Abstract
Glioblastoma multiforme (GBM) is the most common and aggressive form of the primary brain tumors in humans. The intricate pathophysiology, the development of resistance by tumor cells, and the inability of the drugs to effectively cross the blood-brain and blood-tumor barriers result in poor prognosis for GBM patients, with a median survival time of only 1 to 2 years. Nose-to-brain delivery offers an attractive, noninvasive strategy to enhance drug penetration or transport novel drug/gene carriers into the brain. Although the exact mechanism of intranasal delivery remains elusive, the olfactory and trigeminal nerve pathways have been found to play a vital role in circumventing the traditional barriers of brain targeting. This review discusses the intranasal pathway as a novel domain for delivering drugs and nanocarriers encapsulating drugs/genes, as well as stem cell carriers specifically to the glioma cells. Considering the fact that most of these studies are still in preclinical stage, translating such intranasal delivery strategies from bench to bedside would be a critical step for better management and prognosis of GBM.

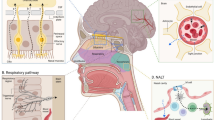
Graphical abstract




Similar content being viewed by others
References
Ostrom QT, Gittleman H, Farah P, Ondracek A, Chen Y, Wolinsky Y, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2006-2010. Neuro Oncol. 2013;15(Suppl 2):ii1–ii56.
de Robles P, Fiest KM, Frolkis AD, Pringsheim T, Atta C, St Germaine-Smith C, et al. The worldwide incidence and prevalence of primary brain tumors: a systematic review and meta-analysis. Neuro-Oncology. 2015;17:776–83.
Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007-2011. Neuro Oncol. 2014;16(Suppl 4):iv1–iv63.
Huse JT, Holland EC. Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat Rev Cancer. 2010;10:319.
Jansen M, de Witt Hamer PC, Witmer AN, Troost D, van Noorden CJF. Current perspectives on antiangiogenesis strategies in the treatment of malignant gliomas. Brain Res Rev. 2004;45:143–63.
Tanaka S, Louis DN, Curry WT, Batchelor TT, Dietrich J. Diagnostic and therapeutic avenues for glioblastoma: no longer a dead end? Nat Rev Clin Oncol. 2012;10:14.
Koo Y-EL, Reddy GR, Bhojani M, Schneider R, Philbert MA, Rehemtulla A, et al. Brain cancer diagnosis and therapy with nanoplatforms. Adv Drug Deliv Rev. 2006;58:1556–77.
Sanai N, Berger MS. Glioma extent of resection and its impact on patient outcome. Neurosurgery. 2008;62:753–66.
Yang Y, He MZ, Li T, Yang X. MRI combined with PET-CT of different tracers to improve the accuracy of glioma diagnosis: a systematic review and meta-analysis. Neurosurg Rev. 2019;42:185–95.
Huk WJ, Gademann G. Magnetic resonance imaging (MRI): method and early clinical experiences in diseases of the central nervous system. Neurosurg Rev. 1984;7:259–80.
Ricci PE, Dungan DH. Imaging of low- and intermediate-grade gliomas. Semin Radiat Oncol. 2001;11:103–12.
Dean BL, Drayer BP, Bird CR, Flom RA, Hodak JA, Coons SW, et al. Gliomas: classification with MR imaging. Radiology. 1990;174:411–5.
Belliveau J-G, Bauman G, Macdonald DR. Detecting tumor progression in glioma: current standards and new techniques. Expert Rev Anticancer Ther. 2016;16:1177–88.
Vézina L-G. Imaging of central nervous system tumors in children: advances and limitations. J Child Neurol. 2008;23:1128–35.
Upadhyay N, Waldman AD. Conventional MRI evaluation of gliomas. Br J Radiol. 2011;84 Spec No 2:S107–SS11.
Puttick S, Bell C, Dowson N, Rose S, Fay M. PET, MRI, and simultaneous PET/MRI in the development of diagnostic and therapeutic strategies for glioma. Drug Discov Today. 2015;20:306–17.
Otakar Belohlavek AU, Pavel Fencl AU, Martin Majovsky AU, Monika Jaruskova AU, Benes V. FLT-PET in previously untreated patients with low-grade glioma can predict their overall survival. FLT-PET in previously untreated patients with low-grade glioma can predict their overall survival. Nucl Med Red Cent East Eur. 2014;17:7–12.
