Abstract
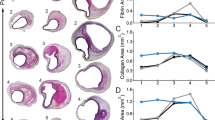
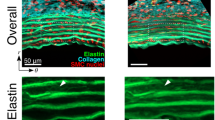
Atherosclerosis is a major risk factor for cardiovascular disease. However, mechanisms of interaction of atherosclerotic plaque development and local stiffness of the lamellar structure of the arterial wall are not well established. In the current study, the local Young’s modulus of the wall and plaque components were determined for three different groups of healthy, mildly diseased and advanced atherosclerotic human abdominal aortas. Histological staining was performed to highlight the atherosclerotic plaque components and lamellar structure of the aortic media, consisting of concentric layers of elastin and interlamellar zones. The force spectroscopy mode of the atomic force microscopy was utilized to determine Young’s moduli of aortic wall lamellae and plaque components at the micron level. The high variability of Young’s moduli (E) at different locations of the atherosclerotic plaque such as the fibrous cap (E = 15.5± 2.6 kPa), calcification zone (E = 103.7±19.5 kPa), and lipid pool (E = 3.5±1.2 kPa) were observed. Reduction of elastin lamellae stiffness (18.6%), as well as stiffening of interlamellar zones (50%), were detected in the diseased portion of the medial layer of abdominal aortic wall compared to the healthy artery. Additionally, significant differences in the stiffness of both elastin lamellae and interlamellar zones were observed between the diseased wall and disease-free wall in incomplete plaques. Our results elucidate the alternation of the stiffness of different lamellae in the human abdominal aortic wall with atherosclerotic plaque development and may provide new insight on the remodeling of the aortic wall during the progression of atherosclerosis.




Similar content being viewed by others
References
Akhtar, R. In vitro characterisation of arterial stiffening: from the macro-to the nano-scale. Artery Res. 8(1):1–8, 2014.
Akhtar, R., H. Graham, B. Derby, M. Sherratt, A. Trafford, R. Chadwick, et al. Frequency-modulated atomic force microscopy localises viscoelastic remodelling in the ageing sheep aorta. J. Mech. Behav. Biomed. Mater. 64:10–17, 2016.
Akyildiz, A. C., L. Speelman, and F. J. Gijsen. Mechanical properties of human atherosclerotic intima tissue. J. Biomech. 47(4):773–783, 2014.
Apostolakis, I. Z., S. D. Nandlall, and E. E. Konofagou. Piecewise pulse wave imaging (pPWI) for detection and monitoring of focal vascular disease in murine aortas and carotids in vivo. IEEE Trans. Med. Imaging 35(1):13–28, 2016.
Avolio, A. Arterial stiffness. Pulse 1(1):14–28, 2013.
Avolio, A., D. Jones, and M. Tafazzoli-Shadpour. Quantification of alterations in structure and function of elastin in the arterial media. Hypertension 32(1):170–175, 1998.
Brüel, A., G. Ørtoft, and H. Oxlund. Inhibition of cross-links in collagen is associated with reduced stiffness of the aorta in young rats. Atherosclerosis. 140(1):135–145, 1998.
Buchanan, J., C. Kleinstreuer, S. Hyun, and G. Truskey. Hemodynamics simulation and identification of susceptible sites of atherosclerotic lesion formation in a model abdominal aorta. J. Biomech. 36(8):1185–1196, 2003.
Burke, A. P., and F. Tavora. Practical cardiovascular pathology. Philadelphia: Lippincott Williams & Wilkins, 2010.
Butt, H.-J., B. Cappella, and M. Kappl. Force measurements with the atomic force microscope: technique, interpretation and applications. Surf. Sci. Rep. 59(1–6):1–152, 2005.
Cattell, M. A., J. C. Anderson, and P. S. Hasleton. Age-related changes in amounts and concentrations of collagen and elastin in normotensive human thoracic aorta. Clin. Chim. Acta 245(1):73–84, 1996.
Chai, C.-K., L. Speelman, C. W. Oomens, and F. P. Baaijens. Compressive mechanical properties of atherosclerotic plaques—indentation test to characterise the local anisotropic behaviour. J. Biomech. 47(4):784–792, 2014.
Discher, D. E., P. Janmey, and Y.-L. Wang. Tissue cells feel and respond to the stiffness of their substrate. Science 310(5751):1139–1143, 2005.
Doran, A. C., N. Meller, and C. A. McNamara. Role of smooth muscle cells in the initiation and early progression of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 28(5):812–819, 2008.
Ebenstein, D. M., D. Coughlin, J. Chapman, C. Li, and L. A. Pruitt. Nanomechanical properties of calcification, fibrous tissue, and hematoma from atherosclerotic plaques. J. Biomed. Mater. Res. Part A 91(4):1028–1037, 2009.
