Abstract
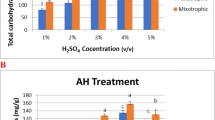
In this study, we have described three steps to produce ethanol from Pogonatherum crinitum, which was derived after the treatment of textile wastewater. (a) Production of biomass: biomass samples collected from a hydroponic P. crinitum phytoreactor treating dye textile effluents and augmented with Ca-alginate immobilized growth-promoting bacterium, Bacillus pumilus strain PgJ (consortium phytoreactor), and waste sorghum husks were collected and dried. Compositional analysis of biomass (consortium phytoreactor) showed that the concentration of cellulose, hemicelluloses and lignin was 42, 30 and 17%, respectively, whereas the biomass samples without the growth-promoting bacterium (normal phytoreactor) was slightly lower, 40, 29 and 16%, respectively. (b) Hydrolysate (sugar) production: a crude sample of the fungus, Phanerochaete chrysosporium containing hydrolytic enzymes such as endoglucanase (53.25 U/ml), exoglucanase (8.38 U/ml), glucoamylase (115.04 U/ml), xylanase (83.88 U/ml), LiP (0.972 U/ml) and MnP (0.459 U/ml) was obtained, and added to consortium, normal and control phytoreactor derived biomass supplemented with Tween-20 (0.2% v/v). The hydrolysate of biomass from consortium phytoreactor produced maximum reducing sugar (0.93 g/l) than hydrolysates of normal phytoreactor biomass (0.82 g/l) and control phytoreactor biomass (0.79 g/l). FTIR and XRD analysis confirmed structural changes in treated biomass. (c) Ethanol production: the bioethanol produced from enzymatic hydrolysates of waste biomass of consortium and normal phytoreactor using Saccharomyces cerevisiae (KCTC 7296) was 42.2 and 39.4 g/l, respectively, while control phytoreactor biomass hydrolysate showed only 25.5 g/l. Thus, the amalgamation of phytoremediation and bioethanol production can be the truly environment-friendly way to eliminate the problem of textile dye along with bioenergy generation.


Similar content being viewed by others
References
Adachi S (1965) Thin-layer chromatography of carbohydrates in the presence of bisulfite. J Chromatogr A 17:295–299. https://doi.org/10.1016/S0021-9673(00)99871-6
Adney B, Baker J (2008) Measurement of cellulase activities, Laboratory Analytical Procedure (LAP). Technical Report NREL/TP-510-42628. pp 1–8
Anto H, Trivedi UB, Patel KC (2006) Glucoamylase production by solid-state fermentation using rice flake manufacturing waste products as substrate. Bioresour Technol 97:1161–1166. https://doi.org/10.1016/j.biortech.2005.05.007
Anwar Z, Gulfraz M, Irshad M (2014) Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: a brief review. J Rad Res Appl Sci 7:163–173. https://doi.org/10.1016/j.jrras.2014.02.003
AOAC (1980) Official methods of analysis of the Association of Official Analytical Chemists. In: Horwitz W (ed), 13th edn. Association of Official Analytical Chemists, Washington DC
Balat M (2011) Production of bioethanol from lignocellulosic materials via the biochemical pathway: a review. Energy Convers Manag 52:858–875. https://doi.org/10.1016/j.enconman.2010.08.013
Banat IM, Nigam P, Singh D, Marchant R (1996) Microbial decolourization of textile dyes containing effluents: a review. Bioresour Technol 58:217–227. https://doi.org/10.1016/S0960-8524(96)00113-7
Belkacemi K, Turcotte G, Savoie P (2002) Aqueous/steam-fractionated agricultural residues as substrates for ethanol production. Ind Eng Chem Res 41:173–179. https://doi.org/10.1021/ie0102246
Berlin A, Gilkes N, Kurabi A, Bura R, Tu MB, Kilburn D, Saddler J (2005) Weak lignin-binding enzymes—a novel approach to improve activity of cellulases for hydrolysis of lignocellulosics. Appl Biochem Biotechnol 121:163–170. https://doi.org/10.1385/ABAB:121:1-3:0163
Binod P, Sindhu R, Singhania RR, Vikram S, Devi L, Nagalakshmi S, Kurien N, Sukumaran RK, Pandey A (2010) Bioethanol production from rice straw: an overview. Bioresour Technol 101:4767–4774. https://doi.org/10.1016/j.biortech.2009.10.079
Cervero JM, Skovgaard PA, Felby C, Sorensen HR, Jorgensen H (2010) Enzymatic hydrolysis and fermentation of palm kernel press cake for production of bioethanol. Enzyme Microb Technol 46:177–184. https://doi.org/10.1016/j.enzmictec.2009.10.012