Floeth FW, Pauleit D, Sabel M, Stoffels G, Reifenberger G, Riemenschneider MJ, et al. Prognostic value of O-(2-18F-Fluoroethyl)-l-tyrosine PET and MRI in low-grade glioma. J Nucl Med. 2007;48:519–27.
Jansen NL, Suchorska B, Wenter V, Schmid-Tannwald C, Todica A, Eigenbrod S, et al. Prognostic significance of dynamic 18F-FET PET in newly diagnosed astrocytic high-grade glioma. J Nucl Med. 2015;56:9–15.
Jung T-Y, Min J-J, Bom H-S, Jung S, Kim I-Y, Lim S-H, et al. Prognostic value of post-treatment metabolic tumor volume from 11C-methionine PET/CT in recurrent malignant glioma. Neurosurg Rev. 2017;40:223–9.
Kobayashi K, Hirata K, Yamaguchi S, Manabe O, Terasaka S, Kobayashi H, et al. Prognostic value of volume-based measurements on 11C-methionine PET in glioma patients. Eur J Nucl Med Mol Imaging. 2015;42:1071–80.
Schwarzenberg J, Czernin J, Cloughesy TF, Ellingson BM, Pope WB, Geist C, et al. 3′-deoxy-3’-18F-fluorothymidine PET and MRI for early survival predictions in patients with recurrent malignant glioma treated with bevacizumab. J Nucl Med. 2012;53:29–36.
Yoo MY, Paeng JC, Cheon GJ, Lee DS, Chung J-K, Kim EE, et al. Prognostic value of metabolic tumor volume on (11)C-methionine PET in predicting progression-free survival in high-grade glioma. Nucl Med Mol Imaging. 2015;49:291–7.
Stummer W, van den Bent MJ, Westphal M. Cytoreductive surgery of glioblastoma as the key to successful adjuvant therapies: new arguments in an old discussion. Acta Neurochir. 2011;153:1211–8.
Wang Y, Jiang T. Understanding high grade glioma: molecular mechanism, therapy and comprehensive management. Cancer Lett. 2013;331:139–46.
van den Bent MJ, Carpentier AF, Brandes AA, Sanson M, Taphoorn MJB, Bernsen HJJA, et al. Adjuvant procarbazine, lomustine, and vincristine improves progression-free survival but not overall survival in newly diagnosed anaplastic oligodendrogliomas and oligoastrocytomas: a randomized European Organisation for Research and Treatment of Cancer phase III trial. JCO. 2006;24:2715–22.
Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, et al. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-Oncology. 2003;5:79–88.
Noël G, Schott R, Froelich S, Gaub M-P, Boyer P, Fischer-Lokou D, et al. Retrospective comparison of chemoradiotherapy followed by adjuvant chemotherapy, with or without prior Gliadel implantation (carmustine) after initial surgery in patients with newly diagnosed high-grade gliomas. Int J Radiat Oncol Biol Phys. 2012;82:749–55.
McGirt MJ, Than KD, Weingart JD, Chaichana KL, Attenello FJ, Olivi A, et al. Gliadel (BCNU) wafer plus concomitant temozolomide therapy after primary resection of glioblastoma multiforme. J Neurosurg. 2009;110:583–8.
Wick W, Weller M. Classification and management of anaplastic gliomas. Curr Opin Neurol[Internet]. 2009;22 Available from: https://journals.lww.com/co-neurology/Fulltext/2009/12000/Classification_and_management_of_anaplastic.17.aspx. Accessed 7/11/2019.
Wick W, Hartmann C, Engel C, Stoffels M, Felsberg J, Stockhammer F, et al. NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine, and vincristine or temozolomide. JCO. 2009;27:5874–80.
Ghadiri M, Young MP, Traini D. Strategies to enhance drug absorption via nasal and pulmonary routes. Pharmaceutics. 2019;11:113–132.
Costantino HR, Illum L, Brandt G, Johnson PH, Quay SC. Intranasal delivery: physicochemical and therapeutic aspects. Int J Pharm. 2007;337:1–24.
