Abstract
Aims
We investigated the mechanism underlying body weight (BW) gain after pioglitazone administration in type 2 diabetes in relation to gender differences in abdominal fat distribution and congestive factors.
Materials and methods
Seventy-five patients without insulin treatment took 15 or 30 mg pioglitazone plus their current medication; abdominal visceral (V) and subcutaneous (S) fat tissues and clinical parameters were compared before and after the 24-week intervention.
Results
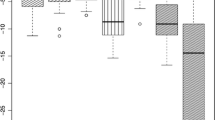
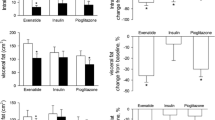
Eleven patients withdrew. Of the remaining 64 patients, hemoglobin A1c improved in 52, indicating that most were pioglitazone responders for glycemic control. Mean BW increased from 64.0 to 66.0 kg. There were gender differences in BW gain. In women, V decreased and S increased; in men, V did not decrease and S markedly increased. Atrial natriuretic peptide increased more significantly than brain natriuretic peptide in women. This suggested that pioglitazone induced a V-to-S abdominal fat distribution shift only in women. In men, the main weight gain mechanism was believed to be an S increase. Adiponectin increase was observed in both groups; however, high-sensitive C-reactive protein decreased only in women. Triglyceride and free fatty acid decreased significantly only in women.
Conclusion
A pioglitazone-induced V-to-S distribution shift of tissue adipocytes occurred only in women. Pioglitazone may be more beneficial for atherosclerotic and metabolic factors in women despite the smaller dose administered. The pioglitazone dose should be carefully considered in women, whereas the beneficial effects of pioglitazone may be limited in men.
Similar content being viewed by others
References
Mazzone T, Meyer PM, Feinstein SB, Davidson MH, Kondos GT, D’Agostino RB Sr, Perez A, Provost JC, Haffner SM. Effect of pioglitazone compared with glimepiride on carotid intima-media thickness in type 2 diabetes: a randomized trial. JAMA. 2006;296:2572–81.
Dormandy JA, Charbonnel B, Eckland DJ, Erdmann E, Massi-Benedetti M, Moules IK, Skene AM, Tan MH, Lefèbvre PJ, Murray GD, Standl E, Wilcox RG, Wilhelmsen L, Betteridge J, Birkeland K, Golay A, Heine RJ, Korányi L, Laakso M, Mokán M, Norkus A, Pirags V, Podar T, Scheen A, Scherbaum W, Schernthaner G, Schmitz O, Skrha J, Smith U, Taton J. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study: a randomized controlled trial. Lancet. 2008;366:1279–89.
Wilcox R, Bousser MG, Betteridge DJ, Schernthaner G, Pirags V, Kupfer S, Dormandy J. Effects of pioglitazone in patients with type 2 diabetes with or without previous stroke. Stroke. 2007;38:865–73.
Erdmann E, Dormandy JA, Charbonnel B, Massi-Benedetti M, Moules IK, Skene AM. The effect of pioglitazone on recurrent myocardial infarction in 2,445 patients with type 2 diabetes and previous myocardial infarction. J Am Col Cardiol. 2007;49:1772–80.
Kawamori R, Kadowaki T, Onji M, Seino Y, Akanuma Y. Hepatic safety profile and glycemic control of pioglitazone in more than 20,000 patients with type 2 diabetes mellitus: postmarketing surveillance study in Japan. Diab Res Clin Pract. 2007;76:229–35.
Nissen SE, Nicholls SJ, Wolski K, Nesto R, Kupfer S, Perez A, Jure H, De Larochellière R, Staniloae CS, Mavromatis K, Saw J, Hu B, Lincoff AM, Tuzcu EM, PERISCOPE intervention. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis on patients with type 2 diabetes: the PERISCOPE randomized controlled trial. JAMA. 2008;299:1561–73.
Miyazaki Y, Mahankali A, Matsuda M, Mahankali S, Hardies J, Cusi K, Mandarino LJ, DeFronzo RA. Effect of pioglitazone on abdominal fat distribution and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab. 2002;87:2784–91.
Kadowaki K, Fukino K, Negishi E, Ueno K. Sex differences in PPAR gamma expressions in rat adipose tissues. Biol Pharm Bull. 2007;30:818–20.
Ueno K, Sugai H, Sato H. Mechanism of sex differences and the clinical significance of PPARγ targeted drugs. Jap J Clin Med. 2010;68:224–8.
Igarashi M, Jimbu Y, Hirata A, Tominaga M. Characterization of plasma brain natriuretic peptide level in patients with type 2 diabetes. Endocr J. 2005;52:353–62.
Makino N, Sugano M, Satoh S, Oyama J, Maeda T. Peroxisome proliferator-activated receptor-gamma ligands attenuate brain natriuretic peptide production and affect remodeling in cardiac fibroblasts in reoxygenation after hypoxia. Cell Biochem Biophys. 2006;44:65–71.
Tokunaga K, Matsuzawa Y, Ishikawa K, Tarui S. Novel technique for determination of body fat by computed tomography. Int J Obes. 1983;7:437–45.
Tsuchiya K, Akaza I, Yoshimoto T, Hirata Y. Pioglitazone improves endothelial function with increased adiponectin and high-density lipoprotein cholesterol levels in type 2 diabetes. Endocr J. 2009;56:691–8.
Aso Y, Hara K, Ozeki N, Yatsuka C, Nakano T, Matsumoto S, Suetsugu M, Nakamachi T, Takebayashi K, Haruki K, Inukai T. Low-dose pioglitazone increases serum high molecular weight adiponectin and improves glycemic control in Japanese patients with poorly controlled type 2 diabetes. Diabetes Res Clin Pract. 2009;85:147–52.
Lefebvre AM, Laville M, Vega N, Riou JP, Gaal L, Auwerx J, Vidal H. Depot-specific differences in adipose tissue gene expression in lean and obese subjects. Diabetes. 1998;47:98–103.
Yasue H, Obata K, Okumura K, Kurose M, Ogawa H, Matsuyama K, Jougasaki M, Saito Y, Nakao K, Imura H. Increased secretion of atrial natriuretic polypeptide from the left ventricle in patients with dilated cardiomyopathy. J Clin Invest. 1989;83:46–51.
Arnetz L, Dorkhan M, Alvarsson M, Brismar K, Ekberg NR: Gender differences in non-glycemic responses to improved insulin sensitivity by pioglitazone treatment in patients with type 2 diabetes. Acta Dibetol. 2013. doi:10.1007/s00592-013-0457-y
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Taneda, S., Bando, H., Misawa, K. et al. Prospective study (Manda study 1) on the clinical effects of pioglitazone in relation to gender differences in Japanese patients with type 2 diabetes. Diabetol Int 5, 92–97 (2014). https://doi.org/10.1007/s13340-013-0136-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-013-0136-9