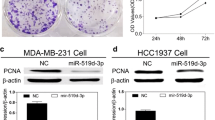
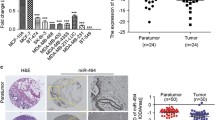
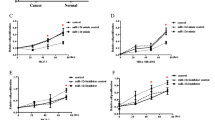
Abstract
Breast cancer is the most common malignant disease amongst women. miRNAs are small, non-coding RNAs that regulate gene expression, thus have the potential to play an important role during cancer development. Emerging evidence shows that miR-135a is down-regulated in breast cancer cells, but the functional roles of miR-135a in breast cancer cells remains unexplored. For this purpose, we investigated the expression of miR-135a in breast cancer cells and explored its functional role during breast cancer progression. In vitro study showed that miR-135a may be a novel tumor suppressor. Further studies showed that transcription factors ELK1 and ELK3 are direct target genes of miR-135a that modulates the suppressive function of miR-135a in breast cancer cells. Induced expression of miR-135a significantly downregulated the expression of ELK1 and ELK3 both at mRNA and protein levels. Furthermore, the effect of miR-135a in MCF-7 and T47D cells was investigated by the overexpression of miR-135a mimics. In vitro, induced expression of miR-135a in breast cancer cells inhibited cell Proliferation and clongenicity. Moreover, a luciferase activity assay revealed that miR-135a could directly target the 3′-untranslated region (3′ UTRS) of ELK1 and ELK3 oncogenes. In addition, rescue experiment demonstrated that the promoted cell growth by transcription factors ELK1 and ELK3 was attenuated by the over-expression of miR-135a. Our study demonstrates that miR-135a regulates cell proliferation in breast cancer by targeting ELK1 and ELK3 oncogenes, and suggests that miR-135a potentially can act as a tumor suppressor.






Similar content being viewed by others
References
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. cell 136:215–233
Boros J, Donaldson IJ, O’Donnell A, Odrowaz ZA, Zeef L, Lupien M, Meyer CA, Liu XS, Brown M, Sharrocks AD (2009) Elucidation of the ELK1 target gene network reveals a role in the coordinate regulation of core components of the gene regulation machinery. Genome Res 19:1963–1973
Calin GA, Croce CM (2006) microRNA-cancer connection: the beginning of a new tale. Cancer Res 66:7390–7394
Colozza M, Cardoso F, Sotiriou C, Larsimont D, Piccart MJ (2005) Bringing molecular prognosis and prediction to the clinic. Clin Breast Cancer 6:61–76
Dumont N, Tlsty TD (2009) Reflections on miR-ing effects in metastasis. Cancer cell 16:3–4
Enright AJ, John B, Gaul U, Tuschl T, Sander C, Marks DS (2003) MicroRNA targets in Drosophila. Genome Biol 5:R1
Giovane A, Pintzas A, Maira SM, Sobieszczuk P, Wasylyk B (1994) Net, a new ets transcription factor that is activated by Ras. Genes Dev 8:1502–1513
Hipskind R, Roa V, Muller C, Raddy E, Nordheim A (1991) Ets-related protein Elk-1 is homologous to the c-fos regulatory factor p62TCF. Nature 354:531–534
Iorio MV, Visone R, Di Leva G, Donati V, Petrocca F, Casalini P, Taccioli C, Volinia S, Liu C-G, Alder H (2007) microRNA signatures in human ovarian cancer. Cancer Res 67:8699–8707
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120:15–20
Liu S, Guo W, Shi J, Li N, Yu X, Xue J, Fu X, Chu K, Lu C, Zhao J (2012) microRNA-135a contributes to the development of portal vein tumor thrombus by promoting metastasis in hepatocellular carcinoma. J Hepatol 56:389–396
Mao XP, Zhang LS, Huang B, Zhou SY, Liao J, Chen LW, Qiu SP, Chen JX (2015) Mir-135a enhances cellular proliferation through post-transcriptionally regulating PHLPP2 and FOXO1 in human bladder cancer. J Transl Med 13:86