Jadhav KR, Gambhire MN, Shaikh IM, Kadam VJ, Pisal SS. Nasal drug delivery system-factors affecting and applications. Current Drug Therapy. 2007;2:27–38.
Furubayashi T, Kamaguchi A, Kawaharada K, Masaoka Y, Kataoka M, Yamashita S, et al. Kinetic model to predict the absorption of nasally applied drugs from in vitro transcellular permeability of drugs. Biol Pharm Bull. 2007;30:1007–10.
Privalova AM, Gulyaeva NV, Bukreeva TV. Intranasal administration: a prospective drug delivery route to the brain. Neurochem J. 2012;6:77–88.
Agrawal M, Saraf S, Saraf S, Antimisiaris SG, Chougule MB, Shoyele SA, et al. Nose-to-brain drug delivery: an update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J Control Release. 2018;281:139–77.
Mistry A, Stolnik S, Illum L. Nanoparticles for direct nose-to-brain delivery of drugs. Int J Pharm. 2009;379:146–57.
Battaglia L, Panciani PP, Muntoni E, Capucchio MT, Biasibetti E, De Bonis P, et al. Lipid nanoparticles for intranasal administration: application to nose-to-brain delivery. Expert Opin Drug Deliv. 2018;15:369–78.
Kulkarni AD, Vanjari YH, Sancheti KH, Belgamwar VS, Surana SJ, Pardeshi CV. Nanotechnology-mediated nose to brain drug delivery for Parkinson’s disease: a mini review. J Drug Target. 2015;23:775–88.
Yang Z-Z, Zhang Y-Q, Wang Z-Z, Wu K, Lou J-N, Qi X-R. Enhanced brain distribution and pharmacodynamics of rivastigmine by liposomes following intranasal administration. Int J Pharm. 2013;452:344–54.
Priprem A, Watanatorn J, Sutthiparinyanont S, Phachonpai W, Muchimapura S. Anxiety and cognitive effects of quercetin liposomes in rats. Nanomedicine. 2008;4:70–8.
Migliore MM, Vyas TK, Campbell RB, Amiji MM, Waszczak BL. Brain delivery of proteins by the intranasal route of administration: a comparison of cationic liposomes versus aqueous solution formulations. J Pharm Sci. 2010;99:1745–61.
Arumugam K, Subramanian G, Mallayasamy S, Averineni R, Reddy M, Udupa N. A study of rivastigmine liposomes for delivery into the brain through intranasal route. Acta Pharm [Internet]. 2008;58 Available from: https://content.sciendo.com/view/journals/acph/58/3/article-p287.xml. Accessed 7/11/2019.
Rinaldi F, Hanieh PN, Chan LKN, Angeloni L, Passeri D, Rossi M, et al. Chitosan glutamate-coated niosomes: a proposal for nose-to-brain delivery. Pharmaceutics. 2018;10:38.
Kumar M, Misra A, Mishra AK, Mishra P, Pathak K. Mucoadhesive nanoemulsion-based intranasal drug delivery system of olanzapine for brain targeting. J Drug Target. 2008;16:806–14.
Kumar M, Misra A, Babbar AK, Mishra AK, Mishra P, Pathak K. Intranasal nanoemulsion based brain targeting drug delivery system of risperidone. Int J Pharm. 2008;358:285–91.
Jogani VV, Shah PJ, Mishra P, Mishra AK, Misra AR. Intranasal mucoadhesive microemulsion of tacrine to improve brain targeting. Alzheimer Dis Assoc Disord [Internet]. 2008;22 Available from: https://journals.lww.com/alzheimerjournal/Fulltext/2008/04000/Intranasal_Mucoadhesive_Microemulsion_of_Tacrine.5.aspx.
Alam MI, Baboota S, Ahuja A, Ali M, Ali J, Sahni JK. Intranasal administration of nanostructured lipid carriers containing CNS acting drug: pharmacodynamic studies and estimation in blood and brain. J Psychiatr Res. 2012;46:1133–8.
Joshi AS, Patel HS, Belgamwar VS, Agrawal A, Tekade AR. Solid lipid nanoparticles of ondansetron HCl for intranasal delivery: development, optimization and evaluation. J Mater Sci Mater Med. 2012;23:2163–75.