Eriksson T, Karlsson J, Tjerneld F (2002) A model explaining declining rate in hydrolysis of lignocellulose substrates with cellobiohydrolase I (Cel7A) and endoglucanase I (Cel7B) of Trichoderma reesei. Appl Biochem Biotechnol 101:41–60. https://doi.org/10.1385/ABAB:101:1:41
Govumoni SP, Gentela J, Koti S, Haragopal V, Venkateshwar S, Rao LV (2015) Extracellular lignocellulolytic enzymes by Phanerochaete chrysosporium (MTCC 787) under solid-state fermentation of agro wastes. Int J Curr Microbiol Appl Sci 4:700–710
Gutierrez-Maneroa FJ, Ramos-Solanoa B, Probanzaa A, Mehouachib J, Tadeob FR, Talon M (2001) The plant-growth-promoting rhizobacteria Bacillus pumilus and Bacillus licheniformis produce high amounts of physiologically active gibberellins. Physiol Plant 111:206–211. https://doi.org/10.1034/j.1399-3054.2001.1110211.x
Hall M, Bansal P, Lee JH, Realff MJ, Bommarius AS (2010) Cellulose crystallinity—a key predictor of the enzymatic hydrolysis rate. FEBS J 277:1571–1582. https://doi.org/10.1111/j.1742-4658.2010.07585.x
Jagtap SS, Woo SM, Kim TS, Dhiman SS, Kim D, Lee JK (2014) Phytoremediation of diesel-contaminated soil and saccharification of the resulting biomass. Fuel 116:292–298. https://doi.org/10.1016/j.fuel.2013.08.017
Janardhan S, Sain M (2011) Targeted disruption of hydroxyl chemistry and crystallinity in natural fibers for the isolation of cellulose nano-fibers via enzymatic treatment. BioRes 6:1242–1250
Kagalkar AN, Govindwar SP (2010) Phytoremediation technologies for removal of textile dyes: an over view and future prospectus. Nova Science Publishers Inc., New York
Kariminiaae HR, Sakurai A, Sakakibara M (2007) Decolorization of synthetic dyes by a new manganese peroxidase-producing white rot fungus. Dyes Pigm 72:157–162. https://doi.org/10.1016/j.dyepig.2005.08.010
Khaliq S, Khalid A, Saba B, Mahmood S, Siddique MT, Aziz I (2013) Effect of acc deaminase bacteria on tomato plants containing azo dye wastewater. Pak J Bot 45:529–534
Khandare RV, Govindwar SP (2015) Phytoremediation of textile dyes and effluents: current scenario and future prospects. Biotechnol Adv 33:1697–1714. https://doi.org/10.1016/j.biotechadv.2015.09.003
Kshirsagar SD, Saratale GD, Saratale RG, Govindwar SP, Oh MK (2016) An isolated Amycolatopsis sp. GDS for cellulase and xylanase production using agricultural waste biomass. J Appl Microbiol 120:112–125. https://doi.org/10.1111/jam.12988
Kuhad RC, Mehta G, Gupta R, Sharma KK (2010) Fed batch enzymatic saccharification of newspaper cellulosics improves the sugar content in the hydrolysates and eventually the ethanol fermentation by Saccharomyces cerevisiae. Biomass Bioenergy 34:1189–1194. https://doi.org/10.1016/j.biombioe.2010.03.009
Lee JW, Kim HY, Koo BW, Choi DH, Kwon M, Choi IG (2008) Enzymatic saccharification of biologically pretreated Pinus densiflora using enzymes from brown rot fungi. J Biosci Bioeng 106:162–167. https://doi.org/10.1263/jbb.106.162
Liu XL, Zeng GM, Tang L, Zhong H, Wang RY, Fu HY, Liu ZF, Huang HL, Zhang JC (2008) Effects of dirhamnolipid and SDS on enzyme production from Phanerochaete chrysosporium in submerged fermentation. Process Biochem 43:1300–1303. https://doi.org/10.1016/j.procbio.2008.06.007
Lo YC, Saratale GD, Chen WM, Bai MD, Chang JS (2009) Isolation of cellulose-hydrolytic bacteria and applications of the cellulolytic enzymes for cellulosic biohydrogen production. Enzyme Microb Technol 44:417–425. https://doi.org/10.1016/j.enzmictec.2009.03.002
Miller GL (1959) Use of dinitrosalicylic reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Mosier N, Wyman C, Dale B, Elander R, Lee YY, Holtzapple M, Ladisch M (2005) Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour Technol 96:673–686. https://doi.org/10.1016/j.biortech.2004.06.025
Nzila C, Dewulf J, Spanjers H, Kiriamiti H, van Langenhove H (2010) Biowaste energy potential in Kenya. Renew Energy 35:2698–2704. https://doi.org/10.1016/j.renene.2010.04.016
Öhgren K, Bura R, Lesnicki G, Saddler J, Zacchi G (2007) A comparison between simultaneous saccharification and fermentation and separate hydrolysis and fermentation using steam-pretreated corn stover. Process Biochem 42:834–839. https://doi.org/10.1016/j.procbio.2007.02.003
Oliveira JG, Cruz CHG (2013) Properties of a biosurfactant produced by Bacillus pumilus using vinasse and waste frying oil as alternative carbon sources. Braz Arch Biol Technol 56:155–160. https://doi.org/10.1590/S1516-89132013000100020
