Abstract
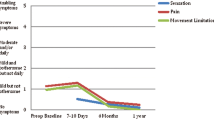
Mesh repair has significantly reduced recurrence rate after groin hernia surgery. Recently, attention has shifted to issues such as chronic pain and discomfort, leading to development of lightweight and partially re-absorbable meshes. The aim of the study was to evaluate the effect of lightweight mesh vs heavyweight mesh on post-operative pain, discomfort and quality of life in short and medium term after inguinal hernia surgery. Eight hundred and eight patients with primary inguinal hernia were allocated to anterior repair (Lichtenstein technique) using a lightweight mesh (Ultrapro®) or a heavyweight mesh (Prolene®). Primary outcomes were incidence of chronic pain and discomfort at 6-month follow-up. Secondary endpoints were quality of life (QoL), pain and complication at 1 week, 1 and 6 months. At 6 months, 25% of patients reported pain of some intensity; severe pain was reported by 1% of patients in both groups. A statistically significant difference in favour of lightweight mesh was found at multivariable analysis for pain (1 week and 6 months after surgery: p = 0.02 and p = 0.04, respectively) and QoL at 1 month and 6 months (p = 0.05 and p = 0.02, respectively). There was no difference in complication rate and no hernia recurrences were detected. The use of lightweight mesh in anterior Lichtenstein inguinal hernia repair significantly reduced the incidence of pain and favourably affected the perceived quality of life at 6 months after surgery compared to heavyweight mesh.
Similar content being viewed by others
References
Rosenberg J, Bisgaard T, Kehlet H et al (2011) Danish Hernia Database recommendations for the management of inguinal and femoral hernia in adults. Dan Med Bull 58:C4243
Amid PK (2004) Lichtenstein tension-free hernioplasty: its inception, evolution, and principles. Hernia 8:1–7
Bay-Nielsen M, Kehlet H, Strand L et al (2001) Quality assessment of 26,304 herniorrhaphies in Denmark: a prospective nationwide study. Lancet 358:1124–1128
Klinge U, Klosterhalfen B, Muller M et al (1999) Foreign body reaction to meshes used for the repair of abdominal wall hernias. Eur J Surg 165:665–673
Simons MP, Aufenacker T, Bay-Nielsen M et al (2009) European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia 13:343–403
Bay-Nielsen M, Perkins FM, Kehlet H (2001) Pain and functional impairment 1 year after inguinal herniorrhaphy: a nationwide questionnaire study. Ann Surg 233:1–7
Weyhe D, Belyaev O, Muller C et al (2007) Improving outcomes in hernia repair by the use of light meshes—a comparison of different implant constructions based on a critical appraisal of the literature. World J Surg 31:234–244
Smietanski M, Bury K, Smietanska IA et al (2011) Five-year results of a randomised controlled multi-centre study comparing heavy-weight knitted versus low-weight, non-woven polypropylene implants in Lichtenstein hernioplasty. Hernia 15:495–501
Li J, Ji Z, Cheng T (2012) Lightweight versus heavyweight in inguinal hernia repair: a meta-analysis. Hernia 16:529–539
Sajid MS, Leaver C, Baig MK et al (2012) Systematic review and meta-analysis of the use of lightweight versus heavyweight mesh in open inguinal hernia repair. Br J Surg 99:29–37
Smietanski M, Smietanska IA, Modrzejewski A et al (2012) Systematic review and meta-analysis on heavy and lightweight polypropylene mesh in Lichtenstein inguinal hernioplasty. Hernia 16:519–528
Uzzaman MM, Ratnasingham K, Ashraf N (2012) Meta-analysis of randomized controlled trials comparing lightweight and heavyweight mesh for Lichtenstein inguinal hernia repair. Hernia 16:505–518
Pocock SJ, Simon R (1975) Sequential treatment assignment with balancing for prognostic factors in the controlled clinical trial. Biometrics 31:103–115
Bharmal M, Thomas J 3rd (2006) Comparing the EQ-5D and the SF-6D descriptive systems to assess their ceiling effects in the US general population. Value Health 9:262–271
