Abstract
Introduction
An Excel® (Microsoft Corporation) model was adapted to estimate the short-term (1-year) cost effectiveness of insulin detemir (IDet) versus neutral protamine Hagedorn (NPH) insulin in patients initiating insulin treatment with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) in Spain.
Methods
Clinical benefits included the non-severe hypoglycemia rate for T1DM and T2DM, and weight change for T2DM. Three scenarios were included with different hypoglycemia rates estimated on the basis of clinical trials and observational studies. Costs, estimated from perspective of the Spanish Public Healthcare System (Euros 2014), included insulin treatment and non-severe hypoglycemia management costs. Non-severe hypoglycemia, defined as a self-managed event, implied the use of extra glucose testing strips and a general practitioner visit during the week following the event for 25% of patients. An average disutility value was associated to non-severe hypoglycemia events and, for T2DM, to one body mass index unit gain to calculate quality-adjusted life years (QALYs).
Results
For the three scenarios a range of 0.025–0.076 QALYs for T1DM and 0.014–0.051 QALYs for T2DM were gained for IDet versus NPH due to non-severe hypoglycemia and weight gain avoidance, in return of an incremental cost of €145–192 for T1DM and €128–206 for T2DM. This resulted in the IDet versus NPH incremental cost-effectiveness ratio (ICER) ranging between €1910/QALY and €7682/QALY for T1DM and €2522/QALY and €15,009/QALY for T2DM.
Conclusion
IDet was a cost-effective alternative to NPH insulin in the first year of treatment of patients with T1DM and patients with T2DM in Spain, with ICERs under the threshold value commonly accepted in Spain (€30,000/QALY).
Funding
Novo Nordisk.
Similar content being viewed by others
Introduction
Diabetes mellitus is a group of heterogeneous metabolic disorders with the common features of hyperglycemia and glucose intolerance, which affects about 10.8% of the Spanish population [1]. In patients with type 1 diabetes mellitus (T1DM), insulin endogenous secretion is impaired, while patients with type 2 diabetes mellitus (T2DM), which represent about 90% of diabetes cases, do not respond to endogenous insulin properly [2]. Therefore, insulin therapy is indicated for all patients with T1DM as they are completely dependent on exogenous insulin; however, only patients with T2DM who are unable to achieve adequate glycemic control by other measures (exercise, diet, and/or other antidiabetic agents) will receive insulin treatment [3].
The main treatment goal in people with diabetes is to achieve a normal blood glucose level to reduce the risk of microvascular and macrovascular long-term complications [2]; however, acute hypoglycemia represents the most common adverse event associated with insulin treatment [4] and have a substantial economic and social impact [5]. Therefore, the principle of ‘basal’ insulin use is the optimization of glycemic control, minimizing the risk of hypoglycemia to improve health-related quality-of-life (HRQoL) and adherence to treatment [6].
Either intermediate-acting (neutral protamine Hagedorn [NPH]) or long-acting (insulin glargine or insulin detemir [IDet]) formulations may be used as initial basal therapy aimed to control blood glucose between meals and overnight [6]. Long-acting insulin analogs more accurately mimic the physiological human insulin profile and provide an alternative to human insulins such as NPH insulin [6]. In addition, long-acting insulins are associated with fewer hypoglycemia events and less weight gain than NPH [7, 8]. These improvements lead to better quality-adjusted life years (QALYs) compared with NPH insulin [9, 10].
Hypoglycemia can be classified on the basis of the event management. While severe hypoglycemia requires the assistance from another person, sometimes from a medical professional and even hospital care [11], non-severe hypoglycemia is defined as an episode that can be managed by the patients themselves. Although non-severe hypoglycemic events are easier to manage, they are more common than severe events [12]. Recurrent episodes of non-severe hypoglycemia may cause behavioral changes and cognitive impairment, leading patients to develop fear of hypoglycemia [13], reducing HRQoL, and affecting treatment adherence [13–15]. Furthermore, severe and non-severe hypoglycemia is associated with direct healthcare costs and indirect costs due to productivity loss [11, 16, 17]. Weight gain is also commonly associated with insulin therapy, especially among patients with T2DM [18], and is linked to an increased risk of cardiovascular morbidity and mortality [19].
