Abstract
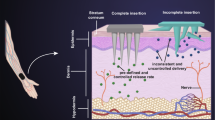
Nanocrystals can enhance skin penetration of drug by increased saturation solubility, dissolution rate and adhesion on the skin. Apremilast is ‘difficult-to-deliver’ in viable layers (epidermis, dermis) and stratum corneum (SC) owing to its poor aqueous solubility and modest lipophilicity, respectively. Apremilast is currently available as oral tablet formulation for the indication of psoriasis but its effectiveness is limited by systemic side effects. Therefore, the present study aimed to develop novel nanocrystal-based formulations of apremilast for improved topical delivery. Nanosuspension was prepared using wet media milling and exhibited a mean particle size of 200 nm. The saturation solubility of nanocrystals was improved by 2-fold than micronized apremilast and showed significant advantage during dissolution study. Nanosuspension and micronized apremilast was incorporated into gel and cream and characterized for rheological properties. Skin permeation and ex vivo dermatokinetic study of topical formulations were performed on pig ear skin at a dose of 1% w/w using Franz diffusion cells. Skin permeation studies indicated that non-detectable amount of apremilast permeated through pig ear skin during exposure of formulations. Nanosuspension showed 2.6- and 3.2-fold drug penetration in SC and viable layers, respectively, over microsuspension. Nanogel showed 2.7- and 2.4-fold drug penetration in SC and viable layers, respectively, over microgel. Nanocream showed 1.2- and 2.8-fold drug penetration in SC and viable layers, respectively, over microcream. Thus, nanocrystal-based formulations of apremilast aid in selective delivery into viable layers by crossing the SC barrier. This is of paramount importance in enhancing therapeutic effectiveness utilizing localized delivery and provides an alternative delivery approach for the treatment of psoriasis.

Graphical abstract














Similar content being viewed by others
References
Wu X, Guy RH. Applications of nanoparticles in topical drug delivery and in cosmetics. J Drug Deliv Sci Technol. 2009;19(6):371–84.
Gao L, Liu G, Ma J, Wang X, Zhou L, Li X. Drug nanocrystals: in vivo performances. J Control Release. 2012;160(3):418–30.
Sinha B, Müller RH, Möschwitzer JP. Bottom-up approaches for preparing drug nanocrystals: formulations and factors affecting particle size. Int J Pharm. 2013;453(1):126–41.
Shete G, Jain H, Punj D, Prajapat H, Akotiya P, Bansal AK. Stabilizers used in nanocrystal based drug delivery systems. J Excip Food Chem. 2014;5(4):184–209.
Müller RH, Zhai X, Romero GB, Keck CM. Nanocrystals for passive dermal penetration enhancement. In: Dragicevic N, Maibach HI, editors. Percutaneous penetration enhancers chemical methods in penetration enhancement: nanocarriers. 1st ed. Berlin: Springer-Verlag Berlin Heidelberg; 2016. p. 283–95.
Zhai X, Lademann J, Keck CM, Müller RH. Nanocrystals of medium soluble actives - novel concept for improved dermal delivery and production strategy. Int J Pharm. 2014;470(1–2):141–50.
Al-Shaal L, Shegokar R, Müller RH. Production and characterization of antioxidant apigenin nanocrystals as a novel UV skin protective formulation. Int J Pharm. 2011;420(1):133–40.
Mitri K, Shegokara R, Gohla S, Anselmi C, Müller RH. Lutein nanocrystals as antioxidant formulation for oral and dermal delivery. Int J Pharm. 2011;420(1):141–6.
Lai F, Pireddu R, Corrias F, Fadda AM, Valenti D, Pini E, et al. Nanosuspension improves tretinoin photostability and delivery to the skin. Int J Pharm. 2013;458(1):104–9.
Pireddu R, Sinico C, Ennas G, Marongiu F, Muzzalupo R, Lai F, et al. Novel nanosized formulations of two diclofenac acid polymorphs to improve topical bioavailability. Eur J Pharm Sci. 2015;77:208–15.
Vidlářová L, Romero GB, Hanuš J, Štěpánek F, Müller RH. Nanocrystals for dermal penetration enhancement - effect of concentration and underlying mechanisms using curcumin as model. Eur J Pharm Sci. 2016;104:216–25.
Hatahet T, Morille M, Hommoss A, Dorandeu C, Müller RH, Bégu S. Dermal quercetin smartCrystals®: formulation development, antioxidant activity and cellular safety. Eur J Pharm Biopharm. 2016;102:51–63.
Sinico C, Pireddu R, Pini E, Valenti D, Caddeo C, Fadda AM, et al. Enhancing topical delivery of resveratrol through a nanosizing approach. Planta Med. 2017;83(5):476–81.
Pyo SM, Meinke M, Keck CM, Müller RH. Rutin - increased antioxidant activity and skin penetration by nanocrystal technology (smartCrystals). Cosmetics. 2016;3(1):1–10.
Shen C, Shen B, Liu X, Yuan H. Nanosuspensions based gel as delivery system of nitrofurazone for enhanced dermal bioavailability. J Drug Deliv Sci Technol. 2018;43:1–11.