Ethier, C. R., and C. A. Simmons. Introductory biomechanics: from cells to organisms. Cambridge: Cambridge University Press, 2007.
Fischer, A. H., K. A. Jacobson, J. Rose, and R. Zeller. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harb. Protoc. 2008. https://doi.org/10.1101/pdb.prot4986.
Fukui, T., T. Matsumoto, T. Tanaka, T. Ohashi, K. Kumagai, H. Akimoto, et al. In vivo mechanical properties of thoracic aortic aneurysmal wall estimated from in vitro biaxial tensile test. Biomed. Mater. Eng. 15(4):295–305, 2004.
Glagov, S., D. Rowley, and R. Kohut. Atherosclerosis of human aorta and its coronary and renal arteries. A consideration of some hemodynamic factors which may be related to the marked differences in atherosclerotic involvement of the coronary and renal arteries. Arch Pathol. 72:558, 1961.
Haghighipour, N., M. Tafazzoli-Shadpour, and A. Avolio. Residual stress distribution in a lamellar model of the arterial wall. J. Med. Eng. Technol. 34(7–8):422–428, 2010.
Hayenga, H., A. Trache, J. Trzeciakowski, and J. Humphrey. Regional atherosclerotic plaque properties in ApoE−/− mice quantified by atomic force, immunofluorescence, and light microscopy. J. Vasc. Res. 48(6):495–504, 2011.
Holzapfel, G. A., G. Sommer, and P. Regitnig. Anisotropic mechanical properties of tissue components in human atherosclerotic plaques. J. Biomech. Eng. 126(5):657–665, 2004.
Hutter, J. L., and J. Bechhoefer. Calibration of atomic-force microscope tips. Rev. Sci. Instrum. 64(7):1868–1873, 1993.
Huynh, J., N. Nishimura, K. Rana, J. M. Peloquin, J. P. Califano, C. R. Montague, et al. Age-related intimal stiffening enhances endothelial permeability and leukocyte transmigration. Sci. Transl. Med. 3(112):112ra122, 2011.
Imura, T., K. Yamamoto, T. Satoh, T. Mikami, and H. Yasuda. Arteriosclerotic change in the human abdominal aorta in vivo in relation to coronary heart disease and risk factors. Atherosclerosis. 73(2):149–155, 1988.
Kamenskiy, A. V., Y. A. Dzenis, S. A. J. Kazmi, M. A. Pemberton, I. I. Pipinos, N. Y. Phillips, et al. Biaxial mechanical properties of the human thoracic and abdominal aorta, common carotid, subclavian, renal and common iliac arteries. Biomech. Model. Mechanobiol. 13(6):1341–1359, 2014.
Kasas, S., G. Longo, and G. Dietler. Mechanical properties of biological specimens explored by atomic force microscopy. J. Phys. D Appl. Phys. 46(13):133001, 2013.
Korshunov, V. A., S. M. Schwartz, and B. C. Berk. Vascular remodeling. Arterioscler. Thromb. Vasc. Biol. 27(8):1722–1728, 2007.
Last, J. A., S. J. Liliensiek, P. F. Nealey, and C. J. Murphy. Determining the mechanical properties of human corneal basement membranes with atomic force microscopy. J. Struct. Biol. 167(1):19–24, 2009.
Lekka, M., D. Gil, K. Pogoda, J. Dulińska-Litewka, R. Jach, J. Gostek, et al. Cancer cell detection in tissue sections using AFM. Arch. Biochem. Biophys. 518(2):151–156, 2012.
Lundkvist, A., E. Lilleodden, W. Siekhaus, J. Kinney, L. Pruitt, and M. Balooch. Viscoelastic properties of healthy human artery measured in saline solution by AFM-based indentation technique. MRS Online Proc. Library Arch. 436:353, 1996.
Maher, E., A. Creane, S. Sultan, N. Hynes, C. Lally, and D. J. Kelly. Tensile and compressive properties of fresh human carotid atherosclerotic plaques. J. Biomech. 42(16):2760–2767, 2009.
Matsumoto, T., T. Goto, T. Furukawa, and M. Sato. Residual stress and strain in the lamellar unit of the porcine aorta: experiment and analysis. J. Biomech. 37(6):807–815, 2004.
Matsumoto, T., and K. Hayashi. Mechanical and dimensional adaptation of rat aorta to hypertension. J. Biomech. Eng. 116(3):278–283, 1994.
McKee, C. T., J. A. Last, P. Russell, and C. J. Murphy. Indentation versus tensile measurements of Young’s modulus for soft biological tissues. Tissue Eng. Part B Rev. 17(3):155–164, 2011.
Murata, K., T. Motayama, and C. Kotake. Collagen types in various layers of the human aorta and their changes with the atherosclerotic process. Atherosclerosis 60(3):251–262, 1986.
Ocallaghan, C. J., and B. Williams. Mechanical strain–induced extracellular matrix production by human vascular smooth muscle cells. Hypertension 36(3):319–324, 2000.