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:10. https://doi.org/10.1186/1754-6834-3-10
Rogalski J, Szczodrak J, Janusz G (2006) Manganese peroxidase production in submerged cultures by free and immobilized mycelia of Nematoloma frowardii. Bioresour Technol 97:469–476. https://doi.org/10.1016/j.biortech.2005.03.002
Saratale GD, Saratale RG, Lo YC, Chang JS (2010) Multicomponent cellulase production by Cellulomonas biazotea NCIM-2550 and its applications for cellulosic biohydrogen production. Biotechnol Prog 26:406–416. https://doi.org/10.1002/btpr.342
Sawada T, Nakamura Y, Kobayashi F, Kuwahara M, Watanabe T (1995) Effects of fungal pretreatment and steam explosion pretreatment on enzymatic saccharification of plant biomass. Biotechnol Bioeng 48:719–724. https://doi.org/10.1002/bit.260480621
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794. https://doi.org/10.1177/004051755902901003
Seo D-J, Fujita H, Sakoda A (2011) Effects of a non-ionic surfactant, Tween-20, on adsorption/desorption of saccharification enzymes onto/from lignocelluloses and saccharification rate. Adsorption 17:813–822. https://doi.org/10.1007/s10450-011-9340-8
Shu H, Zhang P, Chang CC, Wang R, Zhang S (2015) Agricultural waste. Water Environ Res 87:1256–1285. https://doi.org/10.2175/106143013X13698672322345
Taherzadeh MJ, Karimi K (2008) Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: a review. Int J Mol Sci 9:1621–1651. https://doi.org/10.3390/ijms9091621
Tollefson J (2008) Energy: not your father’s biofuels. Nature 451:880–883. https://doi.org/10.1038/451880a
Uma C, Muthulakshmi C, Gomathi D, Gopalkrishan VK (2010) Fungal invertase as aid for production of ethanol from sugarcane bagasse. Res J Microbiol 5:980–985. https://doi.org/10.3923/jm.2010.980.985
UN (2008) The Millennium Development Goals Report 2008, New York
Waghmare PR, Kadam AA, Saratale GD, Govindwar SP (2014) Enzymatic hydrolysis and characterization of waste lignocellulosic biomass produced after dye bioremediation under solid state fermentation. Bioresour Technol 168:136–141. https://doi.org/10.1016/j.biortech.2014.02.099
Watharkar AD, Khandare RV, Waghmare PR, Jagadale AD, Govindwar SP, Jadhav JP (2015) Treatment of textile effluent in a developed phytoreactor with immobilized bacterial augmentation and subsequent toxicity studies on Etheostoma olmstedi fish. J Hazard Mater 283:698–704. https://doi.org/10.1016/j.jhazmat.2014.10.019
Williams MB, Reese HD (1950) Colorimetric determination of ethyl alcohol. Anal Chem 22:1556–1561. https://doi.org/10.1021/ac60048a025
Yeoman CJ, Han Y, Dodd D, Schroeder CM, Mackie RI, Cann IKO (2010) Thermostable enzymes as biocatalysts in the biofuel industry. Adv Appl Microbiol 70:1–55. https://doi.org/10.1016/S0065-2164(10)70001-0
Yoswathana N, Phuriphipat P, Treyawutthiwat P, Eshtiaghi MN (2010) Bioethanol production from rice straw. Energy Res J 1:26–31. https://doi.org/10.3844/erjsp.2010.26.31
Zhong C, Lau MW, Balan V, Dale BE, Yuan YJ (2009) Optimization of enzymatic hydrolysis and ethanol fermentation from AFEX-treated rice straw. Appl Microbiol Biotechnol 84:667–676. https://doi.org/10.1007/s00253-009-2001-0
Zhou D, Zhang L, Guo S (2005) Mechanisms of lead biosorption on cellulose/chitin beads. Water Res 39:3755–3762. https://doi.org/10.1016/j.watres.2005.06.033
Funding
The authors Dr. P. R. Waghmare and Dr. A. D. Watharkar would like to thank UGC (University Grants Commission), New Delhi for providing funding through UGC-NET-SRF fellowship and UGC-Women Postdoctoral fellowship (PDFW), respectively. Corresponding author wishes to thank UGC for providing funding through Special Assistance Program i.e. SAP (Grant No. SU/EST/PG/1328) to the Department of Biochemistry, Shivaji University Kolhapur. Prof. S. P. Govindwar is also thankful to The Korean Federation of Science and Technology Society, South Korea for providing Brain Pool Fellowship (Grant number 172S-5-3-1917).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Waghmare, P.R., Watharkar, A.D., Jeon, BH. et al. Bio-ethanol production from waste biomass of Pogonatherum crinitum phytoremediator: an eco-friendly strategy for renewable energy. 3 Biotech 8, 158 (2018). https://doi.org/10.1007/s13205-018-1188-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1188-0