Staal E, Nienhuijs SW, Keemers-Gels ME et al (2008) The impact of pain on daily activities following open mesh inguinal hernia repair. Hernia 12:153–157
Melzack R (1987) The short-form McGill Pain Questionnaire. Pain 30:191–197
Bender R, Lange S (2001) Adjusting for multiple testing—when and how? J Clin Epidemiol 54:343–349
Harrell FEJ (2013) rms: regression modeling strategies. Rpackage version 4.0-0
R Development Core Team (2012). R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing
Markar SR, Karthikesalingam A, Alam F et al (2010) Partially or completely absorbable versus nonabsorbable mesh repair for inguinal hernia: a systematic review and meta-analysis. Surg Laparosc Endosc 20:213–219
Miserez M, Peeters E, Aufenacker T et al (2014) Update with level 1 studies of the European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia 18:151–163
Zhong C, Wu B, Yang Z et al (2013) A meta-analysis comparing lightweight meshes with heavyweight meshes in Lichtenstein inguinal hernia repair. Surg Innov 20:24–31
Gao M, Han J, Tian J et al (2010) Vypro II mesh for inguinal hernia repair: a meta analysis of randomized controlled trials. Ann Surg 251:838–842
Palmqvist E, Larsson K, Anell A et al (2013) Prospective study of pain, quality of life and the economic impact of open inguinal hernia repair. Br J Surg 2013(100):1483–1488
Nikkolo C, Murruste M, Vaasna T et al (2012) Three-year results of randomised clinical trial comparing lightweight mesh with heavyweight mesh for inguinal hernioplasty. Hernia 16:555–559
Kehlet H, Roumen RM, Reinpold W et al (2013) Invited commentary: persistent pain after inguinal hernia repair: what do we know and what do we need to know? Hernia 17:293–297
Bringman S, Heikkinen TJ, Wollert S et al (2004) Early results of a single-blinded, randomized, controlled, Internet-based multicenter trial comparing Prolene and Vypro II mesh in Lichtenstein hernioplasty. Hernia 8:127–134
Bringman S, Wollert S, Osterberg J et al (2005) One year results of a randomised controlled multi-centre study comparing Prolene and Vypro II-mesh in Lichtenstein hernioplasty. Hernia 9:223–227
Nikkolo C, Lepner U, Murruste M et al (2010) Randomised clinical trial comparing lightweight mesh with heavyweight mesh for inguinal hernioplasty. Hernia 14:253–258
Smietanski M (2008) Randomized clinical trial comparing a polypropylene with a poliglecaprone and polypropylene composite mesh for inguinal hernioplasty. Br J Surg 95:1462–1468
Acknowledgements
SUPERMESH Study Group: Bechi P, Firenze; Bona S, Milano; Carlucci M, Milano; Colizza S, Roma; Danelli PG, Milano; .D’Ambra L, La Spezia; Di Sebastiano P, San Giovanni Rotondo (FG); Fiore B, Milano; Fiscon V, Cittadella (PD); Lo Bello R, Napoli; Logrieco G, Acquaviva delle Fonti (BA); Montorsi M, Milano; Morpurgo E,Camposanpiero (PD); Opocher E, Milano; Perrone G, Bergamo; Rosati R, Milano; Sansonetti G, Cinisello Balsamo (MI); Schillaci V, Catania; Sgroi G, Treviglio (BG).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
S.B. declares potential conflict of interest (he was a paid consultant for Johnson & Johnson Medical until 2013). No other author reports potential conflict of interest.
Research involving human participants and/or animals
This study has been approved by the appropriate institutional research ethics committee and has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Written informed consent was obtained by all patients upon enrolment.
Additional information
The members of SUPERMESH Study Group are listed in Acknowledgements at the end of the article.
Rights and permissions
About this article
Cite this article
Bona, S., Rosati, R., Opocher, E. et al. Pain and quality of life after inguinal hernia surgery: a multicenter randomized controlled trial comparing lightweight vs heavyweight mesh (Supermesh Study). Updates Surg 70, 77–83 (2018). https://doi.org/10.1007/s13304-017-0483-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-017-0483-3