Eskandari S, Varshosaz J, Minaiyan M, Tabbakhian M. Brain delivery of valproic acid via intranasal administration of nanostructured lipid carriers: in vivo pharmacodynamic studies using rat electroshock model. Int J Nanomedicine. 2011;6:363–71.
Gao X, Tao W, Lu W, Zhang Q, Zhang Y, Jiang X, et al. Lectin-conjugated PEG–PLA nanoparticles: preparation and brain delivery after intranasal administration. Biomaterials. 2006;27:3482–90.
Gao X, Wu B, Zhang Q, Chen J, Zhu J, Zhang W, et al. Brain delivery of vasoactive intestinal peptide enhanced with the nanoparticles conjugated with wheat germ agglutinin following intranasal administration. J Control Release. 2007;121:156–67.
Liu Q, Shao X, Chen J, Shen Y, Feng C, Gao X, et al. In vivo toxicity and immunogenicity of wheat germ agglutinin conjugated poly(ethylene glycol)-poly(lactic acid) nanoparticles for intranasal delivery to the brain. Toxicol Appl Pharmacol. 2011;251:79–84.
Park YJ, Chang L-C, Liang JF, Moon C, Chung C-P, Yang VC. Nontoxic membrane translocation peptide from protamine, low molecular weight protamine (LMWP), for enhanced intracellular protein delivery: in vitro and in vivo study. FASEB J. 2005;19:1555–7.
Jain R, Nabar S, Dandekar P, Patravale V. Micellar nanocarriers: potential nose-to-brain delivery of zolmitriptan as novel migraine therapy. Pharm Res. 2010;27:655–64.
Jain R, Nabar S, Dandekar P, Hassan P, Aswal V, Talmon Y, et al. Formulation and evaluation of novel micellar nanocarrier for nasal delivery of sumatriptan. Nanomedicine. 2010;5:575–87.
Shingaki T, Inoue D, Furubayashi T, Sakane T, Katsumi H, Yamamoto A, et al. Transnasal delivery of methotrexate to brain tumors in rats: a new strategy for brain tumor chemotherapy. Mol Pharm. 2010;7:1561–8.
Hashizume R, Ozawa T, Gryaznov SM, Bollen AW, Lamborn KR, Frey WH 2nd, et al. New therapeutic approach for brain tumors: intranasal delivery of telomerase inhibitor GRN163. Neuro-Oncology. 2008;10:112–20.
Özduman K, Wollmann G, Piepmeier JM, van den Pol AN. Systemic vesicular stomatitis virus selectively destroys multifocal glioma and metastatic carcinoma in brain. J Neurosci. 2008;28:1882.
Reitz M, Demestre M, Sedlacik J, Meissner H, Fiehler J, Kim SU, et al. Intranasal delivery of neural stem/progenitor cells: a noninvasive passage to target intracerebral glioma. Stem Cells Transl Med. 2012;1:866–73.
da Fonseca CO, Simão M, Lins IR, Caetano RO, Futuro D, Quirico-Santos T. Efficacy of monoterpene perillyl alcohol upon survival rate of patients with recurrent glioblastoma. J Cancer Res Clin Oncol. 2011;137:287–93.
Colombo M, Figueiró F, de Fraga DA, Teixeira HF, Battastini AMO, Koester LS. Kaempferol-loaded mucoadhesive nanoemulsion for intranasal administration reduces glioma growth in vitro. Int J Pharm. 2018;543:214–23.
Wang W, Swenson S, Cho H-Y, Hofman FM, Schönthal AH, Chen TC. Efficient brain targeting and therapeutic intracranial activity of bortezomib through intranasal co-delivery with NEO100 in rodent glioblastoma models. J Neurosurg JNS. 2019;132:959–967.65.
Yu D, Li G, Lesniak MS, Balyasnikova IV. Intranasal delivery of therapeutic stem cells to glioblastoma in a mouse model. J Vis Exp. 2017;124:55845. https://doi.org/10.3791/55845.
