcı́a F, Torzillo G, Cañizares-Villanueva RO (2013) Scenedesmus incrassatulus CLHE-Si01: a potential source of renewable lipid for high quality biodiesel production. Bioresour Technol 140:158–164. https://doi.org/10.1016/jbiortech201304080
Calixto CD, da Silva Santana JK, Tibúrcio VP, da Costa Sassi CF, da Conceição MM, Sassi R (2018) Productivity and fuel quality parameters of lipids obtained from 12 species of microalgae from the northeastern region of Brazil. Renew Energy 115:1144–1152. https://doi.org/10.1016/jrenene201709029
Battah MG, El-Ayoty YM, Esmael AE, El-Ghany SEA (2014) Effect of different concentrations of sodium nitrate, sodium chloride, and ferrous sulphate on the growth and lipid content of Chlorella vulgaris. Int J Agric Technol 10:339–353. https://doi.org/10.1007/s13213-014-0846-7
Minhas AK, Hodgson P, Barrow CJ, Adholeya A (2016) A review on the assessment of stress conditions for simultaneous production of microalgal lipids and carotenoids. Front Microbiol 7:546. https://doi.org/10.3389/fmicb201600546
Breuer G, Lamers PP, Martens DE, Draaisma RB, Wijffels RH (2012) The impact of nitrogen starvation on the dynamics of triacylglycerol accumulation in nine microalgae strains. Bioresour Technol 124:217–226. https://doi.org/10.1016/jbiortech201208003
Rodolfi L, Chini Zittelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112. https://doi.org/10.1002/bit22033
Simionato D, Block MA, La Rocca N, Jouhet J, Maréchal E, Finazzi G, Morosinotto T (2013) The response of Nannochloropsis gaditana to nitrogen starvation includes de novo biosynthesis of triacylglycerols, a decrease of chloroplast galactolipids, and reorganization of the photosynthetic apparatus. Eukaryot Cell 12:665–676. https://doi.org/10.1128/ec00363-12
Fan J, Yan C, Andre C, Shanklin J, Schwender J, Xu C (2012) Oil accumulation is controlled by carbon precursor supply for fatty acid synthesis in Chlamydomonas reinhardtii. Plant Cell Physiol 53:1380–1390. https://doi.org/10.1093/pcp/pcs082
Pancha I, Chokshi K, George B, Ghosh T, Paliwal C, Maurya R, Mishra S (2014) Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour Technol 156:146–154. https://doi.org/10.1016/jbiortech201401025
Razzak SA, Hossain MM, Lucky RA, Bassi AS, de Lasa H (2013) Integrated CO2 capture, wastewater treatment and biofuel production by microalgae culturing—a review. Renew Sust Energ Rev 27: 622–653. https://doi.org/10.1016/jrser201305063
Beck WS, Hall EK (2018) Confounding factors in algal phosphorus limitation experiments. bioRxiv:298281. https://doi.org/10.1101/298281
Chu FF, Chu PN, Cai PJ, Li WW, Lam PK, Zeng RJ (2013) Phosphorus plays an important role in enhancing biodiesel productivity of Chlorella vulgaris under nitrogen deficiency. Bioresour Technol 134:341–346. https://doi.org/10.1016/jbiortech201301131
Ramírez-Verduzco LF, Rodríguez-Rodríguez JE, del Rayo Jaramillo-Jacob A (2012) Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 91(1):102–111. https://doi.org/10.1016/jfuel201106070
Tuantet K, Temmink H, Zeeman G, Janssen M, Wijffels RH, BuismanC J (2014) Nutrient removal and microalgal biomass production on urine in a short light-path photobioreactor. Water Res 55:162–174. https://doi.org/10.1016/jwatres201402027
Kothari R, Pathak VV, Kumar V, Singh DP (2012) Experimental study for growth potential of unicellular alga Chlorella pyrenoidosa on dairy waste water: an integrated approach for treatment and biofuel production. Bioresour Technol 116:466–470. https://doi.org/10.1016/jbiortech201203121