Migliore C, Petrelli A, Ghiso E, Corso S, Capparuccia L, Eramo A, Comoglio PM, Giordano S (2008) microRNAs impair MET-mediated invasive growth. Cancer Res 68:10128–10136
Nagel R, le Sage C, Diosdado B, van der Waal M, Vrielink JAO, Bolijn A, Meijer GA, Agami R (2008) Regulation of the adenomatous polyposis coli gene by the miR-135 family in colorectal cancer. Cancer Res 68:5795–5802
Navarro A, Diaz T, Martinez A, Gaya A, Pons A, Gel B, Codony C, Ferrer G, Martinez C, Montserrat E (2009) Regulation of JAK2 by miR-135a: prognostic impact in classic Hodgkin lymphoma. Blood 114:2945–2951
Nelson KM, Weiss GJ (2008) microRNAs and cancer: past, present, and potential future. Mol Cancer Ther 7:3655–3660
Pantel K, Alix-Panabières C, Riethdorf S (2009) Cancer micrometastases. Nat Rev Clin Oncol 6:339–351
Ren JW, Li ZJ, Tu C (2015) MiR-135 post-transcriptionally regulates FOXO1 expression and promotes cell proliferation in human malignant melanoma cells. Int J Clin Exp Pathol 8:6356–6366
Sharrocks AD (2002) Complexities in ETS-domain transcription factor function and regulation: lessons from the TCF (ternary complex factor) subfamily. Biochem Soc Trans 30:1–9
Shi H, Ji Y, Zhang D, Liu Y, Fang P (2015) MiR-135a inhibits migration and invasion and regulates EMT-related marker genes by targeting KLF8 in lung cancer cells. Biochem Biophys Res Commun 465:125–130
Tang WW, Wan GP, Wan YC, Zhang L, Cheng WJ (2013) Effects of miR-135a on HOXA10 expression, proliferation and apoptosis of ovarian cancer cells. Zhonghua Fu Chan Ke Za Zhi 48:364–369
Tang W, Jiang Y, Mu X, Xu L, Cheng W, Wang X (2014) MiR-135a functions as a tumor suppressor in epithelial ovarian cancer and regulates HOXA10 expression. Cell Signal 26:1420–1426
Wu H, Huang M, Cao P, Wang T, Shu Y, Liu P (2012a) MiR-135a targets JAK2 and inhibits gastric cancer cell proliferation. Cancer Biol Ther 13:281–288
Wu S, Lin Y, Xu D, Chen J, Shu M, Zhou Y, Zhu W, Su X, Qiu P, Yan G (2012b) MiR-135a functions as a selective killer of malignant glioma. Oncogene 31:3866
Yamada Y, Hidaka H, Seki N, Yoshino H, Yamasaki T, Itesako T, Nakagawa M, Enokida H (2013) Tumor-suppressive microRNA-135a inhibits cancer cell proliferation by targeting the c-MYC oncogene in renal cell carcinoma. Cancer Sci 104:304–312
Zhang L, Huang J, Yang N, Greshock J, Megraw MS, Giannakakis A, Liang S, Naylor TL, Barchetti A, Ward MR (2006) microRNAs exhibit high frequency genomic alterations in human cancer. Proc Natl Acad Sci USA 103:9136–9141
Acknowledgements
This work was supported by The National Key Scientific Programme of China (2016YFC1302305), The National Natural Science Foundation of China (81672609, 81472494, 81502282, 31671299). The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. Akhlaq Ahmad wishes to thank CAS-TWAS presidents PhD fellowship for the advancement of science in developing countries.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Akhlaq Ahmad, Weijie Zhang, Mingming Wu, Sheng Tan and Tao Zhu declare that they have not any conflict of interest.
Ethical approval
This article does not contain any studies with human subjects or animals performed by any of the authors. All the experimental procedures were approved by Scientific Ethics Committee and Review Board of the School of Life Sciences, University of Science and Technology of China.
Rights and permissions
About this article
Cite this article
Ahmad, A., Zhang, W., Wu, M. et al. Tumor-suppressive miRNA-135a inhibits breast cancer cell proliferation by targeting ELK1 and ELK3 oncogenes. Genes Genom 40, 243–251 (2018). https://doi.org/10.1007/s13258-017-0624-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-017-0624-6