JR Pineda, M Jeitany, A Andrieux, MP Junier, H Chneiweiss, Boussin FD. Intranasal Administration of temozolomide delayed the development of brain tumors initiated by human glioma stem-like cell in nude mice. J Cancer Sci Ther [Internet]. 2017 [cited 2019 Jul 11];09. Available from: https://www.omicsonline.org/open-access/intranasal-administration-of-temozolomide-delayed-the-development-ofbrain-tumors-initiated-by-human-glioma-stemlike-cell-in-nude-m-1948-5956-1000445.php?aid=87652
Yu D, Li G, Lesniak MS, Balyasnikova IV. Intranasal delivery of therapeutic stem cells to glioblastoma in a mouse model. J Vis Exp. 2017. https://doi.org/10.3791/55845.
Sekerdag E, Lüle S, Bozdağ Pehlivan S, Öztürk N, Kara A, Kaffashi A, et al. A potential non-invasive glioblastoma treatment: nose-to-brain delivery of farnesylthiosalicylic acid incorporated hybrid nanoparticles. J Control Release. 2017;261:187–98.
Van Woensel M, Wauthoz N, Rosière R, Mathieu V, Kiss R, Lefranc F, et al. Development of siRNA-loaded chitosan nanoparticles targeting Galectin-1 for the treatment of glioblastoma multiforme via intranasal administration. J Control Release. 2016;227:71–81.
Zhuang X, Teng Y, Samykutty A, Mu J, Deng Z, Zhang L, et al. Grapefruit-derived nanovectors delivering therapeutic miR17 through an intranasal route inhibit brain tumor progression. Mol Ther. 2016;24:96–105.
Taki H, Kanazawa T, Akiyama F, Takashima Y, Okada H. Intranasal delivery of camptothecin-loaded tat-modified nanomicells for treatment of intracranial brain tumors. Pharmaceuticals (Basel). 2012;5:1092–102.
Andersson M, Greiff L, Öman H, Svensson C, Wollmer P, Persson CGA. Permeation of polysucrose 15 000 across the human nasal mucosa in vivo. Allergy. 1997;52:1229–32.
Machida M, Sano K, Arakawa M, Hayashi M, Awazu S. Absorption of recombinant human granulocyte colony-stimulating factor (rhG-CSF) from rat nasal mucosa. Pharm Res. 1993;10:1372–7.
Bruce DG, Chisholm DJ, Storlien LH, Borkman M, Kraegen EW. Meal-time intranasal insulin delivery in type 2 diabetes. Diabet Med. 1991;8:366–70.
Hedin L, Olsson B, Diczfalusy M, Flyg C, Petersson AS, Rosberg S, et al. Intranasal administration of human growth hormone (hGH) in combination with a membrane permeation enhancer in patients with GH deficiency: a pharmacokinetic study. J Clin Endocrinol Metab. 1993;76:962–7.
Drejer K, Vaag A, Bech K, Hansen P, Sørensen AR, Mygind N. Intranasal administration of insulin with phospholipid as absorption enhancer: pharmacokinetics in normal subjects. Diabet Med. 1992;9:335–40.
Dua R, Duncan M, Zia H, Needham TE. The influence of the enhancer dimyristoylphosphatidylglycerol and formulation factors on the nasal absorption of salmon calcitonin. Drug Deliv. 1998;5:127–34.
Schipper NGM, Romeijn SG, Verhoef JC, Merkus FWHM. Nasal insulin delivery with dimethyl-β-cyclodextrin as an absorption enhancer in rabbits: powder more effective than liquid formulations. Pharm Res. 1993;10:682–6.
Merkus FWHM, Verhoef JC, Romeijn SG, Schipper NGM. Absorption enhancing effect of cyclodextrins on intranasally administered insulin in rats. Pharm Res. 1991;8:588–92.
Illum L, Farraj NF, Davis SS. Chitosan as a novel nasal delivery system for peptide drugs. Pharm Res. 1994;11:1186–9.
Natsume H, Iwata S, Ohtake K, Miyamoto M, Yamaguchi M, Hosoya K, et al. Screening of cationic compounds as an absorption enhancer for nasal drug delivery. Int J Pharm. 1999;185:1–12.
Kararli TT, Needham TE, Schoenhard G, Baron DA, Schmidt RE, Katz B, et al. Enhancement of nasal delivery of a renin inhibitor in the rat using emulsion formulations. Pharm Res. 1992;9:1024–8.
