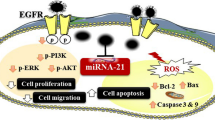
Abstract
Background
Oral tongue squamous cell carcinoma (OTSCC) is a popular aggressive malignancy of the oral cavity. Despite advances in OTSCC therapy, the overall 5-year survival rate is low. The tumor microenvironment resistance factors lead to chemotherapy failure, especially intratumoral hypoxia. HIF-1α, the main protein in hypoxia pathway, influences cell survival and angiogenesis. Hypoxia/HIF-1α system is a potential strategic target in cancer therapeutics. The expression of hypoxia-regulating miRNAs (hypoxamiRs; miR-210 and miR-21), regulators of HIF-1α, is high in OTSCC. Gum Arabic-encapsulated gold nanoparticles (GA-AuNPs) have been reported as promising modality in cancer treatment.
Objective
This study aimed to investigate the influence of GA-AuNPs on the hypoxia regulators in OTSCC (CAL-127 cells). GA-AuNPs cytotoxicity assessed by MTT assay; cell death mode was detected by dual DNA staining; monitoring of cellular hypoxia was followed by pimonidazole; miR-210 and miR-21 expression was assessed by qPCR; and their targets (HIF-1α and c-Myc) assayed by immunocytofluorescence and ELISA, respectively.
Results
GA-AuNPs (75–80 nm; λmax of ~ 540 nm) reduced cell viability with IC50 of 392.3 and 247.3 µg/ml after 24 h and 48 h, respectively. Cell death was mainly due to apoptosis. CAL-27 cells exhibited high hypoxia and the treatment with GA-AuNPs inhibited this hypoxia in a dose-dependent manner, as detected by pimonidazole. GA-AuNPs (30% IC50) significantly reduced miR-210 and miR-21 expression. HIF-1α and c-Myc were inhibited by GA-AuNPs (30% IC50, for 48 h).
Conclusion
The study findings may suggest GA-AuNPs as a promising carrier for chemotherapies to diminish intratumoral hypoxia-stimulated resistance.






Similar content being viewed by others
References
Aebersold DM, Burri P, Beer KT, Laissue J, Djonov V et al (2001) Expression of hypoxia-inducible factor-1alpha: a novel predictive and prognostic parameter in the radiotherapy of oropharyngeal cancer. Cancer Res 61:2911–2916
Almangush A, Heikkinen I, Mäkitie AA, Coletta RD, Läärä E, Leivo I, Salo T (2017) Prognostic biomarkers for oral tongue squamous cell carcinoma: a systematic review and meta-analysis. Br J Cancer 117:856–866. https://doi.org/10.1038/bjc.2017.244
Axiak-Bechtel SM, Upendran A, Lattimer JC, Kelsey J, Cutler CS et al (2014) Gum arabic-coated radioactive gold nanoparticles cause no short-term local or systemic toxicity in the clinically relevant canine model of prostate cancer. Int J Nanomed 9:5001–5011. https://doi.org/10.2147/IJN.S67333
Baskić D, Popović S, Ristić P, Arsenijević NN (2006) Analysis of cycloheximide-induced apoptosis in human leukocytes: fluorescence microscopy using annexin V/propidium iodide versus acridine orange/ethidium bromide. Cell Biol Int 30:924–932. https://doi.org/10.1016/j.cellbi.2006.06.016
Blower PE, Chung JH, Verducci JS, Lin S, Park JK et al (2008) MicroRNAs modulate the chemosensitivity of tumor cells. Mol Cancer Ther 7:1–9. https://doi.org/10.1158/1535-7163.MCT-07-0573
Chen C, Liu R, Wang J, Yan Z, Qian S et al (2015) RNAi knockdown of hypoxia-inducible factor-1α decreased the proliferation, migration, and invasion of hypoxic hepatocellular carcinoma cells. Cell Biochem Biophy 71:1677–1684. https://doi.org/10.1007/s12013-014-0390-x
Cullen BR (2004) Transcription and processing of human microRNA precursors. Mol Cell 16:861–865. https://doi.org/10.1016/j.molcel.2004.12.002
de Carvalho Fraga CA, Alves LR, Marques-Silva L, de Sousa AA, Jorge AS et al (2013) High HIF-1α expression genotypes in oral lichen planus. Clin Oral Inves 17:2011–2015. https://doi.org/10.1007/s00784-013-0920-8
de Souza MG, de Jesus SF, Santos EM, Gomes E, de Paulo SF et al (2020) Radiation therapy reduced blood levels of LDH, HIF-1α, and miR-210 in OSCC. Pathol Oncol Res 26:433–442. https://doi.org/10.1007/s12253-018-0517-2
Dong G, Chen Q, Jiang F, Yu D, Mao Q et al (2016) Diisopropylamine dichloroacetate enhances radiosensitization in esophageal squamous cell carcinoma by increasing mitochondria-derived reactive oxygen species levels. Oncotarget 7:68170–68178. https://doi.org/10.18632/oncotarget.11906
Fasanaro P, D’Alessandra Y, Di Stefano V, Melchionna R, Romani S et al (2008) MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine kinase ligand Ephrin-A3. J Biol Chem 283:15878–15883. https://doi.org/10.1074/jbc.M800731200
