Abstract
Anti-cancer therapy based on the repeated administration of oxaliplatin is limited by the development of a disabling neuropathic syndrome with detrimental effects on the patient’s quality of life. The lack of effective pharmacological approaches calls for the identification of innovative therapeutic strategies based on new targets. We focused our attention on the imidazoline I1 receptor (I1-R) and in particular on the selective I1-R agonist 2-(1-([1,1′-biphenyl]-2-yl)propan-2-yl)-4,5-dihydro-1H-imidazole) (carbophenyline). The purpose of this work was the preclinical evaluation of the efficacy of carbophenyline on oxaliplatin-induced neuropathic pain in mice. Carbophenyline, acutely per os administered (0.1–10 mg kg−1), induced a dose-dependent anti-hyperalgesic effect that was completely blocked by the pre-treatment with the I1-R antagonist 3 or the I1/α2 receptor antagonist efaroxan, confirming the I1-R-dependent mechanism. Conversely, pre-treatment with the I2-R antagonist BU224 did not block the anti-nociceptive effect evoked by carbophenyline. Repeated oral administrations of carbophenyline (1 mg kg−1) for 14 days, starting from the first day of oxaliplatin injection, counteracted the development of neuropathic pain in all behavioral tests (cold plate, Von Frey, and paw pressure tests) carried out 24 h after the last carbophenyline treatment on days 7 and 14. In the dorsal horn of the spinal cord, carbophenyline significantly decreased the oxaliplatin-induced astrocyte activation detected by immunofluorescence staining by the specific labelling with GFAP antibody. In conclusion, carbophenyline showed anti-neuropathic properties both after acute and chronic treatment with preventive effect against oxaliplatin-induced astrocyte activation in the spinal cord. Therefore, I1-R agonists emerge as a new class of candidates for the management of oxaliplatin-induced neuropathic pain.
Similar content being viewed by others
Introduction
Neuropathic pain is now defined by the International Association for the Study of Pain (IASP) as “pain caused by a lesion or disease of the somatosensory nervous system” [1]. Among different factors, nervous lesions can be induced by neurotoxic drugs like several anticancer agents able to evoke painful syndromes defined chemotherapy-induced neuropathies. Oxaliplatin, one of the lead treatments for colorectal cancers [2, 3], is associated with the development of a severe persistent neuropathic pain which remains the dose-limiting toxicity of this treatment [4]. Chronologically, oxaliplatin-induced acute and transient hyperexcitability followed by a chronic cumulative neuropathy experienced by almost the 50% of patients [5]. Signs and symptoms of chronic neuropathy include pain, paresthesia, hypoesthesia, and changes in proprioception that persist during cycles [6]. In the long term, this syndrome has a deleterious impact on cancer survivors, often being associated with sleep disturbance, depressive symptoms, and impaired health-related quality of life [7]. Several molecular mechanisms participate to oxaliplatin neurotoxicity involving early targets in the peripheral nervous system as well as direct and indirect alterations of the neuronal and glial compartments of the central nervous system [8,9,10,11,12,13].
No treatments are currently available to reduce oxaliplatin-dependent neuropathic pain, further pain-relieving drugs used against neuropathic pain are not satisfactory due to limited efficacy and side effects [14, 15]. For these reasons, efforts are necessary to explore novel molecular targets for the pharmacological management of neuropathic pain.
Recent studies have highlighted that the imidazoline I2 receptor (I2-R) activation by selective ligands such as 2-BFI [16] or phenyzoline (1) (Fig. 1) [17], alone or in combination with opioids, triggers a significant anti-hyperalgesic effect. Therefore, this receptor system has been suggested as a promising target for the management of chronic pain [18,19,20]. The I2-R is a member of a wider family of imidazoline receptors, also including I1-R and I3-R, which play important roles in central cardiovascular regulation and in insulin secretion, respectively [21, 22]. In particular, I1-R is involved in the hypotensive activity of clonidine and related compounds supporting the idea that the I1-Rs are upstream from the α2-adrenergic receptors and work in tandem for its effect on blood pressure [23]. Additionally, I1-R activation may suppress sympathetic outflow at postganglionic sympathetic neurons through modulation of several signal transduction systems including N-type calcium channels, G protein inwardly rectifying potassium channel, adenosine receptors, phosphatidyl-choline-specific phospholipase C, and nicotinic receptors [24,25,26,27,28,29]. On this basis, a possible role of I1-R in pain modulation can be theoretically suggested although so far unexplored. In the present work we studied the effects of I1-R modulation against oxaliplatin-induced neuropathic pain by using the selective I1-R agonist compound 2 (in this paper named carbophenyline, highly selective over I2, α2, and 5-HT1A receptors [30, 31]) (Fig. 1). Its effects on pain threshold were evaluated after single or repeated administrations in the absence or presence of the I1-R antagonist 3 [2-(1-methyl-2-phenylethyl)-4,5-dihydro-1H-imidazole] (Fig. 1) [32], highly selective over I2, α2 and 5-HT1A receptor and the well-known I1/α2 receptor antagonist efaroxan [33] which proved to be selective over several other biological targets [33; https://pubchem.ncbi.nlm.nih.gov/compound/Efaroxan#section=BioAssay-Results]. Moreover, the I2 antagonist BU224 was also used to exclude the involvement of I2-R [34]. Finally, to analyze the impact of I1-R stimulation on the maladaptive plasticity of the neuropathic central nervous system, the effect of carbophenyline on oxaliplatin-induced astrocyte activation in the dorsal horn of the spinal cord was assessed.
Materials and Methods
Drugs
The I1-R agonist 2-(1-([1,1′-biphenyl]-2-yl)propan-2-yl)-4,5-dihydro-1H-imidazole) (2, carbophenyline) and the I1-R antagonist 2-(1-phenylpropan-2-yl)-4,5-dihydro-1H-imidazole (3) were obtained treating methyl 3-([1,1′-biphenyl]-2-yl)-2-methylpropanoate and methyl 2-methyl-3-phenylpropanoate, respectively, with ethylenediamine and trimethyl aluminum at 110 °C in toluene [30, 32]. Efaroxan hydrochloride and BU224 hydrochloride were purchased by Tocris. Oxaliplatin and pregabalin were purchased by Carbosynth (Compton, UK).