Wang F, Jiang X, Lu W. Profiles of methotrexate in blood and CSF following intranasal and intravenous administration to rats. Int J Pharm. 2003;263:1–7.
van Woensel M, Wauthoz N, Rosière R, Amighi K, Mathieu V, Lefranc F, et al. Formulations for intranasal delivery of pharmacological agents to combat brain disease: a new opportunity to tackle GBM? Cancers (Basel). 2013;5:1020–48.
Shingaki T, Hidalgo IJ, Furubayashi T, Katsumi H, Sakane T, Yamamoto A, et al. The transnasal delivery of 5-fluorouracil to the rat brain is enhanced by acetazolamide (the inhibitor of the secretion of cerebrospinal fluid). Int J Pharm. 2009;377:85–91.
Cho H-Y, Wang W, Jhaveri N, Torres S, Tseng J, Leong MN, et al. Perillyl alcohol for the treatment of temozolomide-resistant gliomas. Mol Cancer Ther. 2012;11:2462.
Khan AR, Liu M, Khan MW, Zhai G. Progress in brain targeting drug delivery system by nasal route. J Control Release. 2017;268:364–89.
Bourganis V, Kammona O, Alexopoulos A, Kiparissides C. Recent advances in carrier mediated nose-to-brain delivery of pharmaceutics. Eur J Pharm Biopharm. 2018;128:337–62.
Henning A, Schneider M, Nafee N, Muijs L, Rytting E, Wang X, et al. Influence of particle size and material properties on mucociliary clearance from the airways. J Aerosol Med Pulm Drug Deliv. 2010;23:233–41.
Kirch J, Guenther M, Doshi N, Schaefer UF, Schneider M, Mitragotri S, et al. Mucociliary clearance of micro- and nanoparticles is independent of size, shape and charge—an ex vivo and in silico approach. J Control Release. 2012;159:128–34.
Phukan K, Nandy M, Sharma RB, Sharma HK. Nanosized drug delivery systems for direct nose to brain targeting: a review. Recent Pat Drug Deliv Formul. 2016;10:156–64.
Thakur A, Joshi N, Shanmugam T, Banerjee R. Proapoptotic miltefosine nanovesicles show synergism with paclitaxel: implications for glioblastoma multiforme therapy. Cancer Lett. 2013;334:274–83.
Khan A, Imam SS, Aqil M, Ahad A, Sultana Y, Ali A, et al. Brain targeting of temozolomide via the intranasal route using lipid-based nanoparticles: brain pharmacokinetic and scintigraphic analyses. Mol Pharm. 2016;13:3773–82.
Patil NL, Mahajan HS. Quercetin loaded nanostructured lipid carriers for nose to brain delivery: in vitro and in vivo studies. American Journal of Advanced Drug Delivery [Internet]. 2017 [cited 2019 Jul 9];6. Available from: http://www.imedpub.com/abstract/quercetin-loaded-nanostructured-lipid-carriers-for-nose-to-brain-delivery-in-vitro-and-in-vivo-studies-21621.html
Gadhave D, Gorain B, Tagalpallewar A, Kokare C. Intranasal teriflunomide microemulsion: an improved chemotherapeutic approach in glioblastoma. J Drug Deliv Sci Technol. 2019;51:276–89.
Jain DS, Bajaj AN, Athawale RB, Shikhande SS, Pandey A, Goel PN, et al. Thermosensitive PLA based nanodispersion for targeting brain tumor via intranasal route. Mater Sci Eng C. 2016;63:411–21.
Kanazawa T, Taki H, Tanaka K, Takashima Y, Okada H. Cell-penetrating peptide-modified block copolymer micelles promote direct brain delivery via intranasal administration. Pharm Res. 2011;28:2130–9.
Kanazawa T, Akiyama F, Kakizaki S, Takashima Y, Seta Y. Delivery of siRNA to the brain using a combination of nose-to-brain delivery and cell-penetrating peptide-modified nano-micelles. Biomaterials. 2013;34:9220–6.
Kanazawa T, Morisaki K, Suzuki S, Takashima Y. Prolongation of life in rats with malignant glioma by intranasal siRNA/drug codelivery to the brain with cell-penetrating peptide-modified micelles. Mol Pharm. 2014;11:1471–8.