O’Leary, S. A., J. J. Mulvihill, H. E. Barrett, E. G. Kavanagh, M. T. Walsh, T. M. McGloughlin, et al. Determining the influence of calcification on the failure properties of abdominal aortic aneurysm (AAA) tissue. J. Mech. Behav. Biomed. Mater. 42:154–167, 2015.
Peloquin, J., J. Huynh, R. M. Williams, and C. A. Reinhart-King. Indentation measurements of the subendothelial matrix in bovine carotid arteries. J. Biomech. 44(5):815–821, 2011.
Rezvani-Sharif, A., M. Tafazzoli-Shadpour, D. Kazemi-Saleh, and M. Sotoudeh-Anvari. Stress analysis of fracture of atherosclerotic plaques: crack propagation modeling. Med Biol Eng Comput. 55:1389–1400, 2016.
Rho, J. Y., M. E. Roy, T. Y. Tsui, and G. M. Pharr. Elastic properties of microstructural components of human bone tissue as measured by nanoindentation. J. Biomed. Mater. Res. 45(1):48–54, 1999.
Schiffrin, E. L., A. Tedgui, and S. Lehoux. mechanical stress and the arterial wall. Blood pressure and arterial wall mechanics in cardiovascular diseases. New York: Springer, pp. 97–106, 2014.
Stary, H. C., A. B. Chandler, R. E. Dinsmore, V. Fuster, S. Glagov, W. Insull, et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis a report from the committee on vascular lesions of the council on arteriosclerosis. Am. Heart Assoc. Circ. 92(5):1355–1374, 1995.
Taghizadeh, H., M. Tafazzoli-Shadpour, and M. B. Shadmehr. Analysis of arterial wall remodeling in hypertension based on lamellar modeling. J. Am. Soc. Hypertens. 9(9):735–744, 2015.
Taghizadeh, H., M. Tafazzoli-Shadpour, M. B. Shadmehr, and N. Fatouraee. Evaluation of biaxial mechanical properties of aortic media based on the lamellar microstructure. Materials 8(1):302–316, 2015.
Taylor, C. A., T. J. Hughes, and C. K. Zarins. Finite element modeling of three-dimensional pulsatile flow in the abdominal aorta: relevance to atherosclerosis. Ann. Biomed. Eng. 26(6):975–987, 1998.
Teng, Z., Y. Zhang, Y. Huang, J. Feng, J. Yuan, Q. Lu, et al. Material properties of components in human carotid atherosclerotic plaques: a uniaxial extension study. Acta Biomater. 10(12):5055–5063, 2014.
Tracqui, P., A. Broisat, J. Toczek, N. Mesnier, J. Ohayon, and L. Riou. Mapping elasticity moduli of atherosclerotic plaque in situ via atomic force microscopy. J. Struct. Biol. 174(1):115–123, 2011.
VanderBurgh, J. A., and C. A. Reinhart-King. The role of age-related intimal remodeling and stiffening in atherosclerosis. Adv. Pharmacol. 81:365–391, 2018.
Vengrenyuk, Y., S. Carlier, S. Xanthos, L. Cardoso, P. Ganatos, R. Virmani, et al. A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc. Natl. Acad. Sci. 103(40):14678–14683, 2006.
Wayman, B. H., W. R. Taylor, A. Rachev, and R. P. Vito. Arteries respond to independent control of circumferential and shear stress in organ culture. Ann. Biomed. Eng. 36(5):673–684, 2008.
Wolinsky, H., and S. Glagov. A lamellar unit of aortic medial structure and function in mammals. Circ. Res. 20(1):99–111, 1967.
Zieman, S. J., V. Melenovsky, and D. A. Kass. Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler. Thromb. Vasc. Biol. 25(5):932–943, 2005.
Acknowledgments
Authors thank Dr. Davood Kazemi-Saleh and Dr. Zahra Pourjafar at Baghiatallah Hospital for providing tissue specimens. Authors also thank Dr. Amirnader Emami Razavi at Tehran University of Medical Sciences for assistance in the preparation of samples for AFM test and determination of different components of atherosclerotic plaques and arterial wall layers in specimens.
Conflict of interest
Alireza Rezvani-Sharif, Mohammad Tafazzoli-Shadpour and Alberto Avolio declare that they have no conflict of interest.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all patients for being included in the study. No animal studies were carried out by the authors for this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Wei Sun and Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Rezvani-Sharif, A., Tafazzoli-Shadpour, M. & Avolio, A. Mechanical Characterization of the Lamellar Structure of Human Abdominal Aorta in the Development of Atherosclerosis: An Atomic Force Microscopy Study. Cardiovasc Eng Tech 10, 181–192 (2019). https://doi.org/10.1007/s13239-018-0370-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-018-0370-1