Cell Cultures
The HT-29 human colon cancer cell line were acquired from the American Type Culture Collection. HT-29 were cultured in Complete Medium (CM) composed by high-glucose DMEM with 10% FBS, 2 mM l-glutamine, 100 IU/mL penicillin, and 100 μg mL−1 streptomycin (Sigma-Aldrich, Italy).
Cell Viability Assay
To plate HT-29 cells (1 × 104 cells/well), 96-well cell culture plates (Corning) were used. After 48 h, the experiments were performed. Cells were incubated in serum-free DMEM with 0.3–100 μM oxaliplatin (Carbosynth, Compton, Berkshire, UK) in the absence or presence of carbophenyline 10 μM for 48 h. Cell viability was evaluated by the reduction of 3-(4,5-dimethylthiozol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich, Italy) as an index of mitochondrial compartment functionality. After 48 h incubation, 1 mg mL-1 MTT in serum-free DMEM without phenol red was added into each well and incubated for 30 min at 37 °C. After washing the formazan crystals were dissolved in 150 μL dimethyl sulfoxide. The absorbance was measured at 550 nm. Experiments were performed in triplicate.
Animals
CD-1 mice (Envigo, Varese, Italy) weighing 20–25 g at the beginning of the experimental procedure were used. Animals were housed in the Centro Stabulazione Animali da Laboratorio (University of Florence) and used at least 1 week after their arrival. Ten mice were housed per cage (size 26 cm × 41 cm); animals were fed a standard laboratory diet and tap water ad libitum and kept at 23 ± 1 °C with a 12 h light/dark cycle (light at 7 A.M.). All animal manipulations were carried out according to the Directive 2010/63/EU of the European parliament and of the European Union council (22 September 2010) on the protection of animals used for scientific purposes. The ethical policy of the University of Florence complies with the Guide for the Care and Use of Laboratory Animals of the US National Institutes of Health (NIH Publication No. 85–23, revised 1996; University of Florence assurance number: A5278–01). Formal approval to conduct the experiments described was obtained from the Italian Ministry of Health (No. 54/2014-B) and from the Animal Subjects Review Board of the University of Florence. Experiments involving animals have been reported according to ARRIVE guidelines [35]. All efforts were made to minimize animal suffering and to reduce the number of animals used.
Oxaliplatin-Induced Neuropathic Pain
2.4 mg kg−1 oxaliplatin was dissolved in 5% glucose solution and administered intraperitoneally (i.p.) for 5 consecutive days every week for 2 weeks (10 i.p. injections) [36 with minor modification, 37]. Control animals received an equivalent volume of 5% glucose i.p. (vehicle).
Compound Administration and Study of the Pharmacodynamic Mechanisms
When neuropathic pain was fully established (starting from day 14), carbophenyline (0.1–10 mg kg−1) was suspended in 1% solution of carboxymethylcellulose sodium salt (CMC) and per os (p.o.) acutely administered ranging from 0.1 mg kg−1 to 10 mg kg−1 to evaluate its symptomatic efficacy. Pregabalin (30 mg kg−1, p.o.) was used as reference drug [38]. Afterwards, to highlight a preventive effect, repeated per os administrations of 1 mg kg−1 of carbophenyline were carried out daily from the beginning of oxaliplatin administration (day 1) to the end of the experiment (day 14). Control animals were treated with vehicles (5% glucose and CMC 1%).
For the study of the pharmacodynamic mechanisms, the selective I1-R antagonist 3, I1/α2 receptor antagonist efaroxan and selective I2-R antagonist BU224 were used. All antagonists were suspended in CMC 1% and p.o. administered 15 min before carbophenyline. For 3 and BU224 antagonists we used the first dose non hyperalgesic as shown in Supplementary table 1. Control animal were treated with vehicles (5% glucose and CMC 1%).
Paw Pressure Test
Mechanical hyperalgesia was determined by measuring the latency in seconds to withdraw the paw away from a constant mechanical pressure exerted onto the dorsal surface [39]. A 15 g calibrated glass cylindrical rod (diameter = 10 mm) chamfered to a conical point (diameter = 3 mm) was used to exert the mechanical force. The weight was suspended vertically between two rings attached to a stand and was free to move vertically. A single measure was made per animal. A cutoff time of 40 s was used.
Cold Plate
Thermal allodynia was assessed using the cold plate test. With minimal animal–handler interaction, mice were taken from home-cages, and placed onto the surface of the cold plate (Ugo Basile, Varese, Italy) maintained at a constant temperature of 4 °C ± 1 °C. Ambulation was restricted by a cylindrical Plexiglas chamber (diameter, 10 cm; height, 15 cm), with open top. A timer controlled by foot peddle began timing response latency from the moment the mouse was placed onto the cold plate. Pain-related behavior (licking of the hind paw) was observed, and the time (seconds) of the first sign was recorded. The cutoff time of the latency of paw lifting or licking was set at 30 s [40].
Von Frey
The animals were placed in 20 × 20 cm Plexiglas boxes equipped with a metallic meshy floor, 20 cm above the bench. A habituation of 15 min was allowed before the test. An electronic Von Frey hair unit (Ugo Basile, Varese, Italy) was used: the withdrawal threshold was evaluated by applying force ranging from 0 to 5 g with 0.2 g accuracy. Punctuate stimulus was delivered to the mid-plantar area of each anterior paw from below the meshy floor through a plastic tip and the withdrawal threshold was automatically displayed on the screen. Paw sensitivity threshold was defined as the minimum pressure required to elicit a robust and immediate withdrawal reflex of the paw. Voluntary movements associated with locomotion were not taken as a withdrawal response. Stimuli were applied on each anterior paw with an interval of 5 s. The measure was repeated 5 times and the final value was obtained by averaging the 5 measures [41, 42].