For these reasons, the importance of controlling hypoglycemia events in the management of both T1DM and T2DM has been recognized in the reimbursement process in several countries. Insulin analogs with a reduced rate of hypoglycemia and weight gain, like IDet, may provide long- and short-term health benefits with respect to traditional human insulins [20, 21]; however, this clinical benefit and improved quality of life comes with the additional pharmacy costs associated with IDet.
Thus, we aimed to estimate the cost effectiveness of IDet compared with NPH insulin when initiating insulin treatment in patients with T1DM and patients with T2DM using a conservative short-term modeling approach from the perspective of the Spanish National Health System (NHS).
The present study is based on the treatment benefits associated with a reduction in hypoglycemic events and less weight gain as observed in clinical trials and observational studies. Given the short-term design of these studies, a short-term modeling approach is the most conservative option to include clinical data from independent sources and to avoid unrealistic extrapolation derived from long-term horizons. As this kind of cost-effectiveness analysis (CEA) for IDet versus NPH is only available for Scandinavian countries [10, 22], this is the first study, to our knowledge, from the perspective of the Spanish NHS.
Methods
Cost-Effectiveness Model
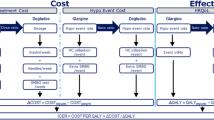
A 1-year CEA model was developed in Excel® 2007 (Microsoft Corporation) to compare basal insulin treatment with IDet against NPH insulin for the management of T1DM and T2DM. For the current analysis, input parameters of the model were adapted to the Spanish NHS. The clinical benefit was defined in terms of QALYs and reflects the effects of hypoglycemic events and weight gain. Only direct health care costs are included (insulins and management of minor hypoglycemic events).
Structure of the Model
The structure of the model is based on two Scandinavian studies: Valentine et al. [10] (T1DM) and Ridderstråle et al. [22] (T2DM). To assess the comparison, two arms are considered, one receiving IDet and one receiving NPH insulin. Each arm is provided with input data obtained from clinical trials [20, 21] and observational studies [23, 24]. As the model was designed to only take into account statistically significant clinical benefits, only the rate of non-severe hypoglycemia for both T1DM and T2DM [10] and the weight change from baseline for T2DM [22] are included in the evaluation. Other clinical parameters, such as severe hypoglycemic events and glycated hemoglobin control were excluded as their differences were found to be not significant. Disutility associated to weight gain and hypoglycemic events is used to estimate QALYs and the incremental cost-effectiveness ratio (ICER; €/QALY) is estimated as the relation between QALYs and costs of one arm versus another.
Study Population
The population consisted in two hypothetical cohorts of patients with either T1DM or T2DM taking up insulin treatment. The number of individuals in the cohort is irrelevant to the result as cost and clinical outcomes are expressed as average yearly costs and rates of event per patient.
Treatments to Compare
One cohort of patients was treated with IDet and the other with NPH. The daily dose was assumed to be 40 IU/day [10], which is the defined daily dose (DDD) indicated by the World Health Organization (WHO) [25].
Time Horizon and Perspective
To estimate the cost-effectiveness of IDet compared to NPH in patients with T1DM and patients with T2DM in Spain, the time horizon considered was one year. The perspective adopted was that of the Spanish NHS.
Clinical Data Inputs
Due to the lack of a study which examines hypoglycemia rates, hypoglycemia IDet/NPH ratios, and weight changes associated to both treatment arms (NPH and IDet for patients with T1DM and patients with T2DM) we have collected clinical data inputs from independent robust sources which are summarized below.
