Abstract
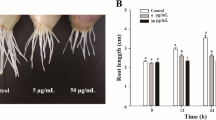
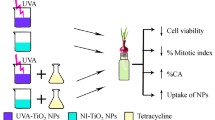
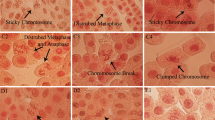
The extensive production and utilisation of titanium dioxide (TiO2) and zinc oxide (ZnO) nanoparticles (NPs) in consumable items may enhance significant increase in fauna and flora exposure. Studies showing the interactive effect of NPs in biological systems are limited. Herein, we showed the cytogenotoxic effects of TiO2 and ZnO NPs, and their mixture (1:1) using the Allium cepa assay. Mitotic index (MI) and chromosomal aberrations (CAs) were assessed in A. cepa L. bulbs exposed to each NP and their mixture at concentrations of 5, 10, 20, 40 and 80 mg L−1, respectively. The recovery effect of the root tip cells from the cytogenotoxic effects of the nanoparticles was also investigated. TiO2, ZnO NPs and their mixture significantly (p < 0.05) induced increase in CA and reduction in MI in A. cepa root cells, but the mixture induced the highest frequency of CA and reduction in MI. When the treated meristematic cells were placed in water for recovery, there were reduction in the number of aberrant cells in A. cepa exposed to TiO2 and the mixture. Interactive factor analysis of the effects of the mixture showed antagonism. The aberrations induced by TiO2 NPs appeared to be transient while those induced by ZnO NPs may be transmissible due to the increase in frequency of aberrations in the recovery test. This finding showed the potential of tested NPs to induce mutation in somatic cells, and is of public and environmental health significance.

Similar content being viewed by others
References
Amer SM, Ali EM. Cytological effects of pesticides V. Effects of some herbicides on Vicia faba. Cytologia. 1974;39:633.
Asztemborska M, Steborowski R, Kowalska J, Bystrzejewska-Piotrowska G. Accumulation of aluminium by plants exposed to nano-and microsized particles of Al. Int J Environ Res. 2015;9(1):109–16.
Atoyebi SM, Oyeyemi IT, Dauda BA, Bakare AA. Genotoxicity and anti- genotoxicity of aqueous extracts of herbal recipes containing Luffa cylindrica (L), Nymphaea lotus (L) and Spondias mombin (L) using the Allium cepa (L) assay. African Journal of Pharmacy and Pharmacology. 2015;9(15):492–9.
Bakare AA, Okunola AA, Adetunji OA, Jenmi HB. Genotoxicity assessment of a pharmaceutical effluent using four bioassays. Genet Mol Biol. 2009;32(2):373–81.
Bakare AA, Adeyemi AO, Adeyemi A, Alabi OA, Osibanjo O. Cytogenotoxic effects of electronic waste leachate in Allium cepa. Caryologia. 2012;65(2):94–100.
Bakare AA, Alabi OA, Gbadebo AM, Ogunsuyi OI, Alimba CG. In vivo cytogenotoxicity and oxidative stress induced by electronic waste leachate and contaminated groundwater. Challenges. 2013;4:169–87.
Balasubramanyam A, Sailaja N, Mahboob M, Rahman MF, Hussain SM, Grover P. In vivo genotoxicity assessment of aluminum oxide nanomaterials in rat peripheral blood cells using the Comet assay and micronucleus test. Mutagenesis. 2009;24(3):245–51.
Bundschuh M, Filser J, Lüderwald S, McKee MS, Metreveli G, Schaumann GE, Schulz R, Wagner S. Nanoparticles in the environment: where do we come from, where do we go to? Environ Sci Eur. 2018;30:6. https://doi.org/10.1186/s12302-018-0132-6.
Choi J, Lee JK, Jeong J, Choy J. Toxicity evaluation of inorganic nanoparticles: considerations and challenges. Mol Cell Toxicol. 2013;9:205–10.
Darlington CD, Mc LL. Action of maleic hydrazide on the cell. Nature. 1951;167:407–8.
Di Virgilio AL, Regiosa M, Arnal PM, Lorenzo F, de Mele M. Comparative study of the cytotoxic and genotoxic effects of titanium dioxide and aluminum oxide nanoparticles in Chinese hamster ovary (CHO-K1) cells. J Hazard Mater. 2010;177:711–8.
Drost W, Matzke M, Backhaus T. Heavy metal toxicity to Lemna minor: studies on the time dependence of growth inhibition and the recovery after exposure. Chemosphere. 2007;67:36–433.
Dutta J, Ahmad A, Singh J. Study of industrial effluents induced genotoxicity on Allium cepa. Caryologia. 2018;71(2):139–45.
Fadoju OM, Ogunsuyi OI, Akanni O, Alabi O, Alimba C, Adaramoye O, Cambier S, Eswara S, Gutleb AC, Bakare AA. Evaluation of cytogenotoxicity and oxidative stress parameters in male Swiss mice co-exposed to titanium dioxide and zinc oxide nanoparticles. Environ Toxicol Pharmacol. 2019;70:103204.