Fent GM, Casteel SW, Kim DY, Kannan R, Katti K et al (2009) Biodistribution of maltose and gum arabic hybrid gold nanoparticles after intravenous injection in juvenile swine. Nanomed 5:128–135. https://doi.org/10.1016/j.nano.2009.01.007
Gamal-Eldeen AM, Moustafa D, El-Daly SM, El-Hussieny EA, Saleh S et al (2016) Photothermal therapy mediated by gum Arabic-conjugated gold nanoparticles suppresses liver preneoplastic lesions in mice. J Photochem Photobiol B 163:47–56. https://doi.org/10.1016/j.jphotobiol.2016.08.009
Gamal-Eldeen AM, Moustafa D, El-Daly SM, Abo-Zeid M, Saleh S et al (2017) Gum Arabic-encapsulated gold nanoparticles for a non-invasive photothermal ablation of lung tumor in mice. Biomed Pharmacother 89:1045–1054. https://doi.org/10.1016/j.biopha.2017.03.006
Gao T, Li JZ, Lu Y, Zhang CY, Li Q, Mao J et al (2016) The mechanism between epithelial mesenchymal transition in breast cancer and hypoxia microenvironment. Biomed Pharmacother 80:393–405. https://doi.org/10.1016/j.biopha.2016.02.044
Gee HE, Camps C, Buffa FM, Patiar S, Winter SC et al (2010) hsa-mir-210 is a marker of tumor hypoxia and a prognostic factor in head and neck cancer. Cancer 116:2148–2158. https://doi.org/10.1002/cncr.25009
Golchin K, Golchin J, Ghaderi S, Alidadiani N, Eslamkhah S et al (2018) Gold nanoparticles applications: from artificial enzyme till drug delivery. Artif Cells Nanomed Biotechnol 46:250–254. https://doi.org/10.1080/21691401.2017.1305393
Guimarães TA, Farias LC, Santos ES, de Carvalho Fraga CA, Orsini LA et al (2016) Metformin increases PDH and suppresses HIF-1α under hypoxic conditions and induces cell death in oral squamous cell carcinoma. Oncotarget 7:55057–55068. https://doi.org/10.18632/oncotarget.10842
Hansen MB, Nielsen SE, Berg A (1989) Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Methods 119:203–210. https://doi.org/10.1016/0022-1759(89)90397-9
Höckel M, Vaupel P (2001) Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J Natl Cancer Inst 93:266–276. https://doi.org/10.1093/jnci/93.4.266
Kannaiyan R, Shanmugam MK, Sethi G (2011) Molecular targets of celastrol derived from Thunder of God Vine: potential role in the treatment of inflammatory disorders and cancer. Cancer Lett 303:9–20. https://doi.org/10.1016/j.canlet.2010.10.025
Kattumuri V, Katti K, Bhaskaran S, Boote EJ, Casteel SW et al (2007) Gum Arabic as a phytochemical construct for the stabilization of gold nanoparticles: in vivo pharmacokinetics and X-ray-contrast-imaging studies. Small 3:333–341. https://doi.org/10.1002/smll.200600427
Koukourakis MI, Giatromanolaki A, Skarlatos J, Corti L, Blandamura S et al (2001) Hypoxia inducible factor (HIF-1a and HIF-2a) expression in early esophageal cancer and response to photodynamic therapy and radiotherapy. Cancer Res 61:1830–1832
Krichevsky AM, Gabriely G (2009) miR-21: a small multi-faceted RNA. J Cell Mol Med 13:39–53. https://doi.org/10.1111/j.1582-4934.2008.00556.x
Li J, Huang H, Sun L, Yang M, Pan C et al (2009) MiR-21 indicates poor prognosis in tongue squamous cell carcinomas as an apoptosis inhibitor. Clin Cancer Res 15:3998–4008. https://doi.org/10.1158/1078-0432.CCR-08-3053
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu Y, Li Y, Wang Z, Xie S, Wang Q et al (2019) Downregulation of RGMA by HIF-1A/miR-210-3p axis promotes cell proliferation in oral squamous cell carcinoma. Biomed Pharmacother 112:108608. https://doi.org/10.1016/j.biopha.2019.108608
Pang X, Yi Z, Zhang J, Lu B, Sung B et al (2010) Celastrol suppresses angiogenesis-mediated tumor growth through inhibition of AKT/mammalian target of rapamycin pathway. Canc Res 70:1951–1959. https://doi.org/10.1158/0008-5472.CAN-09-3201
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S et al (2003) Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 3:347–361
Pérez-Sayáns M, Somoza-Martín JM, Barros-Angueira F, Reboiras-López MD, Gándara Rey JM et al (2009) Genetic and molecular alterations associated with oral squamous cell cancer (Review). Oncol Rep 22:1277–1282. https://doi.org/10.3892/or_00000565
Pickering CR, Zhang J, Yoo SY, Bengtsson L, Moorthy S et al (2013) Integrative genomic characterization of oral squamous cell carcinoma identifies frequent somatic drivers. Cancer Discov 3:770–781. https://doi.org/10.1158/2159-8290.CD-12-0537