Kanazawa T. Brain delivery of small interfering ribonucleic acid and drugs through intranasal administration with nano-sized polymer micelles. Med Devices (Auckl). 2015;8:57–64.
Chu L, Wang A, Ni L, Yan X, Song Y, Zhao M, et al. Nose-to-brain delivery of temozolomide-loaded PLGA nanoparticles functionalized with anti-EPHA3 for glioblastoma targeting. Drug Deliv. 2018;25:1634–41.
Van Woensel M, Mathivet T, Wauthoz N, Rosière R, Garg AD, Agostinis P, et al. Sensitization of glioblastoma tumor micro-environment to chemo- and immunotherapy by galectin-1 intranasal knock-down strategy. Sci Rep. 2017;7:1217–1217.
Akilo OD, Choonara YE, Strydom AM, du Toit LC, Kumar P, Modi G, et al. AN in vitro evaluation of a carmustine-loaded nano-co-Plex for potential magnetic-targeted intranasal delivery to the brain. Int J Pharm. 2016;500:196–209.
Pandey A, Singh K, Patel S, Singh R, Patel K, Sawant K. Hyaluronic acid tethered pH-responsive alloy-drug nanoconjugates for multimodal therapy of glioblastoma: an intranasal route approach. Mater Sci Eng C. 2019;98:419–36.
Li G, Bonamici N, Dey M, Lesniak MS, Balyasnikova IV. Intranasal delivery of stem cell-based therapies for the treatment of brain malignancies. Expert Opin Drug Deliv. 2018;15:163–72.
Jiang Y, Zhu J, Xu G, Liu X. Intranasal delivery of stem cells to the brain. Expert Opin Drug Deliv. 2011;8:623–32.
Balyasnikova IV, Prasol MS, Ferguson SD, Han Y, Ahmed AU, Gutova M, et al. Intranasal delivery of mesenchymal stem cells significantly extends survival of irradiated mice with experimental brain tumors. Mol Ther. 2014;22:140–8.
Clavreul A, Pourbaghi-Masouleh M, Roger E, Lautram N, Montero-Menei CN, Menei P. Human mesenchymal stromal cells as cellular drug-delivery vectors for glioblastoma therapy: a good deal? J Exp Clin Cancer Res. 2017;36:135–135.
Ahmed AU, Tyler MA, Thaci B, Alexiades NG, Han Y, Ulasov IV, et al. A comparative study of neural and mesenchymal stem cell-based carriers for oncolytic adenovirus in a model of malignant glioma. Mol Pharm. 2011;8:1559–72.
Spencer D, Yu D, Morshed RA, Li G, Pituch KC, Gao DX, et al. Pharmacologic modulation of nasal epithelium augments neural stem cell targeting of glioblastoma. Theranostics. 2019;9:2071–83.
Geletneky K, Hajda J, Angelova AL, Leuchs B, Capper D, Bartsch AJ, et al. Oncolytic H-1 parvovirus shows safety and signs of immunogenic activity in a first phase I/IIa glioblastoma trial. Mol Ther. 2017;25:2620–34.
Kiprianova I, Thomas N, Ayache A, Fischer M, Leuchs B, Klein M, et al. Regression of glioma in rat models by intranasal application of parvovirus H-1. Clin Cancer Res. 2011;17:5333.
Dor-On E, Solomon B. Targeting glioblastoma via intranasal administration of Ff bacteriophages. Front Microbiol. 2015;6:530–530.
Funding
We would like to acknowledge University Grants Commission, India, for providing D.S. Kothari Postdoctoral Fellowship to Sreeranjini Pulakkat and Department of Atomic Energy-Board Of Research In Nuclear Sciences (DAE-BRNS) for providing research fellowship to Prashant G. Upadhaya.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Prashant G. Upadhaya, Sreeranjini Pulakkat, and Vandana B. Patravale declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Upadhaya, P.G., Pulakkat, S. & Patravale, V.B. Nose-to-brain delivery: exploring newer domains for glioblastoma multiforme management. Drug Deliv. and Transl. Res. 10, 1044–1056 (2020). https://doi.org/10.1007/s13346-020-00747-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00747-y