Fang J, Shijirbaatar A, Lin DH, Wang DJ, Shen B, Sun PD, Zhou ZQ. Stability of co-existing ZnO and TiO2 nanomaterials in natural water: aggregation and sedimentation mechanisms. Chemosphere. 2017;184:1125–33.
Filho RDS, Vicari T, Santos SA, Felisbino K, Mattoso N, Anna-Santos BFS, Cestari MM, Leme DM. Genotoxicity of titanium dioxide nanoparticles and triggering of defense mechanisms in Allium cepa. Genet Mol Biol. 2019;42(2):425–35.
Fiskesjo G. The Allium test as standard in environmental monitoring. Hereditas. 1985;102:99–112.
Fiskesjo G. Allium test for screening chemicals; evaluation of cytological parameters. In: Wuncheng W, Gorsuch JW, Hughes JS, editors. Plants for environmental studies. Boca Raton: CRC Lewis Publishers; 1997. p. 307–333.
Ghodake G, Seo YD, Lee DS. Hazardous phytotoxic nature of cobalt and zinc oxide nanoparticles assesses using Allium cepa. J Hazard Mater. 2011;186:952–5.
Ghosh M, Bandyopadhyay M, Mukherjee A. Genotoxicity of titanium dioxide (TiO2) nanoparticles at two trophic levels: plant and human lymphocytes. Chemosphere. 2010;81:1253–62.
Ghosh M, Jana A, Sinha S, Jothiramajayam M, Nag A, Chakraborty A, Mukherjee A, Mukherjee A. Effects of ZnO nanoparticles in plants: cytotoxicity, genotoxicity, deregulation of antioxidant defenses, and cell-cycle arrest. Mutat Res. 2016;807:25–322.
Gichner T, Patkova Z, Szakova J, Demnerova K. Cadmium induces DNA damage in tobacco roots, but no DNA damage, somatic mutations or homologous recombinations in tobacco leaves. Mutat Res. 2004;559:49–57.
Grover IS, Kaur S. Genotoxicity of wastewater samples from sewage and industrial effluent detected by the Allium root anaphase aberration and micronucleus assays. Mutat Res. 1999;426:183–8.
Hashimoto M, Imazato S. Cytotoxic and genotoxic characterization of aluminum and silicon oxide nanoparticles in macrophages. Dent Mater. 2015;31(5):556–64.
Jia F, Sun Z, Yan X, Zhou B, Wang J. Effect of pubertal nano-TiO2 exposure on testosterone synthesis and spermatogenesis in mice. Arch Toxicol. 2013;88(3):781–8.
Jiang Y, Sun Y, Liu H, Zhu F, Yin H. Solar photocatalytic decolorisation of C.I. Basic Blue 41 in an aqueous suspension of TiO2-ZnO. Dyes and Pigment. 2008;78(1):77–83.
Katsifis SP, Kinney PL, Hosselet S, Burns FJ, Christie NT. Interaction of nickel with in the induction of sister chromatid exchanges in human lymphocytes. Mutation. 1996;359(1):7–15.
Kaufman BP. Cytochemical studies of changes induced in cellular material by ionization radiations. Ann N Y Acad Sci. 1958;59:553.
Khan M, Naqvi AH, Ahmad M. Comparative study of the cytotoxic and genotoxic potentials of zinc oxide and titanium dioxide nanoparticles. Toxicology Report. 2015;2:765–74.
Kim K, Song J, Kim M, Chung A, Jeong J, Yang J, Choi A, Choi H, Oh J. Physicochemical analysis methods for nanomaterials considering their toxicological evaluations. Mol Cell Toxicol. 2014;10:342–60.
Klancnik K, Drobne D, Valant J, DolencKoce J. Use of a modified Allium test with nanoTiO2. Ecotoxicol Environ Saf. 2011;74:85–92.
Ko K, Koh D, Kong I. Toxicity evaluation of individual and mixtures of nanoparticles based on algal chlorophyll content and cell count. Materials (Basel). 2018;11(1):121.
Kocaman AY, Kilic E. Evaluation of the genotoxicity of commercial formulations of ethephon and ethephon+cyclanilide on Allium cepa L. root meristematic cells. Caryologia. 2017;70(3):229–37.
Kumar A, Najafzadeh M, Jacob BK, Dhawan A, Anderson D. Zinc oxide nanoparticles affect the expression of p53, Ras p21 and JNKs: an ex vivo/in vitro exposure study in respiratory disease patients. Mutagenesis. 2015;30(2):237–45.
Kumar A, Dhawan A. Genotoxic and carcinogenic potential of engineered nanoparticles: an update. Arch Toxicol. 2013;87:1883–900.