Hypoglycemia Rates
Three scenarios were estimated in which the NPH arm was assigned three different hypoglycemia rates (Table 1). Two scenarios were defined based on data from an observational prospective study conducted in six secondary care diabetes centers in the UK over 9–12 months [23]. The UK Hypoglycemia Study [23] hypothesized that diabetes type and the duration of insulin treatment influenced the risk of hypoglycemia. To test this hypothesis, they explored the epidemiology of hypoglycemia in individuals with diabetes. A total of 383 patients aged 17–75 years were included in three treatment groups for T2DM: (1) sulfonylurea, (2) insulin for <2 years, and (3) insulin for >5 years, and into two treatment groups for T1DM: (1) insulin for <5 years and (2) insulin for >15 years and asked to self-report all non-severe (self-treated) and severe hypoglycemic episodes (requiring medical assistance). Hypoglycemic episodes were recorded using two complementary methods: Self-reported, when a patient experiences symptoms usually associated with hypoglycemia, or biochemical, reporting all episodes with glucose levels <3.0 mmol/L. The hypoglycemia rates were extracted from self-reported hypoglycemic episodes corresponding to the cohort of patients that recently began insulin treatment. The follow-up period ranged from a mean of 8.8 [standard deviation (SD) 2.6] months in sulfonylurea-treated subjects to 10.3 (SD 3.0) months in longstanding T1DM. As a result, for patients with T1DM treated <5 years a mean of 35.5 [95% confidence interval (CI) 22.8–48.2] events were self-reported, while a mean of 4.08 (95% CI 2.4–5.8) episodes were reported for patients with T2DM treated <2 years. From a cohort with more than 15 years of insulin treatment, a mean of 29.0 (95% CI 16.4–41.8) events for T1DM and 10.2 (95% CI 5.4–15.0) episodes for T2DM were selected.
A more recent observational cross-sectional study carried out in Spain [24] was chosen to estimate the third scenario, describing the frequency of self-reported non-severe and severe hypoglycemic events in patients with T1DM and patients with T2DM. A total of 630 individuals [n = 294 (47%) with T1DM and n = 336 (53%) with T2DM] aged 15 years or older and receiving insulin were enrolled to fill out a questionnaire survey, and a total of 506 patients completed the four questionnaires comprised in the study. The first questionnaire collected the self-reported frequency of non-severe and severe hypoglycemia in the previous year, while the other three questionnaires were administered weekly, to record the frequency of non-severe and severe hypoglycemia in the preceding 7 days. From this study, a mean yearly non-severe hypoglycemia rate of 88.0 for T1DM and of 18.3 for T2DM receiving basal-only therapy based on long-acting insulin were selected to feed the model.
Hypoglycemia IDet/NPH Ratios
Lacking a robust source for the non-severe hypoglycemia IDet/NPH ratio, the IDet/NPH rate ratio (RR) for overall hypoglycemic events in patients with T1DM was extracted from a systematic review and meta-analysis study carried out by the Canadian Agency for Drugs and Technologies in Health (CADTH) [21]. The study aimed to evaluate the clinical efficacy and safety of the long-acting insulin analogs compared with intermediate- and long-acting unmodified human insulins and oral antidiabetic drugs (OADs) for the treatment of T1DM, T2DM, and gestational diabetes [21]. MedLine, Embase, and BIOSIS previews and the Cochrane library were searched for the terms “diabetes,” “long-acting insulin,” and “short-acting insulin analogs,” and filtered by randomized controlled trials (RCTs), over the period of 1990–2007. Outcomes of interest included glycemic control (glycosylated hemoglobin or A1C, fasting plasma glucose), hypoglycemia, body weight, quality of life, patient satisfaction, healthcare utilization, and long-term complications of diabetes. From a meta-analysis of six RCTs (2109 patients), the reduction of non-severe hypoglycemia was statistically significant when IDet was compared with NPH, with an IDet/NPH overall hypoglycemia RR of 0.84 (95% CI 0.74–0.97).
On the other hand, IDet/NPH RR was estimated at 0.52 (0.44–0.61) for patients with T2DM [20, 22], on the basis of a multicenter, randomized, open-label, 3-arm (morning or evening IDet vs. evening NPH), parallel-group clinical trial [20]. This trial was conducted in 91 centers across Europe and the United States over 20 weeks. It was aimed at comparing the effectiveness and tolerability of IDet administered once daily with those of NPH insulin. Patients eligible for inclusion were adults, with a body mass index (BMI) ≤40 kg/m2, who had a diagnosis of T2DM of at least 12 months, and were receiving ≥1 OAD but were insulin naïve. A subanalysis comparing IDet clinical outcomes by time of administration was also conducted. A total of 504 patients were enrolled (285 male, 219 female); patients were randomly assigned in a 1:1:1 ratio to receive either IDet before breakfast (n = 168); IDet in the evening (n = 170), or NPH insulin in the evening (n = 166). When evening administration of IDet was compared with NPH, the hypoglycemic RR obtained was 0.52 for patients with T2DM.
