Abstract
Background
Transglutaminase 2 (TG2) mediates protein modifications by crosslinking or by incorporating polyamine in response to oxidative or DNA-damaging stress, thereby regulating apoptosis, extracellular matrix formation, and inflammation. The regulation of transcriptional activity by TG2-mediated histone serotonylation or by Sp1 crosslinking may also contribute to cellular stress responses.
Objective
In this study, we attempted to identify TG2-interacting proteins to better understand the role of TG2 in transcriptional regulation.
Methods
Using a yeast two-hybrid assay to screen a HeLa cell cDNA library, we found that TG2 bound BAF250a, a core subunit of the cBAF chromatin remodeling complex, through an interaction between the TG2 barrel 1 and BAF250a C-terminal domains.
Results
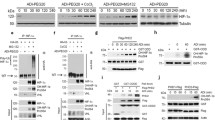
TG2 was pulled down with a GST-BAF250a C-term fusion protein. Moreover, TG2 and BAF250a were co-fractionated using P11 chromatography, and co-immunoprecipitated. A transamidation reaction showed that TG2 mediated incorporation of polyamine into BAF250a. In glucocorticoid response-element reporter-expressing cells, TG2 overexpression increased the luciferase reporter activity in a transamidation-dependent manner. In addition, a comparison of genome-wide gene expression between wild-type and TG2-deficient primary hepatocytes in response to dexamethasone treatment showed that TG2 further enhanced or suppressed the expression of dexamethasone-regulated genes that were identified by a gene ontology enrichment analysis.
Conclusion
Thus, our results indicate that TG2 regulates transcriptional activity through BAF250a polyamination.



Similar content being viewed by others
References
Aeschlimann D et al (1998) Isolation of a cDNA encoding a novel member of the transglutaminase gene family from human keratinocytes. Detection and identification of transglutaminase gene products based on reverse transcription-polymerase chain reaction with degenerate primers. J Biol Chem 273:3452–3460. https://doi.org/10.1074/jbc.273.6.3452
Ai L et al (2008) The transglutaminase 2 gene (TGM2), a potential molecular marker for chemotherapeutic drug sensitivity, is epigenetically silenced in breast cancer. Carcinogenesis 29:510–518. https://doi.org/10.1093/carcin/bgm280
Cho SY et al (2020) Transglutaminase 2 mediates hypoxia-induced selective mRNA translation via polyamination of 4EBPs. Life Sci Alliance. https://doi.org/10.26508/lsa.201900565
Choi K et al (2005) Chemistry and biology of dihydroisoxazole derivatives: selective inhibitors of human transglutaminase 2. Chem Biol 12:469–475. https://doi.org/10.1016/j.chembiol.2005.02.007
De Laurenzi V, Melino G (2001) Gene disruption of tissue transglutaminase. Mol Cell Biol 21:148–155. https://doi.org/10.1128/mcb.21.1.148-155.2001
Farrelly LA et al (2019) Histone serotonylation is a permissive modification that enhances TFIID binding to H3K4me3. Nature 567:535–539. https://doi.org/10.1038/s41586-019-1024-7
Fryer CJ, Archer TK (1998) Chromatin remodelling by the glucocorticoid receptor requires the BRG1 complex. Nature 393:88–91. https://doi.org/10.1038/30032
Jang GY et al (2010) Transglutaminase 2 suppresses apoptosis by modulating caspase 3 and NF-kappaB activity in hypoxic tumor cells. Oncogene 29:356–367. https://doi.org/10.1038/onc.2009.342
Jeon JH et al (2003a) Transglutaminase 2 inhibits Rb binding of human papillomavirus E7 by incorporating polyamine. EMBO J 22:5273–5282. https://doi.org/10.1093/emboj/cdg495
Jeon JH et al (2003b) Differential incorporation of biotinylated polyamines by transglutaminase 2. FEBS Lett 534:180–184. https://doi.org/10.1016/s0014-5793(02)03836-x
Jeon BH, Yoo YM, Jung EM, Jeung EB (2020) Dexamethasone treatment increases the intracellular calcium level through TRPV6 in A549 cells. Int J Mol Sci. https://doi.org/10.3390/ijms21031050
Kozmik Z et al (2001) Characterization of mammalian orthologues of the Drosophila osa gene: cDNA cloning, expression, chromosomal localization, and direct physical interaction with Brahma chromatin-remodeling complex. Genomics 73:140–148. https://doi.org/10.1006/geno.2001.6477
Lee SM et al (2012) Cysteamine prevents the development of lens opacity in a rat model of selenite-induced cataract. Investig Ophthalmol Vis Sci 53:1452–1459. https://doi.org/10.1167/iovs.11-8636
Lee JH et al (2014) Endoplasmic reticulum stress activates transglutaminase 2 leading to protein aggregation. Int J Mol Med 33:849–855. https://doi.org/10.3892/ijmm.2014.1640
Lee SJ et al (2017) Transglutaminase 2 mediates UV-induced skin inflammation by enhancing inflammatory cytokine production. Cell Death Dis 8:e3148. https://doi.org/10.1038/cddis.2017.550
Masliah-Planchon J et al (2015) SWI/SNF chromatin remodeling and human malignancies. Annu Rev Pathol 10:145–171. https://doi.org/10.1146/annurev-pathol-012414-040445