Balato A, Balato N, Megna M, Schiattarella M, Lembo S, Ayala F. Pathogenesis of psoriasis: the role of pro-inflammatory cytokines produced by keratinocytes. In: Soung J, Koo B, editor. Psoriasis. IntechOpen; 2012. p. 9–28.
Chandra A, Ray A, Senapati S, Chatterjee R. Genetic and epigenetic basis of psoriasis pathogenesis. Mol Immunol. 2015;64(2):313–23.
Rahman M, Alam K, Ahmad MZ, Gupta G, Afzal M, Akhter S, et al. Classical to current approach for treatment of psoriasis: a review. Endocr Metab Immune Disord Drug Targets. 2012;12(3):287–302.
Laws PM, Young HS. Topical treatment of psoriasis. Expert Opin Pharmacother. 2010;11(12):1999–2009.
Shutty B, West C, Pellerin M, Feldman S. Apremilast as a treatment for psoriasis. Expert Opin Pharmacother. 2012;13(12):1761–70.
Celgene-Corporation. OTEZLA® (apremilast) tablets, for oral use: US prescribing information. 2019. http://www.celgene.com/content/uploads/otezla-pi.pdf. Accessed 8 Feb 2020.
Keating GM. Apremilast: a review in psoriasis and psoriatic arthritis. Drugs. 2017;77(4):459–72.
EMA. Assessment report Otezla. 2014. https://www.ema.europa.eu/en/documents/assessment-report/otezla-epar-public-assessment-report_en.pdf. Accessed 5 April 2020.
Drugbank. Apremilast. 2020. https://www.drugbank.ca/drugs/DB05676. Accessed 25 May 2020.
Lu Y, Shen X, Hang T, Song M. Identification and characterization of process-related substances and degradation products in apremilast: process optimization and degradation pathway elucidation. J Pharm Biomed Anal. 2017;141:70–8.
Zhang Y, Huo M, Zhou J, Zou A, Li W, Yao C, et al. DDSolver: an add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 2010;12(3):263–71.
FDA. Guidance for industry dissolution testing of immediate release solid oral dosage forms. 1997. https://www.fda.gov/media/70936/download. Accessed 8 Feb 2020.
Merisko-Liversidge E, Liversidge GG, Cooper ER. Nanosizing: a formulation approach for poorly-water-soluble compounds. Eur J Pharm Sci. 2003;18(2):113–20.
Peltonen L, Hirvonen J. Pharmaceutical nanocrystals by nanomilling: critical process parameters, particle fracturing and stabilization methods. J Pharm Pharmacol. 2010;62(11):1569–79.
Seweryn A. Interactions between surfactants and the skin - theory and practice. Adv Colloid Interf Sci. 2018;256:242–55.
Dumortier G, Grossiord JL, Agnely F, Chaumeil JC. A review of poloxamer 407 pharmaceutical and pharmacological characteristics. Pharm Res. 2006;23(12):2709–28.
Tuomela A, Hirvonen J, Peltonen L. Stabilizing agents for drug nanocrystals: effect on bioavailability. Pharmaceutics. 2016;8(2):1–18.
Bodratti AM, Alexandridis P. Formulation of poloxamers for drug delivery. J Funct Biomater. 2018;9(1):1–24.
Bhattacharjee S. DLS and zeta potential - what they are and what they are not? J Control Release. 2016;235:337–51.
Muller GW, Schafer PH, Man H, Ge C, Xu J, inventors; Solid forms comprising (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, compositions thereof, and uses thereof. United States patent 7,893,101. 2011 22 February.
Murdande SB, Shah DA, Dave RH. Impact of nanosizing on solubility and dissolution rate of poorly soluble pharmaceuticals. J Pharm Sci. 2015;104(6):2094–102.
Chiarappa G, Piccolo A, Colombo I, Hasa D, Voinovich D, Moneghini M, et al. Exploring the shape influence on melting temperature, enthalpy, and solubility of organic drug nanocrystals by a thermodynamic model. Cryst Growth Des. 2017;17(8):4072–83.
Liu P, Wulf OD, Laru J, Heikkilä T, van Veen B, Kiesvaara J, et al. Dissolution studies of poorly soluble drug nanosuspensions in non-sink conditions. AAPS PharmSciTech. 2013;14(2):748–56.
Eccleston GM, Bakhshaee M, Hudson NE, Richards DH. Rheological behavior of nasal sprays in shear and extension. Drug Dev Ind Pharm. 2000;26(9):975–83.
Kumbhar D, Wavikar P, Vavia P. Niosomal gel of lornoxicam for topical delivery: in vitro assessment and pharmacodynamic activity. AAPS PharmSciTech. 2013;14(3):1072–82.
Acknowledgements
Prashantkumar K. Parmar would like to gratefully acknowledge Rajiv Gandhi National Fellowship (RGNF, New Delhi, India) for providing Ph.D. Scholarship. Prashantkumar K. Parmar would also like to thank Sonu Dalsania, Pooja Sharma, Dnayaneshwar Kale, Payal Sharma and Jhanvi Wadhawan for their support and help during this project. The authors would like to acknowledge NIPER - S.A.S. Nagar for providing necessary facilities for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Parmar, P.K., Bansal, A.K. Novel nanocrystal-based formulations of apremilast for improved topical delivery. Drug Deliv. and Transl. Res. 11, 966–983 (2021). https://doi.org/10.1007/s13346-020-00809-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00809-1