Considering the rate of non-severe hypoglycemia in the NPH arm and the overall hypoglycemia IDet/NPH RR in T1DM [21] and T2DM [20], the rate of approximate non-severe hypoglycemia episodes for patients with T1DM and patients with T2DM who received IDet was calculated by multiplying the respective hypoglycemic rate for patients receiving NPH insulin by the IDet/NPH RR, as reported in Table 1 for both T1DM and T2DM.
Weight
The same IDet versus NPH RCT described above [20] was the source of the IDet versus NPH mean weight change difference for patients with T2DM. The study reported an increase in body weight following insulin initiation in both evening IDet and NPH insulin groups (0.7 vs. 1.6 kg, respectively) with respect to the baseline, with the mean difference being 0.91 kg (P < 0.005) [20].
Source of Costs Data
Only pharmacy and non-severe hypoglycemia management costs were included in the model. Costs were computed from the perspective of the Spanish NHS and are expressed in Euros of the year 2014 [26]. Pharmacy daily costs are detailed in Table 2. Pharmacy costs were reimbursed by the Spanish NHS. IDet cost is discounted by 7.5% (discount in force at the time of the estimation, i.e., September 2014).
Non-severe hypoglycemia management costs were assumed to consist only of the cost of an average of 5.6 extra glucose test strips per event, as it was considered that this would be the only measurable cost of self-managed hypoglycemia [10, 17, 22]. Additionally, it was assumed that 25% of the cohort visits a general practitioner as a consequence of a non-severe hypoglycemic event, as is reported by Brod et al. [17]. The non-severe hypoglycemia management costs are detailed in Table 3.
Utilities
The utility value for symptomatic non-severe hypoglycemia for patients with T1DM and patients with T2DM was −0.0054, which was calculated by averaging the utility associated to nocturnal (−0.0067) and diurnal (−0.0041) episodes [29].
For patients with T2DM, the utility value assigned for the BMI increment was estimated in −0.0100 per BMI unit increase [30]. To estimate the utility value assigned for weight increments, weight changes were transformed in BMI changes multiplying the weight change by 0.346 (ΔBMI = Δweight/[height2]; considering the mean height of the Spanish population is 1.7 m [31], ΔBMI = Δweight × 0.346).
Sensitivity Analysis
A one-way sensitivity analysis (OWSA) was performed to assess model uncertainty. The variables were assigned consecutively an upper and a higher value to estimate the resulting ICER in each case. Intervals included in the analysis are detailed in Table 4 for both T1DM and T2DM. The values of ICER obtained with each parameter variation were represented using tornado-type diagrams. Due to the small differences in weight gain between both treatment arms, an additional scenario was evaluated dismissing the utility loss due to BMI increase.
A probabilistic sensitivity analysis (PSA) with 1000 simulations was also performed to assess model outcome stability when varying all parameters at once, according to suitable probability distribution [22]. Distributions considered for each parameter were: log-normal for hypoglycemia rates and RRs, normal for the weight changes, beta for disutilities, and gamma for hypoglycemia costs and insulin doses. Results were represented using cost-effectiveness acceptability curves and scatterplots.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Results
Base Case
Due to lower non-severe hypoglycemia rates in patients with T1DM, IDet treatment yields an improvement with respect to NPH insulin ranging between 0.025 and 0.076 QALYs, at an incremental cost between €145 and €192 per patient per year for the three scenarios considered in the base case. Therefore, the ICER for IDet versus NPH insulin was estimated at €1910/QALY to €7682/QALY, depending on the three different hypoglycemia rates considered (Table 5).
For patients with T2DM, IDet was associated with significantly fewer non-severe hypoglycemic episodes and less weight gain compared to NPH insulin, resulting in a 0.014–0.051 QALY gain for the three base-case scenarios, in return of an incremental cost of €128–€206 for the Spanish NHS. Therefore, the IDet versus NPH insulin ICER was estimated at €2522/QALY to €15,009/QALY in Spain, in relation to the three different hypoglycemia rates considered (Table 5).
