Abstract
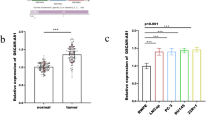
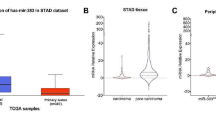
miR-92 has been reported to be upregulated in several human cancers. Until now, its expression pattern and biological roles in human bladder cancer still remains unexplored. The present study aims to clarify its expression, function, and potential molecular mechanisms in bladder cancer. Using real-time PCR, we found that miR-92 was upregulated in bladder cancer tissues compared with normal bladder tissues. We transfected miR-92 mimic and inhibitor in T24 and 5637 bladder cancer cells separately. We found that miR-92 mimic promoted T24 proliferation and invasion, with increased expression of cyclin D1, c-myc, and MMP7 at both mRNA and protein levels. Further investigation found that miR-92 could also promote epithelial-mesenchymal transition by downregulating E-cadherin protein and upregulating vimentin. In addition, miR-92 mimic also promoted activation of Wnt signaling. Meanwhile, miR-92 inhibitor displayed the opposite effects in 5637 cell line. By use of bioinformatic prediction software and luciferase reporter assay, we discovered that GSK3β acted as a direct target of miR-92. Additionally, GSK3β siRNA abrogated the effects of miR-92 mimic on cyclin D1 and MMP7. Moreover, we observed a negative correlation between GSK3β and miR-92 in bladder cancer tissues. In conclusion, our study demonstrated that upregulation of miR-92 is closely related with malignant progression of bladder cancer and miR-92 promotes proliferation, invasion, and Wnt/c-myc/MMP7 signaling by targeting GSK3β.







Similar content being viewed by others
References
Siegel R et al. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29.
Reddy OL et al. Loss of FOXA1 drives sexually dimorphic changes in urothelial differentiation and is an independent predictor of poor prognosis in bladder cancer. Am J Pathol. 2015;185(5):1385–95.
Szarvas T et al. Serum endostatin levels correlate with enhanced extracellular matrix degradation and poor patients’ prognosis in bladder cancer. Int J Cancer. 2012;130(12):2922–9.
Yang GL et al. Increased expression of HMGB1 is associated with poor prognosis in human bladder cancer. J Surg Oncol. 2012;106(1):57–61.
Lin S, Gregory RI. MicroRNA biogenesis pathways in cancer. Nat Rev Cancer. 2015;15(6):321–33.
Lu J et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435(7043):834–8.
Takamizawa J et al. Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res. 2004;64(11):3753–6.
Ota A et al. Identification and characterization of a novel gene, C13orf25, as a target for 13q31-q32 amplification in malignant lymphoma. Cancer Res. 2004;64(9):3087–95.
Hayashita Y et al. A polycistronic microRNA cluster, miR-17-92, is overexpressed in human lung cancers and enhances cell proliferation. Cancer Res. 2005;65(21):9628–32.
Diosdado B et al. MiR-17-92 cluster is associated with 13q gain and c-myc expression during colorectal adenoma to adenocarcinoma progression. Br J Cancer. 2009;101(4):707–14.
He L et al. A microRNA polycistron as a potential human oncogene. Nature. 2005;435(7043):828–33.
Su X et al. An in vivo method to identify microRNA targets not predicted by computation algorithms: p21 targeting by miR-92a in cancer. Cancer Res. 2015;75(14):2875–85.
Wu Q et al. MiR-19b/20a/92a regulates the self-renewal and proliferation of gastric cancer stem cells. J Cell Sci. 2013;126(Pt 18):4220–9.
Ke TW et al. MiR-92a promotes cell metastasis of colorectal cancer through PTEN-mediated PI3K/AKT pathway. Ann Surg Oncol. 2015;22(8):2649–55.
Liu GH et al. Serum miR-21 and miR-92a as biomarkers in the diagnosis and prognosis of colorectal cancer. Tumour Biol. 2013;34(4):2175–81.
Lin HY, Chiang CH, Hung WC. STAT3 upregulates miR-92a to inhibit RECK expression and to promote invasiveness of lung cancer cells. Br J Cancer. 2013;109(3):731–8.
Zhou C et al. miR-92a is upregulated in cervical cancer and promotes cell proliferation and invasion by targeting FBXW7. Biochem Biophys Res Commun. 2015;458(1):63–9.
Ohyagi-Hara C et al. miR-92a inhibits peritoneal dissemination of ovarian cancer cells by inhibiting integrin alpha5 expression. Am J Pathol. 2013;182(5):1876–89.
Chen ZL et al. microRNA-92a promotes lymph node metastasis of human esophageal squamous cell carcinoma via E-cadherin. J Biol Chem. 2011;286(12):10725–34.
Yoshizawa S et al. Downregulated plasma miR-92a levels have clinical impact on multiple myeloma and related disorders. Blood Cancer J. 2012;2(1):e53.
Ohyashiki K et al. Clinical impact of down-regulated plasma miR-92a levels in non-Hodgkin’s lymphoma. PLoS One. 2011;6(2):e16408.
Si H et al. Circulating microRNA-92a and microRNA-21 as novel minimally invasive biomarkers for primary breast cancer. J Cancer Res Clin Oncol. 2013;139(2):223–9.
Nilsson S et al. Downregulation of miR-92a is associated with aggressive breast cancer features and increased tumour macrophage infiltration. PLoS One. 2012;7(4):e36051.
Zhang L et al. microRNAs exhibit high frequency genomic alterations in human cancer. Proc Natl Acad Sci U S A. 2006;103(24):9136–41.