Kumari M, Khan SS, Pakrashi S, Mukherjee A, Chandrasekaran N. Cytogenetic and genotoxic effects of zinc oxide nanoparticles on root cells of Allium cepa. J Hazard Mater. 2011;190:613–21.
Larue C, Laurette J, Herlin-Boime N, Khodja H, Favard B, Flank A, Brisset F, Carriere M. Accumulation, trans location and impact of TiO2 nanoparticles in wheat: influence of diameter and crystal phase. Sci Total Environ. 2012;431:197–208.
Lee CW, Mahendra S, Zodrow K, Li D, Tsai YC, Braam J. Developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environ Toxicol Chem. 2010;29:669–75.
Lee S, Chung H, Kim S, Lee I. The genotoxic effect of ZnO and CuO nanoparticles on early growth of Buckwheat, Fagopyrum esculentum. Water, Air, Soil Pollut. 2013;224:1668.
Leme DM, Marin-Morales MA. Allium cepa test in environmental monitoring: a review on its application. Mutat Res. 2009;682:71–81.
Lin D, Xing B. Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ Pollut. 2008;150(2):243–50.
Magdolenova Z, Collins A, Kumar A, Dhawan A, Stone V, Dusinska M. Mechanisms of genotoxicity: a review of in vitro and in vivo studies with engineered nanoparticles. Nanotoxicology. 2014;8(3):233–78.
Mesi A, Kopliku D. Cytotoxic and genotoxic potency screening of two pesticides on Allium cepa. Procedia Technol. 2013;8:19–26.
Nam D, Lee B, Eom I, Kim P, Yeo M. Uptake and bioaccumulation of titanium- and silver- nanoaparticles in aquatic ecosystems. Mol Cell Toxicol. 2014;10:9–17.
Ogunsuyi OI, Fadoju OM, Coker MM, Akinrinade SO, Oyeyemi IT, Alabi OA, Alimba CG, Bakare AA. Nano-genotoxicity evaluation: a review. In: Kumar VN, Ranjan S, editors. Nanotoxicology: toxicity, risk assessment and management. Boca Raton: CRC Press Taylor and Francis Group; 2018. p. 463–504.
Oyeyemi IT, Bakare AA. Genotoxic and anti-genotoxic effect of aqueous extracts of Spondias mombin L., Nymphea lotus L. and Luffa cylindrica L. on Allium cepa root tip cells. Caryologia. 2013;66(4):360–7.
Pakrashi S, Jain N, Dalai S, Jayakumar J, Chandrasekaran PT, Raichur AM, Chandrasekaran N, Mukherjee A. In vivo genotoxicity assessment of titanium dioxide nanoparticles by Allium cepa root tip assay at high exposure concentrations. PLoS ONE. 2014;9(2):e87789.
Park C, Jung J, Baek M, Sung B, Park J, Seol Y, Yeom D, Park J, Kin Y. Mixture toxicity of metal oxide nanoparticles and silver ions on Daphnia magna. J Nanopart Res. 2019;21:166.
Ramesh M, Palanisamy K, Babu K, Sharma NK. Effects of bulk and nano-titanium dioxide and zinc oxide on physio-morphological changes in Triticum aestivum Linn. J Glob Biosci. 2014;3(2):415–22.
Sharma V, Singh P, Pandey A, Dhawan A. Induction of oxidative stress, DNA damage and apoptosis in mouse liver after sub-acute oral exposure to zinc oxide nanoparticles. Mutat Res. 2012;745:84–91.
Shaymurat T, Gu J, Xu C, Yang Z, Zhao Q, Liu Y, Liu Y. Phytotoxic and genotoxic effects of ZnO nanoparticles on garlic (Allium sativum L.): a morphological study. Nanotechnology. 2012;6(3):241–8.
Shukla RK, Sharma V, Pandey AK, Singh S, Sultana S, Dhawan A. ROS-mediated genotoxicity induced by titanium dioxide nanoparticles in human epidermal cells. Toxicol In Vitro. 2011;25:231–41.
Singh S. Nanomaterials as non-viral siRNA delivery agents for cancer therapy. Bioimpacts. 2013;3(2):53–655.
Sun Z, Xiong T, Zhang T, Wang N, Chen D, Li S. Influences of zinc oxide nanoparticles on Allium cepa root cells and the primary cause of phytotoxicity. Ecotoxicology. 2019;28(2):175–88.
Yah CS, Simate GS, Iyuke SE. Nanoparticles toxicity and their routes of exposures. J Pharm Sci. 2012;25(2):477–91.
Zhang H, Jiang Y, He Z, Ma M. Cadmium accumulation and oxidative burst in garlic (Allium sativum). J Plant Physiol. 2005;162:977–84.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fadoju, O.M., Osinowo, O.A., Ogunsuyi, O.I. et al. Interaction of titanium dioxide and zinc oxide nanoparticles induced cytogenotoxicity in Allium cepa. Nucleus 63, 159–166 (2020). https://doi.org/10.1007/s13237-020-00308-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13237-020-00308-1