Pugh CW, Ratcliffe PJ (2003) Regulation of angiogenesis by hypoxia: role of the HIF system. Nat Med 9:677–684. https://doi.org/10.1038/nm0603-677
Puisségur MP, Mazure NM, Bertero T, Pradelli L, Grosso S et al (2011) miR-210 is overexpressed in late stages of lung cancer and mediates mitochondrial alterations associated with modulation of HIF-1 activity. Cell Death Diff 18:465–478. https://doi.org/10.1038/cdd.2010.119
Sano D, Myers JN (2007) Metastasis of squamous cell carcinoma of the oral tongue. Canc Metastasis Rev 26(3–4):645–662. https://doi.org/10.1007/s10555-007-9082-y
Selcuklu SD, Donoghue MT, Spillane C (2009) miR-21 as a key regulator of oncogenic processes. Biochem Soc Trans 37:918–925. https://doi.org/10.1042/BST0370918
Semenza GL (1999) Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Ann Rev Cell Dev Biol 15:551–578. https://doi.org/10.1146/annurev.cellbio.15.1.551
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. Br J Cancer 69:7–34. https://doi.org/10.3322/caac.21551
Talks KL, Turley H, Gatter KC, Maxwell PH, Pugh CW et al (2000) The expression and distribution of the hypoxia-inducible factors HIF-1alpha and HIF-2alpha in normal human tissues, cancers, and tumor-associated macrophages. Am J Pathol 157:411–421. https://doi.org/10.1016/s0002-9440(10)64554-3
van Dijk BA, Brands MT, Geurts SM, Merkx MA, Roodenburg JL (2016) Trends in oral cavity cancer incidence, mortality, survival and treatment in the Netherlands. Int J Cancer 139:574–583. https://doi.org/10.1002/ijc.30107
Varol N, Konac E, Gurocak OS, Sozen S (2011) The realm of microRNAs in cancers. Mol Biol Rep 38:1079–1089. https://doi.org/10.1007/s11033-010-0205-0
Warnakulasuriya S (2009) Global epidemiology of oral and oropharyngeal cancer. Oral Oncol 45(4–5):309–316. https://doi.org/10.1016/j.oraloncology.2008.06.002
Wong TS, Liu XB, Wong BY, Ng RW, Yuen AP, Wei WI (2008) Mature miR-184 as potential oncogenic microRNA of squamous cell carcinoma of tongue. Clin Cancer Res 14:2588–2592. https://doi.org/10.1158/1078-0432.CCR-07-0666
Wu BH, Xiong XP, Jia J, Zhang WF (2011) MicroRNAs: new actors in the oral cancer scene. Oral Oncol 47:314–319. https://doi.org/10.1016/j.oraloncology.2011.03.019
Yang X, Zhu H, Ge Y, Liu J, Cai J et al (2014) Melittin enhances radiosensitivity of hypoxic head and neck squamous cell carcinoma by suppressing HIF-1α. Tumour Biol 35:10443–10448. https://doi.org/10.1007/s13277-014-2218-0
Ye H, Yu T, Temam S, Ziober BL, Wang J, Schwartz JL, Mao L, Wong DT, Zhou X (2008) Transcriptomic dissection of tongue squamous cell carcinoma. BMC Genomics 9:69. https://doi.org/10.1186/1471-2164-9-69
Ye H, Yu T, Temam S, Ziober BL, Wang J, Schwartz JL et al (2010) MicroRNAs contribute to the chemoresistance of cisplatin in tongue squamous cell carcinoma lines. Oral Oncol 46:317–322. https://doi.org/10.3349/ymj.2017.58.3.489
Yu T, Tang B, Sun X (2017) Development of inhibitors targeting hypoxia-inducible factor 1 and 2 for cancer therapy. Yonsei Med J 58:489–496. https://doi.org/10.3349/ymj.2017.58.3.489
Zhu S, Zhu L, Yu J, Wang Y, Peng B (2019) Anti-osteoclastogenic effect of epigallocatechin gallate-functionalized gold nanoparticles in vitro and in vivo. Int J Nanomed 14:5017–5032. https://doi.org/10.2147/IJN.S204628
Acknowledgements
This work was supported by Taif University Researchers Supporting Project Number (TURSP-2020/103).
Author information
Authors and Affiliations
Contributions
AMG-E designed the study, carried out the molecular biology experiments, and wrote the manuscript; HMB reviewed the manuscript; NSA reviewed the manuscript; EMI contributed in the cell biology part; WFA carried out the fluorescence microscopy analysis; FA contributed in cell biology part and statistics; and BMR contributed in nanoparticles preparation and characterization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Ethical approval is not applicable.
Rights and permissions
About this article
Cite this article
Gamal-Eldeen, A.M., Baghdadi, H.M., Afifi, N.S. et al. Gum arabic-encapsulated gold nanoparticles modulate hypoxamiRs expression in tongue squamous cell carcinoma. Mol. Cell. Toxicol. 17, 111–121 (2021). https://doi.org/10.1007/s13273-021-00117-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-021-00117-w