One-Way Sensitivity Analysis
OWSA was conducted for key input parameters. Overall, results are detailed in Figs. 1 and 2 for T1DM and T2DM, respectively. The variable that has the highest impact on the ICER for T1DM is IDet/NPH hypoglycemia RR, with ICER values varying from €1965/QALY to €36,592/QALY, when varying the IDet/NPH hypoglycemia RR between 0.74 and 0.97. The second variable with the highest impact on ICER is the daily cost of IDet treatment, which makes ICER decrease to €3152/QALY when decreased by 20%, and increase to €9058/QALY when increased by 20%. All other variables have reduced impacts on ICER (Fig. 1). In T2DM, the variable with the highest impact on ICER is IDet cost. When increasing or decreasing it by 20%, the ICER varies between €8408/QALY and €21,610/QALY. Disutility due to weight gain is the second most sensitive variable, making the ICER increase to €19,478/QALY when it is not considered. NPH cost variation (±20%) induces a variation of ICER slightly higher than €5000/QALY. All other variables have a smaller impact on ICER (Fig. 2).
One-way sensitivity analyses for IDet versus NPH for T1DM in Spain (for NPH non-severe hypoglycemia rate = 35.5). UKHSG UK Hypoglycemic Study Group, RR CI Rate ratio confidence interval, ICER Incremental cost-effectiveness ratio, IDet Insulin detemir, NPH Neutral protamine Hagedorn, QALY Quality-adjusted life year, RR Rate ratio, T2DM Type 2 diabetes mellitus
One-way sensitivity analyses for IDet versus NPH for T2DM in Spain (for NPH non-severe hypoglycemia rate = 4.08). UKHSG UK Hypoglycemia Study Group, RR CI Rate ratio confidence interval, ICER Incremental cost-effectiveness ratio, IDet Insulin detemir, NPH Neutral protamine Hagedorn, QALY Quality-adjusted life year, RR Rate ratio, T2DM Type 2 diabetes mellitus
Probabilistic Sensitivity Analysis
Cost-effectiveness acceptability curves allow estimation of the NHS willingness-to-pay for an additional QALY at different ICER thresholds. For the threshold value currently accepted in Spain of €30,000/QALY, IDet has 89.5% and 78.7% probabilities of being cost effective versus NPH for T1DM and T2DM, respectively (Figs. 3, 4).
According to the cost-effectiveness scatterplots, IDet has a 32.9% probability of being more effective and less costly than NPH (dominant) for T1DM (Fig. 5) and 20.7% probability for T2DM (Fig. 6).
Discussion
This direct and transparent short-term CEA was planned to rely on statistically significant efficacy differences between IDet and NPH; therefore, only non-severe hypoglycemia rates for T1DM and T2DM, and weight gain differences for T2DM were included. The model is based on previously published short-term economical evaluations for Scandinavian countries (Denmark, Finland, the Netherlands, Norway, and Sweden), and, to our knowledge, is the first of its kind in Spain.
This evaluation showed that due to lower hypoglycemic rate and less weight gain, IDet versus NPH insulin is associated with an ICER that stays below the threshold commonly accepted for Spain (€30,000/QALY) [32] both in patients with T1DM and patients with T2DM, and can, therefore, be considered as cost-effective in this country.
In general, the model results are quite robust; in fact, all estimated ICERs in the OWSA remain below the cost-effectiveness acceptability threshold implicitly mentioned above. The only exception is attributing an IDet/NPH hypoglycemia RR close to 1 (RR = 0.97; upper extreme of the 95% CI from the CADTH meta-analysis average [21]), for which IDet would not be considered cost-effective with respect to NPH in patients with T1DM. This is because an RR close to 1 would imply that IDet does not provide any relevant incremental health benefit on hypoglycemia rate versus NPH; thus, its higher daily price would not be justified. Another sensitive variable, in both types of diabetes, is the treatment cost. Increasing or decreasing the cost of IDet, makes the ICER vary in an interval up to €6000/QALY wide (T1DM). These estimations are consistent with the structure of the model. Moreover, PSA simulations reveal a probability of cost-effectiveness at a €30,000/QALY threshold of about 90% and 80% in T1DM and T2DM, respectively.
The results of this evaluation must be read in the context of its limitations. First, only the short term is considered. This might underestimate the real clinical benefits of IDet with respect to NPH; in fact, the hypoglycemia rate is expected to increase with treatment duration [33].