Immunohistochemistry
On day 14, after behavioral measurements, mice were sacrificed and the lumbar spinal cord segments were removed, postfixed in 4% paraformaldehyde, and then cryoprotected in 30% sucrose solution at 4 °C. Slide-mounted cryostat sections (5 μm) were processed for indirect immunofluorescence histochemistry.
Formalin-fixed cryostat sections (5 μm) were incubated for 30 min in ready-to-go blocking solution (Bio-Optica, Milan, Italy) at room temperature to block unspecific binding. The primary antibodies, overnight incubated at 4 °C, were directed against Iba1 (rabbit, 1:200; Wako Chemicals, Richmond, USA) for microglial staining or against glial fibrillary acidic protein (GFAP; rabbit, 1:200; Dako, USA) for astrocyte staining. After rinsing in PBST, sections were incubated in donkey anti-rabbit IgG secondary antibody labelled with Alexa Fluor 488 (1:500, Invitrogen, Milan, Italy) at room temperature for 1 h. Nuclei were stained with DAPI (4′,6-diamidin-2-fenilindolo; 1:2000; Invitrogen, Milan, Italy).
Negative control sections (no exposure to the primary antisera) were processed concurrently with the other sections for all immunohistochemical studies. We obtained a single optical density value for the dorsal horns by averaging the two sides in each mouse, and these values were compared to the homologous average values from the vehicle-treated animals.
Images were acquired by a motorized Leica DM6000B microscope equipped with a DFC350FX camera (Leica, Mannheim, Germany). Microglia and astrocyte morphology were assessed by inspection of at least three fields (× 40 0.75NA objective) in the dorsal horn.
Quantitative analysis of GFAP and Iba1-positive cells was performed by collecting at least three independent fields through a × 20 0.5NA objective. Immunofluorescent staining was measured as mean fluorescence intensity using ImageJ software (ImageJ, National Institute of Health, USA, https://imagej.nih.gov/) by automatic thresholding algorithm. Furthermore, GFAP or Iba-1 -positive cells were quantified by thresholding automatic count. Results, given as mean fluorescent intensity (arbitrary units) by the thresholded fluorescent signal, revealed a common trend between GFAP/Iba-1 expression and astrocyte/microglial cell number (data not shown). Five spinal cord sections were analyzed for each animal.
Statistical Analysis
Behavioral measurements were performed on ten mice for each treatment carried out in 2 different experimental sets. All assessments were made by researchers blinded to animal treatments. Results were expressed as mean ± (S.E.M.) with one-way analysis of variance. A Bonferroni’s significant difference procedure was used as a post hoc comparison. P values < 0.05 or < 0.01 were considered significant. Data were analyzed using the Origin 9 software (OriginLab, Northampton, MA, USA).
Results
Behavioral Evaluations
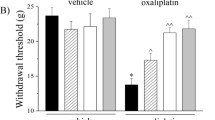
The effects of carbophenyline against neuropathic pain were studied in a mouse model of chemotherapy-induced neuropathic pain. The neurotoxicity of oxaliplatin was used to evoke a neuropathic syndrome characterized by thermal and mechanical hypersensitivity and allodynia after repeated administrations (2.4 mg kg−1, 10 intraperitoneal injections) (Fig. 2). On day 14, the response to a cold non-noxious stimulus (allodynia-like symptom) was measured by the cold plate test. The pain threshold of oxaliplatin-treated animals decreased to 9.8 ± 0.2 s in comparison to 18.2 ± 0.4 s of the control group (vehicle + vehicle-treated animals). Increasing doses of carbophenyline (0.1–10 mg kg−1), acutely per os administered, reduced thermal allodynia in a dose-dependent manner. The single administration of compound at 10 mg kg−1 was able to fully counteract oxaliplatin-induced neuropathic pain 30 min after injection with the onset 15 min after treatment that lasted up to 45 min. A dose of 1 mg kg−1 increased the pain threshold of the animals starting 15 min after treatment with a peak at 45 min. The anti-hypersensitivity effect vanished at 60 min. The lowest dose (0.1 mg kg−1) was inactive. Pregabalin was used as reference drug. The administration of the drug at the dose of 30 mg kg−1 significantly increased the pain threshold 30 min after treatment with the effect that remained active up 45 min without completely counteract oxaliplatin-induced thermal allodynia (Fig. 2a). The acute anti-hyperalgesic and anti-allodynic effects of carbophenyline were also evaluated by paw pressure test and Von Frey test, respectively (Fig. 2 b and c). Results obtained were comparable to that achieved by the cold plate test. Carbophenyline evoked a dose-dependent anti-neuropathic effect that reached the higher effect with the dose of 10 mg kg−1, 30 min after the administration.
Effect of acute carbophenyline administrations on pain behavior induced by oxaliplatin. Pain threshold to (a) a non-noxious thermal stimulus (cold plate test), (b) to a noxious mechanical stimulus (paw pressure test) and (c) to a non-noxious mechanical stimulus (Von Frey test) were measured. Oxaliplatin (2.4 mg kg−1, i.p.) was administered for 2 weeks (10 injections). Starting from day 14, carbophenyline was acutely per os administered (0.1–10 mg kg−1) and measurements assessed before and 15, 30, 45, 60 min after injection. Control animals were treated with vehicles. Each value represents the mean ± S.E.M. of 10 animals per group performed in 2 experimental sets. Statistical analysis is one-way ANOVA followed by Bonferroni’s post hoc comparison. ^^P < 0.01 vs vehicle + vehicle; **P < 0.01 vs oxaliplatin + vehicle
To confirm the pharmacodynamics of carbophenyline, three specific antagonists were used: 3 (selective I1-R antagonist), efaroxan (I/α2 receptor antagonist), and BU224 (selective I2-R antagonist). As shown in Fig. 3, the pre-treatment (15 min before carbophenyline administration) with 3 10 (mg kg−1, p.o.) was able to completely abolish the pain-relieving effect of carbophenyline till the end of the experiment. The same result was obtained with the pre-treatment with efaroxan (10 mg kg−1, p.o., 15 min before carbophenyline administration). BU224 (3 mg kg−1 p.o., 15 min before carbophenyline administration) was not able to modify the efficacy of carbophenyline over the time of observation (Fig. 3).