Zhou T et al. Overexpression of miR-92a correlates with tumor metastasis and poor prognosis in patients with colorectal cancer. Int J Color Dis. 2013;28(1):19–24.
Valera VA et al. Regulatory effects of microRNA-92 (miR-92) on VHL gene expression and the hypoxic activation of miR-210 in clear cell renal cell carcinoma. J Cancer. 2011;2:515–26.
Rao E et al. The miRNA-17 approximately 92 cluster mediates chemoresistance and enhances tumor growth in mantle cell lymphoma via PI3K/AKT pathway activation. Leukemia. 2012;26(5):1064–72.
Ghosh AK et al. Aberrant regulation of pVHL levels by microRNA promotes the HIF/VEGF axis in CLL B cells. Blood. 2009;113(22):5568–74.
Shigoka M et al. Deregulation of miR-92a expression is implicated in hepatocellular carcinoma development. Pathol Int. 2010;60(5):351–7.
He G et al. miR-92a/DUSP10/JNK signalling axis promotes human pancreatic cancer cells proliferation. Biomed Pharmacother. 2014;68(1):25–30.
Tsuchida A et al. miR-92 is a key oncogenic component of the miR-17-92 cluster in colon cancer. Cancer Sci. 2011;102(12):2264–71.
Knudsen KE et al. Cyclin D1: polymorphism, aberrant splicing and cancer risk. Oncogene. 2006;25(11):1620–8.
Ratschiller D et al. Cyclin D1 overexpression in bronchial epithelia of patients with lung cancer is associated with smoking and predicts survival. J Clin Oncol. 2003;21(11):2085–93.
Roy PG, Thompson AM. Cyclin D1 and breast cancer. Breast. 2006;15(6):718–27.
Keum JS et al. Cyclin D1 overexpression is an indicator of poor prognosis in resectable non-small cell lung cancer. Br J Cancer. 1999;81(1):127–32.
Wieczorek E et al. MMP7 and MMP8 genetic polymorphisms in bladder cancer patients. Cent European J Urol. 2014;66(4):405–10.
Liu G et al. MiRNA-34a inhibits EGFR-signaling-dependent MMP7 activation in gastric cancer. Tumour Biol. 2014;35(10):9801–6.
Sakamoto N et al. MicroRNA-148a is downregulated in gastric cancer, targets MMP7, and indicates tumor invasiveness and poor prognosis. Cancer Sci. 2014;105(2):236–43.
Dey N et al. Differential activation of Wnt-beta-catenin pathway in triple negative breast cancer increases MMP7 in a PTEN dependent manner. PLoS One. 2013;8(10):e77425.
Schmitz-Drager BJ et al. C-myc in bladder cancer. Clinical findings and analysis of mechanism. Urol Res. 1997;25(Suppl 1):S45–9.
Fan Y et al. Long non-coding RNA UCA1 increases chemoresistance of bladder cancer cells by regulating Wnt signaling. FEBS J. 2014;281(7):1750–8.
Le Bras GF, Taubenslag KJ, Andl CD. The regulation of cell-cell adhesion during epithelial-mesenchymal transition, motility and tumor progression. Cell Adhes Migr. 2012;6(4):365–73.
Zhao J et al. Prognostic significance of the epithelial-to-mesenchymal transition markers e-cadherin, vimentin and twist in bladder cancer. Int Braz J Urol. 2014;40(2):179–89.
Kim MK et al. The differential expression of TGF-beta1, ILK and wnt signaling inducing epithelial to mesenchymal transition in human renal fibrogenesis: an immunohistochemical study. Int J Clin Exp Pathol. 2013;6(9):1747–58.
Zhang Q et al. Wnt/beta-catenin signaling enhances hypoxia-induced epithelial-mesenchymal transition in hepatocellular carcinoma via crosstalk with hif-1alpha signaling. Carcinogenesis. 2013;34(5):962–73.
Chen HC et al. Wnt signaling induces epithelial-mesenchymal transition with proliferation in ARPE-19 cells upon loss of contact inhibition. Lab Investig. 2012;92(5):676–87.
Benelli R et al. The chemopreventive retinoid 4HPR impairs prostate cancer cell migration and invasion by interfering with FAK/AKT/GSK3beta pathway and beta-catenin stability. Mol Cancer. 2010;9:142.
Luo J. Glycogen synthase kinase 3beta (GSK3beta) in tumorigenesis and cancer chemotherapy. Cancer Lett. 2009;273(2):194–200.
Yook JI et al. A Wnt-Axin2-GSK3beta cascade regulates Snail1 activity in breast cancer cells. Nat Cell Biol. 2006;8(12):1398–406.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81260374, 81460384); Yunnan Provincial Department of Education Fund (No. 2014Z072); Joint Project of Science and Technology, Department of Yunnan and Kunming Medical University (Nos. 2014FA015, 2014FZ031); Project of Yunnan Provincial Health Department (No. 2014NS081); and Project of Yunnan Provincial Science and Technology (No. 2015FB196).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This study was conducted with the approval of the ethics committee of Kunming Medical University. Written informed consent was obtained from all patients and all clinical investigation has been conducted.
Conflicts of interests
None.
Additional information
Haifeng Wang and Changxing Ke contributed equally to this work.
Electronic supplementary material
Supplement Figure 1
Prediction results from miRNAorg. miRNAorg showed that GSK3β was a potential target for miR-92. (GIF 83 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Ke, C., Ma, X. et al. MicroRNA-92 promotes invasion and chemoresistance by targeting GSK3β and activating Wnt signaling in bladder cancer cells. Tumor Biol. 37, 16295–16304 (2016). https://doi.org/10.1007/s13277-016-5460-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5460-9