With reference to the absolute rate of non-severe hypoglycemia in the NPH treatment arm, this is a crucial datum that affects the cost-effectiveness of IDet versus NPH for which the three scenarios were estimated in the base-case analysis. The first two scenarios use non-severe hypoglycemia rates coming from the UK Hypoglycemia Study [23] which is a reference observational study for hypoglycemia in diabetic patients. From this study, two non-severe hypoglycemia rates have been selected: one corresponding to patients with recent insulinization (<5 and <2 years for T1DM and T2DM, respectively), and one corresponding to patients with long-run insulinization (>15 and >5 years for T1DM and T2DM, respectively) [23]. The third scenario is based on non-severe hypoglycemia rates from a recently published observational study performed in Spain [24], which should be considered the closest to the “real-world” situation of the Spanish NHS, showing the highest non-severe hypoglycemia rates. The three scenarios all give cost-effective results and contemplate hypoglycemia rates. It must be considered that the non-severe hypoglycemia rate of these studies was assumed to correspond to treatment with NPH, although insulin type information was not available and most probably included different insulin types. However, this is a conservative assumption in that NPH is known to be associated to higher hypoglycemia rates than more modern insulin analogs [34]. In addition, the use of an overall hypoglycemia RR for IDet versus NPH, instead of a specific RR for non-severe hypoglycemia episodes, due to the lack of a robust source for this datum in the literature, may be considered a limitation of the model. Anyway, the approximated values that are maintained from the original models by Valentine et al. [10] and Ridderstråle et al. [22] give conservative estimations of the non-severe hypoglycemia RR, as already discussed by Valentine et al. [10], and offer the advantage of integrating data from a very controlled setting (RCTs [20, 21]), with a setting that is closer to the “real-world” data (observational study [23]), as explained by Ridderstråle et al. [22].
As the hypoglycemia rate was an overall value (no distinction between nocturnal and diurnal non-severe hypoglycemia events), the utility values associated to diurnal and nocturnal events [17] were averaged to obtain a unique value. However, this simplification was tested in the OWSA by assuming 100% nocturnal or 100% diurnal events and attributing the corresponding utility values. In both ICER estimations, IDet was considered as cost-effective with respect to NPH.
Another limitation may be the use of the WHO DDD of insulin [25]. Even though 40 IU/day is not a “real-world” dose for patients with diabetes, robust data in this sense are not available due to the dosing variability in clinical practice. Considering all exposed assumptions, this may be considered a conservative estimation.
Long-term cost effectiveness of IDet versus NPH insulin in Spain was previously assessed for basal-bolus therapy, together with four other European countries (Belgium, France, Germany, and Italy) [35]. This evaluation showed that, in the long term, IDet is dominant (in Spain, Belgium, and Germany) or very cost effective (France and Italy) with respect to NPH, due to the reduced cumulative incidence of most diabetes-related complications over the patient lifetime. Other long-term evaluations carried out for Germany, Sweden, and the UK [9, 36–38] have also shown IDet long-term cost effectiveness compared to NPH insulin in both patients with T1DM and patients with T2DM.
However, short-term cost-effectiveness analyses for IDet versus NPH are only available for Scandinavian countries, estimating ICERs in the range of €12,216/QALY to €16,568/QALY (year 2010) for T1DM [10] and €21,768/QALY to €28,349/QALY (year 2012) for T2DM [20] over a 1-year period.
Therefore, a short-term cost-effectiveness estimation was needed in Spain to address the requirements of healthcare decision makers for whom short-term cost effectiveness may be more relevant to immediate budget considerations.
Conclusions
In conclusion, this analysis shows in a direct manner that IDet can be considered cost-effective with respect to NPH insulin in a Spanish setting for the treatment of both patients with T1DM and patients with T2DM, with ICERs that are in line with or even smaller than those calculated for other European countries, and in the range commonly accepted for Spain.
References
IDF Diabetes Atlas. Available from: http://www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf. Accessed Sept 2014.
World Health Organization. Definition, diagnosis and classification of diabetes mellitus and its complications: report of a WHO Consultation. Part1: Diagnosis and classification of diabetes mellitus. Geneve: World Health Organization; 1999.
American Diabet es Association. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S14–80.
Seaquist ER, Fish L, Anderson J, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and The Endocrine Society Diabetes Care. Diabetes Care. 2013;36:1384–95.
Cryer PE, Axelrod L, Grossman AB, et al. Evaluation and management of adult hypoglycemic disorders: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2009;94(3):709–28.
Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2012;55(6):1577–96.
Hermansen K, Davies M, Derezinski T, et al. A 26-week, randomized, parallel, treat-to-target trial comparing insulin detemir with NPH insulin as add-on therapy to oral glucose-lowering drugs in insulin-naive people with type 2 diabetes. Diabetes Care. 2006;29(6):1269–74.