Study of carbophenyline pharmacodynamic profile. Pain was induced by repeated treatment with oxaliplatin. The hypersensitivity to a cold stimulus was measured by the cold plate test. Carbophenyline was administered per os at 10 mg kg−1. The I1 receptor antagonist 3 (10 mg kg−1), the I1/α2 receptor antagonist efaroxan (10 mg kg−1) and the I2 receptor antagonist BU224 (3 mg kg−1) were p.o. administered 15 min before carbophenyline; measurements were assessed before and 15, 30, 45, 60 min after carbophenyline injection. Control animals were treated with vehicles. Each value represents the mean ± S.E.M. of 10 animals per group performed in 2 experimental sets. Statistical analysis is one-way ANOVA followed by Bonferroni’s post hoc comparison. ^^P < 0.01 vs vehicle + vehicle; **P < 0.01 vs oxaliplatin + vehicle; °°P < 0.01 vs oxaliplatin + carbophenyline
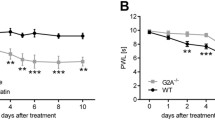
Repeated treatments of carbophenyline (1 mg kg−1 p.o.) were performed to evaluate the preventive effect of the compound in the same model of oxaliplatin-induced neuropathic pain. Oxaliplatin-treated animals were daily injected with carbophenyline starting from the same day of antitumor administration. The response to a thermal (cold plate test) and mechanical (paw pressure and Von Frey tests) stimuli was measured on days 7 and 14, 24 h after the last treatment (Fig. 4). Oxaliplatin-induced neuropathic pain increased during time reaching the plateau on week 2 (day 14) as shown by Fig. 4. Repeated daily administration of carbophenyline increased the pain threshold of oxaliplatin-treated animals at all time points without development of tolerance to the anti-hypersensitivity effect. The effect evoked by carbophenyline remained stable on days 7 and 14 as highlighted by the cold plate and paw pressure tests (Fig. 4 a and b, respectively) counteracting oxaliplatin-induced neuropathic pain. Similar results were obtained measuring the response to a mechanical non-noxious stimulus (Von Frey test, Fig. 4c), carbophenyline increased its efficacy during time completely abolishing mechanical allodynia on day 14.
Effects of repeated administration of carbophenyline against pain induced by oxaliplatin. Pain threshold to a non-noxious thermal stimulus as measured by the cold plate test (a). Sensitivity to a noxious mechanical stimulus as measured by the paw pressure test (b). Pain threshold to a non-noxious mechanical stimulus as measured by the Von Frey test (c). Behavioral tests were performed on days 7 and 14 after the beginning of oxaliplatin and carbophenyline administrations, 24 h after the last treatment. Oxaliplatin (2.4 mg kg−1, i.p.) was administered for 2 weeks (10 injections) whereas carbophenyline (1 mg kg−1, p.o.) was daily administered, starting from day 1 of oxaliplatin injection. Control animals were treated with vehicles. Each value represents the mean ± S.E.M. of 10 animals per group performed in 2 experimental sets. Statistical analysis is one-way ANOVA followed by Bonferroni’s post hoc comparison. ^^P < 0.01 vs vehicle + vehicle; **P < 0.01 vs oxaliplatin + vehicle
Analysis of Glial Cells in the Spinal Cord
The central nervous system was analyzed to assess the capability of carbophenyline to intervene against the maladaptive plasticity induced by oxaliplatin treatment. In the spinal cord, repeated oxaliplatin injections induced astrocyte activation, measured as increased GFAP fluorescence intensity (day 14) on the entire surface and, particularly, in the superficial laminae (Fig. 5b) with respect to the control animals (Fig. 5a). The repeated administration of carbophenyline significantly prevented the astrocytic response to oxaliplatin as shown in Fig. 5c. As previously reported [9], spinal microglia was not altered by oxaliplatin on day 14 (Supplementary Figure S1); the co-treatment with carbophenyline did not modify this condition (Fig. S1).
Astrocytic profile in the spinal cord. The effect of repeated treatment with carbophenyline (1 mg kg−1) was evaluated in oxaliplatin-treated mice on day 14. The number of GFAP-positive cells was measured in the dorsal horn of the L4–L5 spinal cord. Transverse sections of spinal cord imaged with × 20 objective (scale bar = 50 μm); insert shows morphological characteristics of a representative astrocytic cell. Histograms show the quantitative analysis of GFAP fluorescence intensity. Each value represents the mean of 6 mice, performed by analyzing 4 independent fields for both sides of the dorsal horn of the lumbar spinal cord. Statistical analysis is one-way ANOVA followed by Bonferroni’s post hoc comparison. ^^P < 0.05 vs vehicle + vehicle; **P < 0.01 vs oxaliplatin + vehicle
HT-29 Cell Line
Aimed to evaluate a possible interaction between the treatment with carbophenyline and the therapeutic properties of oxaliplatin, we analyzed the vitality of the human colorectal cancer cell line HT-29. Table 1 shows oxaliplatin lethality after 48 h incubation (0.3–100 μM) in the absence or presence of carbophenyline. Ten μM carbophenyline did not significantly alter the oxaliplatin-dependent decrease of cell viability. Carbophenyline per se did not influence HT-29 viability up to 100 μM as shown in Table 2.
Discussion
The present data show for the first time the involvement of the I1-R in the signaling of neuropathic pain, highlighting the properties of the selective I1-R agonist carbophenyline in relieving pain alterations in a mouse model of oxaliplatin-induced neurotoxicity. Moreover, repeated administration of carbophenyline prevented the maladaptive changes that occur to the astrocyte population in the central nervous system. A new class of potential neuropathic pain relievers has been outlined.