Arias-Forero D, Hayashida G, Aranda M, Araya S, Portilla T, García A, Díaz-Palma P (2013) Protocol for maximizing the triglycerides-enriched lipids production from Dunaliella salina SA32007 biomass, isolated from the Salar de Atacama (Northern Chile). Adv Biosci Biotechnol 4:830–839. https://doi.org/10.4236/abb201348110
Converti A, Casazza AA, Ortiz EY, Perego P, Del Borghi M (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem Eng Process 48:1146–1151. https://doi.org/10.1016/jcep200903006
Ruangsomboon S, Ganmanee M, Choochote S (2013) Effects of different nitrogen, phosphorus, and iron concentrations and salinity on lipid production in newly isolated strain of the tropical green microalga, Scenedesmus dimorphus KMITL. J Appl Phycol 25:867–874. https://doi.org/10.5772/19358
Mandal S, Mallick N (2009) Microalga Scenedesmus obliquus as a potential source for biodiesel production. Appl Microbiol Biotechnol 84:281–291. https://doi.org/10.1007/s00253-009-1935-6
Kiran B, Pathak K, Kumar R, Deshmukh D (2016) Statistical optimization using central composite design for biomass and lipid productivity of microalga: a step towards enhanced biodiesel production. Ecol Eng 92:73–81. https://doi.org/10.1016/jecoleng201603026
Dahmen I, Chtourou H, Jebali A, Daassi D, Karray F, Hassairi I, Dhouib A (2014) Optimisation of the critical medium components for better growth of Picochlorum sp. and the role of stressful environments for higher lipid production. J Sci Food Agric 38:1381–1392. https://doi.org/10.1007/s00449-015-1379-6
Pruvost J, Pottier L, Legrand J (2006) Numerical investigation of hydrodynamic and mixing conditions in a torus photobioreactor. Chem Eng Sci 61:4476–4489. https://doi.org/10.1016/jces200602027
Ahmad M, Ullah K, Khan MA, Zafar M, Tariq M, Ali S, Sultana S (2011) Physicochemical analysis of hemp oil biodiesel: a promising non edible new source for bioenergy. Energy Sources Part A 33(14):1365–1137. https://doi.org/10.1080/155670362010499420
Bücker F, Santestevan NA, Roesch LF, Jacques RJS, Peralba MDCR, de Oliveira Camargo FA, Bento FM (2011) Impact of biodiesel on biodeterioration of stored Brazilian diesel oil. Int Biodeterior Biodegrad 65(1):172–178. https://doi.org/10.1016/jibiod201009008
Acharya N, Nanda P, Panda S, Acharya S (2017) Analysis of properties and estimation of optimum blending ratio of blended mahua biodiesel. Eng Sci Technol Int J 20(2):511–517. https://doi.org/10.1016/jjestch201612005
Gopinath A, Sairam K, Velraj R, Kumaresan G (2015) Effects of the properties and the structural configurations of fatty acid methyl esters on the properties of biodiesel fuel: a review. Proc Inst Mech Eng D 229(3):357–390. https://doi.org/10.1177/0954407014541103
Sivaramakrishnan K, Ravikumar P (2012) Determination of cetane number of biodiesel and its influence on physical properties. ARPN J Eng Appl Sci 7(2):205–211. https://doi.org/10.4271/2007-01-0077
Funding
The authors of the manuscript are grateful to the University Grant Commission (UGC), India for providing financial support and are thankful to the Head, Department of Environmental Science and Microbiology and Director, USIC of Babasaheb Bhimrao Ambedkar University (BBAU), Lucknow, India for providing the instrumentation facilities for this research study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ahmad, S., Kothari, R., Pathania, D. et al. Optimization of nutrients from wastewater using RSMfor augmentation of Chlorella pyrenoidosa with enhanced lipid productivity, FAME content, and its quality assessment using fuel quality index. Biomass Conv. Bioref. 10, 495–512 (2020). https://doi.org/10.1007/s13399-019-00443-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-019-00443-z