Meneghini LF, Rosenberg KH, Koenen C, et al. Insulin detemir improves glycaemic control with less hypoglycaemia and no weight gain in patients with type 2 diabetes who were insulin naive or treated with NPH or insulin glargine: clinical practice experience from a German subgroup of the PREDICTIVE study. Diabetes Obes Metab. 2007;9(3):418–27.
Valentine WJ, Aagren M, Haglund M, et al. Evaluation of the long-term cost-effectiveness of insulin detemir compared with neutral protamine Hagedorn insulin in patients with type 1 diabetes using a basal-bolus regimen in Sweden. Scand J Public Health. 2011;39(1):79–87.
Valentine WJ, Jendle J, Saraheimo M, et al. Evaluating the cost-effectiveness of reduced mild hypoglycaemia in subjects with Type 1 diabetes treated with insulin detemir or NPH insulin in Denmark, Sweden, Finland and the Netherlands. Diabet Med. 2012;29(3):303–12.
Hammer M, Lammert M, Mejías SM, et al. Costs of managing severe hypoglycaemia in three European countries. J Med Econ. 2009;12(4):281–90.
Cryer PE. Hypoglycemia: the limiting factor in the glycemic management of type 1 and type 2 diabetes. Diabetol. 2002;45:937–48.
Frier BM. Hypoglycaemia in diabetes mellitus: epidemiology and clinical implications. Nat Rev Endocrinol. 2014;10(12):711–22.
Jódar-Gimeno E, Alvarez-Guisasola F, Avila-Lachica L, et al. Quality of life and fear for hypoglycaemia in patients with type 2 diabetes mellitus. Rev Clin Esp. 2014. pii:S0014-2565(14)00325-7.
Alvarez Guisasola F, Tofé Povedano S, Krishnarajah G, et al. Hypoglycaemic symptoms, treatment satisfaction, adherence and their associations with glycaemic goal in patients with type 2 diabetes mellitus findings from the Real Life Effectiveness and Care Patterns of Diabetes Management (RECAP-DM) Study. Diab Obesity Metab. 2008;10:25–32.
Williams SA, Shi L, Brenneman SK, et al. The burden of hypoglycemia on healthcare utilization, costs, and quality of life among type 2 diabetes mellitus patients. J Diabetes Complicat. 2012;26(5):399–406.
Brod M, Wolden M, Christensen T, et al. A nine country study of the burden of non-severe nocturnal hypoglycaemic events on diabetes management and daily function. Diabetes Obes Metab. 2013;15(6):546–57.
Pontiroli AE, Miele L, Morabito A. Increase of body weight during the first year of intensive insulin treatment in type 2 diabetes: systematic review and metaanalysis. Diabetes Obes Metab. 2011;13:1008–19.
Eeg-Olofsson K, Cederholm J, Nilsson PM, et al. Risk of cardiovascular disease and mortality in overweight and obese patients with type 2 diabetes: an observational study in 13,087 patients. Diabetologia. 2009;52:65–73.
Philis-Tsimikas A, Charpentier G, Clauson P, et al. Comparison of once-daily insulin detemir with NPH insulin added to a regimen of oral antidiabetic drugs in poorly controlled type 2 diabetes. Clin Ther. 2006;28(10):1569–81.
Canadian Agency for Drugs and Technology in Health. Long-Acting Insulin Analogues for the Treatment of Diabetes Mellitus: Metaanalyses of Clinical Outcomes—Update of CADTH Technology Report No. 92. COMPUS Vol. 2, Issue 1, March 2008. Available from: http://www.cadth.ca/media/pdf/341b_Long-acting-insulin_tr_e.pdf. Accessed Sept 2014.
Ridderstråle M, Jensen MM, Gjesing RP, Niskanen L. Cost-effectiveness of insulin detemir compared with NPH insulin in people with type 2 diabetes in Denmark, Finland, Norway, and Sweden. J Med Econ. 2013;16(4):468–78.
UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes:effects of treatment modalities and their duration. Diabetologia. 2007;50(6):1140–7.
Orozco-Beltrán D, Mezquita-Raya P, Ramírez de Arellano A, et al. Self-reported frequency and impact of hypoglycemic events in Spain. Diabetes Ther. 2014;5(1):155–68.