Many different pharmacological actions have been associated with imidazoline compounds [21] and our research on them has highlighted that the 2-substituted imidazoline ring linked to an aromatic moiety (Ar) by a biatomic bridge (X) is a bioversatile scaffold to obtain agents interacting with different targets [43,44,45,46] (Fig. 1). Some of these imidazoline ligands demonstrated to be efficacious as anti-neuropathic agents. In particular, in addition to the already mentioned I2 ligand phenyzoline (1), characterized by a –CH2CH2- bridge, the dual α2/5-HT1A receptor agonist S-(-)-biphenyline (4) [47, 48], bearing an –OCH(CH3)- bridge and an ortho phenyl substituent in the aromatic ring, showed a significant pain-relieving effect in a rat model of neuropathic pain [49]. The isosteric substitution of the oxygen atom of the bridge with a CH2 group produced derivative carbophenyline, characterized by an interesting modulation of the biological profile. Indeed, carbophenyline behaves as an I1-R agonist with high selectivity over I2, α2, and 5-HT1A receptors [30, 31, 48]. The profitable activation of I1-R makes carbophenyline a potent anti-hypertensive agent [30, 31].
Although imidazoline binding sites attracted attention in nociception, only the I2-R was studied. A possible role of I1-R can be hypothesized analyzing the efficacy of clonidine (a molecule possessing I1-R affinity in addition to the well-known adrenergic α2-mediated effects) against neuropathic and cancer pain [50,51,52,53], even if a possible I1-R component in clonidine anti-nociceptive effects was not described. Stone et al. [54] showed that there is a possible synergy between I1-R and opioid receptors at the spinal level based on the use of the selective I1-R agonist, diethyl-phenyl-amino-imidazoline, ST91. Exploring the possible anatomical and molecular impact of I1-R in the nervous system for justifying the pain-relieving effect, some interesting data emerge. I1 binding sites were found in bovine, rabbit, rat, and human brainstem and brain [55]. In the brainstem, I1-Rs were detected predominantly on non-mitochondrial membranes [56]; in the brain, they are particularly expressed by synaptic membranes, and most likely from presynaptic terminals [55]. Among molecular mechanisms mediated by I1-R, Edwards and Ernsberger [57] showed that the receptor activation can abolish the nerve growth factor–activated signalling pathway by increasing the levels of a specific phosphatase through ERK dephosphorylation. This effect could be particularly relevant in pain modulation since the nerve growth factor (as others growth factors) is a potent pain inducer [58] that seems unable to separate the neuroprotective effect from the algic one probably following the evolutionary positive alarm role of physiological pain. Moreover, moxonidine, an anti-hypertensive drug, inhibited voltage-dependent N-type Ca2+ current in rat ganglion neurons via activating I1-R attenuating repetitive firing [29]. Neuronal N-type Ca2+ current is closely related to nociception and the pivotal role of its modulation in the pharmacodynamic of anti-neuropathic drugs was recently described by our group [59]. On the contrary, other authors correlated the I1-R activation in the striatum with pro-excitatory signals [60] that cannot fit with pain relief indicating that the nervous I1-R signaling is complex and dependent on the anatomical localization.
Furthermore, a few data indicate a I1-R role in neuroprotection. The stimulation of this receptor participates in the improvement of locomotor recovery in mice subjected to spinal cord injury treated with agmatine [61]. Treatment with moxonidine significantly attenuated locomotor activity, grip strength, learning, and memory impairments in a model of Huntington’s disease [62].
Considering the wide expression of I1-R in several areas of the CNS [55] and that no studies reported on the anti-neuropathic efficacy of selective I1-R ligands, we thought it interesting to investigate the role played by I1-R in the oxaliplatin-induced neuropathic pain using the selective I1-R agonist carbophenyline. The selection of carbophenyline is rationally justified on the basis of its structural analogy with other anti-neuropathic imidazoline compounds at our disposal, in particular derivative 4 showing a significant pain-relieving effect in a rat model of neuropathic pain [49].
In vivo experiments demonstrated the symptomatic properties of carbophenyline in relieving oxaliplatin-induced allodynia and hyperalgesia after a single acute injection. The relevance of the I1-R in the pain-relieving pharmacodynamic of carbophenyline has been validated through the use of 3, efaroxan, and BU224 (I1-, I1/α2-, and I2-R antagonists, respectively) demonstrating the selective involvement of type I1-R. Moreover, carbophenyline prevented the development of oxaliplatin-induced neuropathic pain after a repeated administration, and the effect remained stable both 7 and 14 days over treatment. The described preventive profile allows us to exclude the development of tolerance to the anti-hypersensitivity effects. Further, it suggests possible neuroprotective properties of carbophenyline able to modify the nervous system alterations leading the establishment and persistence of neuropathic pain.
This aspect was studied in the present research analyzing the possible impact of I1-R stimulation on spinal cord alterations evoked by oxaliplatin. The repeated treatment with carbophenyline prevented the activation of astrocytes in the dorsal horns. Glia cells contribute to the persistence of pain, an effect that in neuropathic conditions prevails on the usual homeostatic functions [63]. The metabolic activation of microglia and astrocytes participates in the maladaptive plasticity of the central nervous system, facilitating nociceptive processes, generating clinical pain hypersensitivity, and making neuropathic pain an autonomous disease state [64]. Despite the limited ability of oxaliplatin to cross the blood–brain barrier [65], in our previous study we demonstrated that microglia and astrocytes cooperated to promote the initial phase of spinal cord sensitization but, although microglia conclude the task in the early phase of oxaliplatin administration, astrocytes maintain their activation contributing to the pain persistence [10]. These data are supported by the information that the spinal intrathecal infusion of fluorocitrate, an astrocyte selective inhibitor, fully prevented oxaliplatin-evoked pain alterations [10]. The present results revealed the modulatory role of carbophenyline on oxaliplatin-induced spinal astrocyte activation suggesting its ability to protect from the maladaptive plasticity of the central nervous system, a mechanism that could offer an explanation for the prevention of pain development demonstrated after a repeated treatment. The present first demonstration regarding the potential of I1-R agonists in pain relief adds a fragment to the very limited information regarding the role of this receptor in nervous signals.