Mathur R, Roberts CWM (2018) SWI/SNF (BAF) complexes: guardians of the epigenome. Annu Rev Cancer Biol 2:413–427. https://doi.org/10.1146/annurev-cancerbio-030617-050151
McConoughey SJ et al (2010) Inhibition of transglutaminase 2 mitigates transcriptional dysregulation in models of Huntington disease. EMBO Mol Med 2:349–370. https://doi.org/10.1002/emmm.201000084
Mehta K, Kumar A, Kim HI (2010) Transglutaminase 2: a multi-tasking protein in the complex circuitry of inflammation and cancer. Biochem Pharmacol 80:1921–1929. https://doi.org/10.1016/j.bcp.2010.06.029
Mittal P, Roberts CWM (2020) The SWI/SNF complex in cancer - biology, biomarkers and therapy. Nat Rev Clin Oncol 17:435–448. https://doi.org/10.1038/s41571-020-0357-3
Muratcioglu S et al (2015) Structural Modeling of GR Interactions with the SWI/SNF Chromatin Remodeling Complex and C/EBP. Biophys J 109:1227–1239. https://doi.org/10.1016/j.bpj.2015.06.044
Myneni VD, Melino G, Kaartinen MT (2015) Transglutaminase 2–a novel inhibitor of adipogenesis. Cell Death Dis 6:e1868. https://doi.org/10.1038/cddis.2015.238
Nanda N et al (2001) Targeted inactivation of Gh/tissue transglutaminase II. J Biol Chem 276:20673–20678. https://doi.org/10.1074/jbc.M010846200
Nie Z et al (2000) A specificity and targeting subunit of a human SWI/SNF family-related chromatin-remodeling complex. Mol Cell Biol 20:8879–8888. https://doi.org/10.1128/mcb.20.23.8879-8888.2000
Olsen KC et al (2011) Transglutaminase 2 and its role in pulmonary fibrosis. Am J Respir Crit Care Med 184:699–707. https://doi.org/10.1164/rccm.201101-0013OC
Sandhya S et al (2018) Domain architecture of BAF250a reveals the ARID and ARM-repeat domains with implication in function and assembly of the BAF remodeling complex. PLoS ONE 13:e0205267. https://doi.org/10.1371/journal.pone.0205267
Shin DM et al (2004) Cell type-specific activation of intracellular transglutaminase 2 by oxidative stress or ultraviolet irradiation: implications of transglutaminase 2 in age-related cataractogenesis. J Biol Chem 279:15032–15039. https://doi.org/10.1074/jbc.M308734200
Shin DM et al (2008) Cystamine prevents ischemia-reperfusion injury by inhibiting polyamination of RhoA. Biochem Biophys Res Commun 365:509–514. https://doi.org/10.1016/j.bbrc.2007.11.007
Shin JW et al (2020) Keratinocyte transglutaminase 2 promotes CCR6(+) γδT-cell recruitment by upregulating CCL20 in psoriatic inflammation. Cell Death Dis 11:301. https://doi.org/10.1038/s41419-020-2495-z
Szondy Z et al (2003) Transglutaminase 2-/- mice reveal a phagocytosis-associated crosstalk between macrophages and apoptotic cells. Proc Natl Acad Sci USA 100:7812–7817. https://doi.org/10.1073/pnas.0832466100
Tang L, Nogales E, Ciferri C (2010) Structure and function of SWI/SNF chromatin remodeling complexes and mechanistic implications for transcription. Prog Biophys Mol Biol 102:122–128. https://doi.org/10.1016/j.pbiomolbio.2010.05.001
Tatsukawa H et al (2009) Role of transglutaminase 2 in liver injury via cross-linking and silencing of transcription factor Sp1. Gastroenterology 136:1783-1795.e1710. https://doi.org/10.1053/j.gastro.2009.01.007
Tatsukawa H et al (2016) Transglutaminase 2 has opposing roles in the regulation of cellular functions as well as cell growth and death. Cell Death Dis 7:e2244. https://doi.org/10.1038/cddis.2016.150
Tordjmann T et al (1997) An improved digitonin-collagenase perfusion technique for the isolation of periportal and perivenous hepatocytes from a single rat liver: physiological implications for lobular heterogeneity. Hepatology (Baltimore, MD) 26:1592–1599. https://doi.org/10.1053/jhep.1997.v26.pm0009398003
Verderio EA, Johnson TS, Griffin M (2005) Transglutaminases in wound healing and inflammation. Prog Exp Tumor Res 38:89–114. https://doi.org/10.1159/000084235
Vihervaara A, Duarte FM, Lis JT (2018) Molecular mechanisms driving transcriptional stress responses. Nat Rev Genet 19:385–397. https://doi.org/10.1038/s41576-018-0001-6
Zhang J, Lesort M, Guttmann RP, Johnson GV (1998) Modulation of the in situ activity of tissue transglutaminase by calcium and GTP. J Biol Chem 273:2288–2295. https://doi.org/10.1074/jbc.273.4.2288
Acknowledgements
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (grant No. 2012R1A1A2005188 and 2017R1C1B2002183); and the Bio & Medical Technology Development Program of the NRF funded by the Ministry of Science & ICT (grant No. 2018M3A9F3056902).
Author information
Authors and Affiliations
Contributions
JHJ and IGK conceived and designed the experiments. HJK, JHL and SYC performed the experiments and collected the data. IGK wrote the manuscript with assistance and final approval by all the coauthors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, HJ., Lee, JH., Cho, SY. et al. Transglutaminase 2 mediates transcriptional regulation through BAF250a polyamination. Genes Genom 43, 333–342 (2021). https://doi.org/10.1007/s13258-021-01055-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-021-01055-6