Abstract
Background
Studies investigating bladder cancer risk in pioglitazone-treated type 2 diabetes mellitus patients report conflicting results. Previous meta-analyses on this topic utilized publications prior to 2013. More long-term observational studies have been published since then. We reviewed the accumulated evidence and updated findings from previous meta-analyses.
Methods
This meta-analysis was based on a systematic review of peer-reviewed observational studies published prior to September 30, 2016. Eligible studies were identified using a specified MEDLINE search. References from included studies and from previous meta-analyses were screened for additional records. Meta-analysis hazards ratios were derived using a random-effects model. Several sensitivity analyses including hierarchical Bayesian meta-analysis with country-specific effects were conducted.
Results
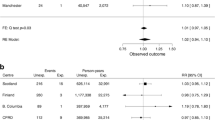
Of 363 identified records, 23 studies were included in this review and 18 in the actual meta-analyses. For bladder cancer outcome, the estimated effect size for ever vs. never use of pioglitazone was 1.16 [95% confidence interval (CI), 1.04–1.28]. In the cumulative dose and duration analyses, highest effect was observed in the highest/longest exposure group, but substantial heterogeneity was present. In the sensitivity analysis, only studies adjusted for lifestyle-related factors were included and the frequentist effect size was 1.18 (95% CI, 1.00–1.40, p = 0.054). However, the risk was not verified in the Bayesian framework with an effect size of 1.17 [95% credible interval (CrI), 0.94–1.54].
Conclusions
In line with previous meta-analyses, we observed a small but statistically significant association between ever (vs. never) use of pioglitazone and bladder cancer risk; however, causality is not established and alternative explanations cannot be ruled out.






Similar content being viewed by others
References
World Health Organization. Global Report on Diabetes. 2016. http://apps.who.int/iris/bitstream/10665/204871/1/9789241565257_eng.pdf?ua=1. Accessed 23 Aug 2016.
Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. http://www.cdc.gov/diabetes/pubs/statsreport14/national-diabetes-report-web.pdf. Accessed 23 Aug 2016.
American Diabetes Association. Economic Costs of Diabetes in the US in 2012. Diabetes Care. 2013;36:1033–46. https://doi.org/10.2337/dc12-2625.
DeFronzo RA, Banerji M, Bray GA, et al. Actos now for the prevention of diabetes (ACT NOW) study. BMC Endocr Disord. 2009. https://doi.org/10.1186/1472-6823-9-17.
DeFronzo RA, Tripathy D, Schwenke DC, et al. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med. 2011;364:1104–15. https://doi.org/10.1056/NEJMoa1010949.
Kipnes MS, Krosnick A, Rendell MS, et al. Pioglitazone hydrochloride in combination with sulfonylurea therapy improves glycemic control in patients with type 2 diabetes mellitus: a randomized, placebo-controlled study. Am J Med. 2001;111:10–7.
Einhorn D, Rendell M, Rosenzweig J, et al. Pioglitazone hydrochloride in combination with metformin in the treatment of type 2 diabetes mellitus: a randomized, placebo-controlled study. The Pioglitazone 027 Study Group. Clin Ther. 2000;22:1395–409.
Rosenstock J, Einhorn D, Hershon K, et al. Efficacy and safety of pioglitazone in type 2 diabetes: a randomised, placebo-controlled study in patients receiving stable insulin therapy. Int J Clin Pract. 2002;56:251–7.
Schernthaner G, Matthews DR, Charbonnel B, et al. Efficacy and safety of pioglitazone versus metformin in patients with type 2 diabetes mellitus: a double-blind, Randomized Trial. J Clin Endocrinol Metab. 2004;89:6068–76. https://doi.org/10.1210/jc.2003-030861.
Takeda Pharmaceuticals America, Inc. Actos (Pioglitazone Hydrochloride) Tablets: Full Prescribing Information. 2009. http://www.actos.com/actospro/home.aspx. Accessed 12 Aug 2016.
Piccinni C, Motola D, Marchesini G, Poluzzi E. Assessing the association of pioglitazone use and bladder cancer through drug adverse event reporting. Diabetes Care. 2011;34(6):1369–71. https://doi.org/10.2337/dc10-2412.
Lewis JD, Ferrara A, Peng T, et al. Risk of bladder cancer among diabetic patients treated with pioglitazone: interim report of a longitudinal cohort study. Diabetes Care. 2011;34:916–22. https://doi.org/10.2337/dc10-1068.
Tseng C-H. Pioglitazone and bladder cancer: a population-based study of Taiwanese. Diabetes Care. 2012;35:278–80. https://doi.org/10.2337/dc11-1449.