A crucial point in the study of novel treatments against chemotherapy-induced neuropathic pain is the evaluation of possible interactions with the anticancer effect of the drug as the primary treatment outcome. As a preliminary evaluation we analyzed the effect of carbophenyline on oxaliplatin lethality against the human colon adenocarcinoma cell line HT-29, representing a main clinical target for this platin derivative [2, 3]. No interferences with the concentration-dependent effect of oxaliplatin were registered after 48 h treatment with 10 μM of carbophenyline, a concentration about 65-fold higher in comparison to the I1-R affinity [30, 31]. Carbophenyline per se did not alter HT-29 cell viability suggesting the lack of a direct effect against cancer cells. On the base of pharmacophore modelling and virtual docking study, I1-R agonists were suggested as possible apoptotic agents in cancer cells [66]. In the pheochromocytoma-derived cell line PC12, not specific I1-R ligands showed antiproliferative effect [55]. Generally, imidazolines can sensitize cancer cells to antineoplastic DNA damaging agents [67]. The role of I1-R in cancer and the effects of specific ligands, like carbophenyline, in cancer mechanisms as well as in the possible pharmacological interactions with antitumor drugs deserve deep investigation.
The development of oxaliplatin neuropathy is a clinical issue in patients receiving a cumulative dose exceed 780–850 mg/m2, approximately two-thirds will have symptoms 1 year after treatment or beyond [4, 68]. The lack of effective therapies claims for new pharmacological approaches: in this context I1-R stimulation rises to a very interesting potential.
In conclusion, for the first time the I1-R has been demonstrated to be a valuable and effective target for the management of oxaliplatin-induced neuropathic pain. The potent and selective I1-R agonist carbophenyline is a lead compound for the development of an innovative class of pain killers. The novelty of the obtained results stimulates the continuation of the research both from chemical and biological point of view. In fact, the presence in carbophenyline of an asymmetric carbon atom suggests the examination of the influence of stereochemistry on its anti-neuropathic effect. Moreover, it might be of interest to investigate whether I1-R ligands can be active also in other type of neuropathic pain as well as to highlight the molecular pharmacodynamic of pain relief.
References
International Association for the Study of Pain. IASP Taxonomy. Pain terms. Neuropathic pain. Updated 2017. www.iasp-pain.org/Taxonomy#Neuropathicpain
André T, Boni C, Mounedji-Boudiaf L, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004;350:2343–51.
Nordlinger B, Sorbye H, Glimelius B, et al. Perioperative FOLFOX4 chemotherapy and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC 40983): long-term results of a randomised, controlled, phase 3 trial. Lancet Oncol. 2013;14:1208–15.
Beijers AJM, Mols F, Vreugdenhil G. A systematic review on chronic oxaliplatin-induced peripheral neuropathy and the relation with oxaliplatin administration. Support Care Cancer. 2014;22:1999–2007.
Balayssac D, Ferrier J, Descoeur J, et al. Chemotherapy-induced peripheral neuropathies: from clinical relevance to preclinical evidence. Expert Opin Drug Saf. 2011;10:407–17.
Kiernan MC. The pain with platinum: oxaliplatin and neuropathy. Eur J Cancer. 2007;43:2631-2633.
Argyriou AA, Cavaletti G, Briani C, et al. Clinical pattern and associations of oxaliplatin acute neurotoxicity: a prospective study in 170 patients with colorectal cancer. Cancer. 2013;119(2):438-44.
Ta LE, Espeset L, Podratz J, Windebank AJ. Neurotoxicity of oxaliplatin and cisplatin for dorsal root ganglion neurons correlates with platinum-DNA binding. Neurotoxicol. 2006;27:992-1002.
Di Cesare Mannelli L, Pacini A, Bonaccini L, Zanardelli M, Mello T, Ghelardini C. Morphologic features and glial activation in rat oxaliplatin-dependent neuropathic pain. J Pain. 2013;14:1585-600.
Di Cesare Mannelli L, Pacini A, Micheli L, Tani A, Zanardelli M, Ghelardini C. Glial role in oxaliplatin-induced neuropathic pain. Exp Neurol. 2014;261:22-33.
Di Cesare Mannelli L, Pacini A, Matera C, et al. Involvement of α7 nAChR subtype in rat oxaliplatin-induced neuropathy: effects of selective activation. Neuropharmacol. 2014;79:37-48.
Di Cesare Mannelli L, Marcoli M, Micheli L, et al. Oxaliplatin evokes P2X7-dependent glutamate release in the cerebral cortex: A pain mechanism mediated by Pannexin 1. Neuropharmacol. 2015;97:133-41.
Ibrahim EY, Ehrlich BE. Prevention of chemotherapy-induced peripheral neuropathy: A review of recent findings. Crit Rev Oncol Hematol. 2020;145:102831.
Hershman DL, Lacchetti C, Loprinzi CL. Prevention and Management of Chemotherapy-Induced Peripheral Neuropathy in Survivors of Adult Cancers: American Society of Clinical Oncology Clinical Practice Guideline Summary. J Oncol Pract. 2014;10(6):e421-e424.
Miltenburg NC, Boogerd W. Chemotherapy-induced neuropathy: A comprehensive survey. Cancer Treat Rev. 2014;40(7):872-82.
Alemany R, Olmos G, GarciaSevilla JA. Labelling I-2B-imidazoline receptors by [H-3]2-(2-benzofuranyl)-2-imidazoline (2-BFI) in rat brain and liver: characterization, regulation and relation to monoamine oxidase enzymes. Naunyn-Schmied. Arch Pharmacol. 1997;356:39-47.
Gentili F, Cardinaletti C, Carrieri A, et al. Involvement of I2-imidazoline binding sites in positive and negative morphine analgesia modulatory effects. Eur J Pharmacol. 2006;553:73–81.