WHO. ATC/DDD Index 2014. Available at: http://www.whocc.no/atc_ddd_index/. Accessed Jun. 2014.
Nómenclator de Facturación del Ministerio de Sanidad, Servicios Sociales e Igualdad. Available from: http://www.msssi.gob.es/profesionales/nomenclator.do. Accessed Sept 2014.
Consejo General de Colegios Oficiales de Farmacéuticos. Bot Plus Web 2.0. Available at: https://botplusweb.portalfarma.com/. Accessed Sept 2014.
eSalud—Información económica del sector sanitario. Available at: http://www.oblikue.com/bddcostes/. Accessed Sept 2014.
Evans M, Wolden M, Gundgaard J, et al. Cost-effectiveness of insulin degludec compared with insulin glargine for patients with type 2 diabetes treated with basal insulin—from the UK health care cost perspective. Diabetes Obes Metab. 2014;16(4):366–75.
Lee AJ, Morgan CL, Morrissey M, et al. Evaluation of the association between the EQ-5D (health-related utility) and body mass index (obesity) in hospital-treated people with Type 1 diabetes, Type 2 diabetes and with no diagnosed diabetes. Diabet Med. 2005;22(11):1482–6.
Instituto Nacional de Estadística (INE). Altura media de la población por países, sexo, periodo y edad. http://www.ine.es. Accessed Sept 2014.
Sacristán JA, Oliva J, Del Llano J, et al. ¿Qué es una tecnología sanitaria eficiente en España? Gac Sanit. 2002;16(4):334–43.
Bartley PC, Bogoev M, Larsen J, Philotheou A. Long-term efficacy and safety of insulin detemir compared to Neutral Protamine Hagedorn insulin in patients with Type 1 diabetes using a treat-to-target basal-bolus regimen with insulin aspart at meals: a 2-year, randomized, controlled trial. Diabet Med. 2008;25(4):442–9.
Monami M, Marchionni N, Mannucci E. Long-acting insulin analogues versus NPH human insulin in type 2 diabetes: a meta-analysis. Diabetes Res Clin Pract. 2008;81(2):184–9.
Gschwend MH, Aagren M, Valentine WJ. Cost-effectiveness of insulin detemir compared with neutral protamine Hagedorn insulin in patients with type 1 diabetes using a basal-bolus regimen in five European countries. J Med Econ. 2009;12(2):114–23.
Smith-Palmer J, Fajardo-Montañana C, Pollock RF, et al. Long-term cost-effectiveness of insulin detemir versus NPH insulin in type 2 diabetes in Sweden. J Med Econ. 2012;15(5):977–86.
Valentine WJ, Goodall G, Aagren M, et al. Evaluating the cost-effectiveness of therapy conversion to insulin detemir in patients with type 2 diabetes in Germany: a modelling study of long-term clinical and cost outcomes. Adv Ther. 2008;25(6):567–84.
Palmer AJ, Valentine WJ, Ray JA, et al. An economic assessment of analogue basal-bolus insulin versus human basal-bolus insulin in subjects with type 1 diabetes in the UK. Curr Med Res Opin. 2007;23(4):895–901.
Acknowledgments
This study has been funded by Novo Nordisk. The article processing charges for this publication were funded by Novo Nordisk. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
Dr. Cristóbal Morales and Dr. Daniel de Luis acted as expert advisors in the development of the study. They work as independent medical professionals at their corresponding hospital services. Mr. Antonio Ramírez de Arellano Works at NovoNordisk. NovoNordisk sponsored the study. Ms. Maria Giovanna Ferrario and Dr. Luis Lizán work at Outcomes’10 an independent clinical research organization that receives a grant from NovoNordisk to develop the study and to draft the manuscript. All authors contributed in writing the manuscript. The results showed in the manuscript resulted from their freely calculation and interpretation. They declare no conflict of interests.
Compliance with ethics guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Morales, C., de Luis, D., de Arellano, A.R. et al. Cost-Effectiveness Analysis of Insulin Detemir Compared to Neutral Protamine Hagedorn (NPH) in Patients with Type 1 and Type 2 Diabetes Mellitus in Spain. Diabetes Ther 6, 593–610 (2015). https://doi.org/10.1007/s13300-015-0143-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-015-0143-x