Neumann A, Weill A, Ricordeau P, et al. Pioglitazone and risk of bladder cancer among diabetic patients in France: a population-based cohort study. Diabetologia. 2012;55:1953–62. https://doi.org/10.1007/s00125-012-2538-9.
Azoulay L, Yin H, Filion KB, et al. The use of pioglitazone and the risk of bladder cancer in people with type 2 diabetes: nested case-control study. BMJ. 2012;344:e3645.
Fujimoto K, Hamamoto Y, Honjo S, et al. Possible link of pioglitazone with bladder cancer in Japanese patients with type 2 diabetes. Diabetes Res Clin Pract. 2013;99:e21–3. https://doi.org/10.1016/j.diabres.2012.11.013.
Song SO, Kim KJ, Lee B-W, et al. The risk of bladder cancer in korean diabetic subjects treated with pioglitazone. Diabetes Metab J. 2012;36:371–8. https://doi.org/10.4093/dmj.2012.36.5.371.
Colmers IN, Bowker SL, Majumdar SR, et al. Use of thiazolidinediones and the risk of bladder cancer among people with type 2 diabetes: a meta-analysis. CMAJ Can Med Assoc J J Assoc Medicale Can. 2012;184:E675–83. https://doi.org/10.1503/cmaj.112102.
Bosetti C, Rosato V, Buniato D, et al. Cancer risk for patients using thiazolidinediones for type 2 diabetes: a meta-analysis. Oncologist. 2013;18:148–56. https://doi.org/10.1634/theoncologist.2012-0302.
Ferwana M, Firwana B, Hasan R, et al. Pioglitazone and risk of bladder cancer: a meta-analysis of controlled studies. Diabet Med. 2013;30:1026–32. https://doi.org/10.1111/dme.12144.
Turner RM, Kwok CS, Chen-Turner C, et al. Thiazolidinediones and associated risk of bladder cancer: a systematic review and meta-analysis: thiazolidinediones and bladder cancer. Br J Clin Pharmacol. 2014;78:258–73. https://doi.org/10.1111/bcp.12306.
Zhu Z, Shen Z, Lu Y, et al. Increased risk of bladder cancer with pioglitazone therapy in patients with diabetes: a meta-analysis. Diabetes Res Clin Pract. 2012;98:159–63. https://doi.org/10.1016/j.diabres.2012.05.006.
He S, Tang Y, Zhao G, et al. Pioglitazone prescription increases risk of bladder cancer in patients with type 2 diabetes: an updated meta-analysis. Tumour Biol J Int Soc Oncodevelopmental Biol Med. 2014;35:2095–102. https://doi.org/10.1007/s13277-013-1278-x.
Levin D, Bell S, Sund R, et al. Pioglitazone and bladder cancer risk: a multipopulation pooled, cumulative exposure analysis. Diabetologia. 2015;58:493–504. https://doi.org/10.1007/s00125-014-3456-9.
Lewis JD, Habel LA, Quesenberry CP, et al. Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA. 2015;314:265. https://doi.org/10.1001/jama.2015.7996.
Tuccori M, Filion KB, Yin H, et al. Pioglitazone use and risk of bladder cancer: population based cohort study. BMJ. 2016;352:i1541.
Korhonen P, Heintjes EM, Williams R, et al. Pioglitazone use and risk of bladder cancer in patients with type 2 diabetes: retrospective cohort study using datasets from four European countries. BMJ. 2016. https://doi.org/10.1136/bmj.i3903.
von Hippel PT. The heterogeneity statistic I 2 can be biased in small meta-analyses. BMC Med Res Methodol. 2015. https://doi.org/10.1186/s12874-015-0024-z.
Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58. https://doi.org/10.1002/sim.1186.
Higgins JPT. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60. https://doi.org/10.1136/bmj.327.7414.557.
Sterne JA, Egger M. Funnel plots for detecting bias in meta-analysis. J Clin Epidemiol. 2001;54:1046–55. https://doi.org/10.1016/S0895-4356(01)00377-8.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101.
Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Jin S-M, Song SO, Jung CH, et al. Risk of bladder cancer among patients with diabetes treated with a 15 mg pioglitazone dose in Korea: a multi-center retrospective cohort study. J Korean Med Sci. 2014;29:238. https://doi.org/10.3346/jkms.2014.29.2.238.
Hsiao F-Y, Hsieh P-H, Huang W-F, et al. Risk of bladder cancer in diabetic patients treated with rosiglitazone or pioglitazone: a nested case-control study. Drug Saf. 2013;36:643–9. https://doi.org/10.1007/s40264-013-0080-4.