Li JX, Thorn DA, Qiu Y, Peng BW, Zhang Y. Antihyperalgesic effects of imidazoline I2 receptor ligands in rat models of inflammatory and neuropathic pain. Br J Pharmacol. 2014;171:1580-90.
Bektas N, Nemutlu D, Arsla R. The imidazoline receptors and ligands in pain modulation. Indian J Pharmacol. 2015;47:472-478.
Thorn DA, Zhang Y, Li JX. Tolerance and cross-tolerance to the antinociceptive effects of oxycodone and the imidazoline I2 receptor agonist phenyzoline in adult male rats. Psychopharmacol. 2017;234:1871-1880.
Nikolic K, Agbaba D. Pharmacophore development and SAR studies of imidazoline receptor ligands. Mini-Rev Med Chem. 2012;12:1542−1555.
Cobos-Puc L, Aguayo-Morales H. Cardiovascular Effects Mediated by Imidazoline Drugs: An Update. Cardiovasc Hematol Disord Drug Targets. 2019;19(2):95-108.
Head GA, Burke SL. I1 imidazoline receptors in cardiovascular regulation: the place of rilmenidine. Am J Hypertens. 2000;13:89S–98S.
Ernsberger P. The I1-imidazoline receptor and its cellular signaling pathways. Ann N Y Acad Sci. 1999;881:35–53.
Musgrave IF, Hughes R. 1999. Novel targets and techniques in imidazoline receptor research. Ann N Y Acad Sci. 1999;881:217–228.
Greney H, Ronde P, Magnier C, et al. Coupling of I(1) imidazoline receptors to the cAMP pathway: studies with a highly selective ligand, benazoline. Mol Pharmacol. 2000;57:1142–1151.
Yoro SG, Urosevic D, Fellmann L, Greney H, Bousquet P, Feldman J. G-protein inwardly rectifying potassium channels are involved in the hypotensive effect of I1-imidazoline receptor selective ligands. J Hypertens. 2008;26:1025–1032.
Kim YH, Nam TS, Ahn DS, Chung S. Modulation of N-type Ca(2)(+) currents by moxonidine via imidazoline I(1) receptor activation in rat superior cervical ganglion neurons. Biochem Biophys Res Commun. 2011;409:645–650.
Harraz OF, El-Gowelli HM, Mohy El-Din MM, Ghazal AR, El-Mas MM. Adenosinergic modulation of the imidazoline I(1)- receptor-dependent hypotensive effect of ethanol in acute renal failure. Food Chem Toxicol. 2012;50:2622–2628.
Gentili F, Bousquet P, Carrieri A, et al. Rational design of the new antihypertensive I1-receptor ligand 2-(2-biphenyl-2-yl-1-methyl-ethyl)-4,5-dihydro-1H-imidazole. Lett Drug Des Discov. 2005;2:571-578.
Del Bello F, Bargelli V, Cifani C, et al. Antagonism/agonism modulation to build novel antihypertensives selectively triggering I1-imidazoline receptor activation. ACS Med Chem Lett. 2015;6:496-501.
Gentili F, Bousquet P, Brasili L, et al. Imidazoline Binding Sites (IBS) profile modulation: key role of the bridge in determining I1-IBS or I2-IBS selectivity within a series of 2-phenoxymethylimidazoline analogues. J Med Chem. 2003;46:2169-2176.
Eglen RM, Hudson AL, Kendall DA, et al. “Seeing through a glass darkly”: casting light on imidazoline “I” sites. Trends Pharmacol Sci. 1998;19:381-390.
Hudson AL, Gough R, Tyacke R, et al. Novel selective compounds for the investigation of imidazoline receptors. Ann N Y Acad Sci. 1999;881:81-91.
McGrath JC, Lilley E. Implementing guidelines on reporting research using animals (ARRIVE etc.): new requirements for publication in BJP. Br J Pharmacol. 2015;172:3189–3193.
Cavaletti GG, Tredici MG, Petruccioli E, et al. Effects of different schedules of oxaliplatin treatment on the peripheral nervous system of the rat. Eur. J. Cancer. 2001;37:2457-2463.
Micheli L, Di Cesare Mannelli L, Rizzi A, et al. 2015. Intrathecal administration of nociceptin/orphanin FQ receptor agonists in rats: A strategy to relieve chemotherapy-induced neuropathic hypersensitivity. Eur J Pharmacol. 2015;766:155-62.
Tanabe M, Takasu K, Takeuchi Y, Ono H. Pain relief by gabapentin and pregabalin via supraspinal mechanisms after peripheral nerve injury. J Neurosci Res. 2008;86(15):3258-64.
Russo R, D'Agostino G, Mattace Raso G, et al. Central administration of oxytocin reduces hyperalgesia in mice: implication for cannabinoid and opioid systems. Peptides. 2012;38(1):81-8.
Baptista-De-Souza D, Di Cesare Mannelli L, Zanardelli M, et al. Serotonergic modulation in neuropathy induced by oxaliplatin: effect on the 5HT2C receptor. Eur J Pharmacol. 2014;735:141-9.
Sakurai M, Egashira N, Kawashiri T, Yano T, Ikesue H, Oishi R. Oxaliplatin-induced neuropathy in the rat: involvement of oxalate in cold hyperalgesia but not mechanical allodynia. Pain. 2009;147:165–174.
Di Cesare Mannelli L, Micheli L, Maresca M, et al. Anti-neuropathic effects of Rosmarinus officinalis L. terpenoid fraction: relevance of nicotinic receptors. Sci Rep. 2016;6:34832
Cardinaletti C, Mattioli L, Ghelfi F, et al. Might adrenergic α2C-agonists/α2A-antagonists become novel therapeutic tools for pain treatment with Morphine? J Med Chem. 2009;52:7319–7322.
Del Bello F, Diamanti E, Giannella M, et al. Low doses of allyphenyline and cyclomethyline, effective against morphine dependence, elicit antidepressant-like effect. ACS Med Chem Lett. 2012;3:535-539.