Mackenzie TA, Zaha R, Smith J, et al. Diabetes pharmacotherapies and bladder cancer: a medicare epidemiologic study. Diabetes Ther. 2016;7:61–73. https://doi.org/10.1007/s13300-016-0152-4.
Shapiro S. Causation, bias and confounding: a hitchhiker’s guide to the epidemiological galaxy part 2. Principles of causality in epidemiological research: confounding, effect modification and strength of association. J Fam Plann Reprod Health Care. 2008;34:185–90. https://doi.org/10.1783/147118908784734873.
Greco T, Zangrillo A, Biondi-Zoccai G, et al. Meta-analysis: pitfalls and hints. Heart Lung Vessels. 2013;5:219–25.
Chang C-H, Lin J-W, Wu L-C, et al. Association of thiazolidinediones with liver cancer and colorectal cancer in type 2 diabetes mellitus. Hepatology. 2012;55:1462–72. https://doi.org/10.1002/hep.25509.
Kuo H-W, Tiao M-M, Ho S-C, et al. Pioglitazone use and the risk of bladder cancer. Kaohsiung J Med Sci. 2014;30:94–7. https://doi.org/10.1016/j.kjms.2013.09.011.
Han E, Jang S-Y, Kim G, et al. Rosiglitazone use and the risk of bladder cancer in patients with type 2 diabetes. Medicine (Baltimore). 2016;95:e2786. https://doi.org/10.1097/MD.0000000000002786.
Mamtani R, Haynes K, Bilker WB, et al. Association between longer therapy with thiazolidinediones and risk of bladder cancer: a cohort study. JNCI J Natl Cancer Inst. 2012;104:1411–21. https://doi.org/10.1093/jnci/djs328.
Tseng C-H. Pioglitazone and bladder cancer: a population-based study of Taiwanese. Diabetes Care. 2012;35:278–80. https://doi.org/10.2337/dc11-1449.
Wei L, MacDonald TM, Mackenzie IS. Pioglitazone and bladder cancer: a propensity score matched cohort study. Br J Clin Pharmacol. 2013;75:254–9. https://doi.org/10.1111/j.1365-2125.2012.04325.x.
Vallarino C, Perez A, Fusco G, et al. Comparing pioglitazone to insulin with respect to cancer, cardiovascular and bone fracture endpoints, using propensity score weights. Clin Drug Investig. 2013;33:621–31. https://doi.org/10.1007/s40261-013-0106-9.
Lee M-Y, Hsiao P-J, Yang Y-H, et al. The association of pioglitazone and urinary tract disease in type 2 diabetic Taiwanese: bladder cancer and chronic kidney disease. PLoS One. 2014;9:e85479. https://doi.org/10.1371/journal.pone.0085479.
Balaji V, Seshiah V, Ashtalakshmi G, et al. A retrospective study on finding correlation of pioglitazone and incidences of bladder cancer in the Indian population. Indian J Endocrinol Metab. 2014;18:425. https://doi.org/10.4103/2230-8210.131223.
Lewis JD, Habel L, Quesenberry C, et al. Proteinuria testing among patients with diabetes mellitus is associated with bladder cancer diagnosis: potential for unmeasured confounding in studies of pioglitazone and bladder cancer: proteinuria and bladder cancer diagnosis. Pharmacoepidemiol Drug Saf. 2014;23:636–45. https://doi.org/10.1002/pds.3619.
Acknowledgements
This study was sponsored by Takeda Pharmaceuticals Company Limited.
Author information
Authors and Affiliations
Contributions
DB and YY conceived and designed the study and wrote a first protocol outline. JM, HK, PK, and FH critically reviewed the outline and developed a full protocol. JM, HK, PK, YY, and FH participated in the development of the literature search strategy, conducted literature search, and abstracted and compiled the data. JM, HK, PK, and FH performed the analyses. All authors participated in the discussion and interpretation of the study findings. JM, HK, PK, and FH drafted the manuscript. All authors critically reviewed, revised, and approved the final version submitted for publication.
Corresponding author
Ethics declarations
Conflict of interest
YY and DB are employees of Takeda Pharmaceutical Company Limited. JM, HK, FH, and PK are employees of EPID Research Oy, which is a contract research organization that performs commissioned pharmacoepidemiologic studies, and thus its employees have been and currently are working in collaboration with several pharmaceutical companies.
Ethics approval
This systematic literature review does not involve any animal or human subjects, including research on identifiable human material and data.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Mehtälä, J., Khanfir, H., Bennett, D. et al. Pioglitazone use and risk of bladder cancer: a systematic literature review and meta-analysis of observational studies. Diabetol Int 10, 24–36 (2019). https://doi.org/10.1007/s13340-018-0360-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-018-0360-4