Del Bello F, Giannella M, Piergentili A, Quaglia W. The 2-substituted imidazoline ring linked to an aromatic moiety by a biatomic bridge: a bioversatile scaffold. Glob Drugs Therap. 2016;1:1-4.
Giusepponi ME, Cifani C, Micioni Di Bonaventura MV, et al. Combined interactions with I1-, I2-imidazoline binding sites and α2-adrenoceptors to manage opioid addiction. ACS Med Chem Lett. 2016;7:956-961.
Gentili F, Bousquet P, Brasili L, et al. α2-Adrenoreceptors profile modulation and high antinociceptive activity of (S)-(-)-2-[1-(biphenyl-2-yloxy)ethyl]-4,5-dihydro-1H-imidazole. J Med Chem. 2002;45:32–40.
Del Bello F, Cilia A, Carrieri A, et al. The versatile 2-substituted imidazoline nucleus as a structural motif of ligands directed to the serotonin 5-HT1A receptor. Chem Med Chem. 2016;11:2287–98.
Di Cesare Mannelli L, Ghelardini C, Micheli L, et al. Synergic stimulation of serotonin 5-HT1A receptor and α2-adrenoceptors for neuropathic pain relief: Preclinical effects of 2-substituted imidazoline derivatives. Eur J Med Chem. 2017;810:128-133.
Eisenach JC, DuPen S, Dubois M, Miguel R, Allin D. Epidural clonidine analgesia for intractable cancer pain. The Epidural Clonidine Study Group. Pain. 1995;61:391-9.
Pan HL, Chen SR, Eisenach JC. Role of spinal NO in antiallodynic effect of intrathecal clonidine in neuropathic rats. Anesthesiology. 1998;89:1518-23.
Szabo B. Imidazoline antihypertensive drugs: a critical review on their mechanism of action. Pharmacol Ther. 2002;93(1):1-35.
Wrzosek A, Woron J, Dobrogowski J, Jakowicka-Wordliczek J, Wordliczek J. Topical clonidine for neuropathic pain. Cochrane Database Syst Rev. 2015;318:CD010967.
Stone LS, Kitto KF, Eisenach JC, Fairbanks CA, Wilcox GL. ST91 [2-(2,6-diethylphenylamino)-2-imidazoline hydrochloride]-mediated spinal antinociception and synergy with opioids persists in the absence of functional alpha-2A- or alpha-2C-adrenergic receptors. J Pharmacol Exp Ther. 2007;323:899–906.
Bousquet P, Hudson A, García-Sevilla JA, Li JX. Imidazoline Receptor System: The Past, the Present, and the Future. Pharmacol Rev. 2020;72:50-79.
Ernsberger P, Shen IH. Membrane localization and guanine nucleotide sensitivity of medullary I1-imidazoline binding sites. Neurochem Int. 1997;30:17–23.
Edwards L, Ernsberger P. The I(1)-imidazoline receptor in PC12 pheochromocytoma cells reverses NGF-induced ERK activation and induces MKP-2 phosphatase. Brain Res. 2003;980:71–79.
Alles SRA, Smith PA. Etiology and Pharmacology of Neuropathic Pain. Pharmacol Rev. 2018;70:315-347.
Coppi E, Cherchi F, Fusco I, et al. Adenosine A3 receptor activation inhibits pronociceptive N-type Ca2+ currents and cell excitability in dorsal root ganglion neurons. Pain. 2019;160:1103-1118.
Tanabe M, Kino Y, Honda M, Ono H. Presynaptic I1-imidazoline receptors reduce GABAergic synaptic transmission in striatal medium spiny neurons. J Neurosci. 2006;26:1795–1802.
Dixit MP, Upadhya MA, Taksande B, et al. Neuroprotective effect of agmatine in mouse spinal cord injury model: Modulation by imidazoline receptors. J Nat Sci Biol Med. 2018;9:115-120.
Gupta S, Sharma B. Pharmacological benefit of I(1)-imidazoline receptors activation and nuclear factor kappa-B (NF-κB) modulation in experimental Huntington's disease. Brain Res Bull. 2014;102:57-68.
Milligan ED, Watkins LR. Pathological and protective roles of glia in chronic pain. Nat Rev Neurosci. 2009;10(1):23-36.
Von Hehn CA, Baron R, Woolf CJ. Deconstructing the Neuropathic Pain Phenotype to Reveal Neural Mechanisms. Neuron. 2012;73(4):638–652.
Jacob A, Hack B, Chiang E, Garcia JG, Quigg RJ, Alexander JJ. C5a alters blood-brain barrier integrity in experimental lupus. FASEB J. 2010;24(6):1682-8.
Nikolic K, Veljkovic N, Gemovic B, Srdic-Rajic T, Agbaba D. Imidazoline-1 receptor ligands as apoptotic agents: pharmacophore modeling and virtual docking study. Comb Chem High Throughput Screen. 2013;16:298-319
Sharma V, Peddibhotla S, Tepe JJ. Sensitization of cancer cells to DNA damaging agents by imidazolines. J Am Chem Soc. 2006;128:9137-43.
Seretny M, Currie GL, Sena ES, et al. Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: A systematic review and meta-analysis. Pain. 2014;155:2461–2470.
Acknowledgments
This research was funded by the Italian Ministry of Instruction, University and Research (MIUR), by the University of Florence and by the University of Camerino (Fondo di Ateneo per la Ricerca 2018).
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Dedicated to Prof. Maria Pigini
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Micheli, L., Di Cesare Mannelli, L., Del Bello, F. et al. The Use of the Selective Imidazoline I1 Receptor Agonist Carbophenyline as a Strategy for Neuropathic Pain Relief: Preclinical Evaluation in a Mouse Model of Oxaliplatin-Induced Neurotoxicity. Neurotherapeutics 17, 1005–1015 (2020). https://doi.org/10.1007/s13311-020-00873-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13311-020-00873-y