Abstract
Madhuca indica provides livelihood to several tribal people in India, where the flowers are used for extraction of sweet juices having multiple applications. Certain trees have more value as judged by the tribal people mainly based on yield and quality performance of the trees, and these trees were selected for the genetic diversity analyses. Genetic diversity of 48 candidate Mahua trees from Etapalli, Dadagaon, and Jawhar, Maharashtra, India, was assessed using ISSR markers. Fourteen ISSR primers revealed a total of 132 polymorphic bands giving overall 92% polymorphism. Genetic diversity, in terms of expected number of alleles (Ne), the observed number of alleles (Na), Nei’s genetic diversity (H), and Shannon’s information index (I) was 1.921, 1.333, 0.211, and 0.337, respectively, and suggested lower genetic diversity. Region wise analysis revealed higher genetic diversity for site Etapalli (H = 0.206) and lowest at Dhadgaon (H = 0.140). Etapalli area possesses higher forest cover than Dhadgaon and Jawhar. Additionally, in Dhadgaon and Jawhar M. indica trees are restricted to field bunds; both reasons might contribute to lower genetic diversity in these regions. The dendrogram and the principal coordinate analyses showed no region-specific clustering. The clustering patterns were supported by AMOVA where higher genetic variance was observed within trees and lower variance among regions. Long-distance dispersal and/or higher human interference might be responsible for low diversity and higher genetic variance within the candidate trees.


Similar content being viewed by others
References
Aabd NA, Msanda F, Mousadik AE (2015) Genetic diversity of the endangered argan tree (Argania spinosa L.) (sapotaceae) revealed by ISSR analysis. Basic Res J Agri Sci Rev 4:176–186
Apte GS, Bahulikar RA, Kulkarni RS, Lagu MD, Kulkarni BG, Suresh HS, Rao PSN, Gupta VS (2006) Genetic diversity analysis in Gaultheria fragrantissima Wall. (Ericaceae) from the two biodiversity hotspots in India using ISSR markers. Curr Sci 91(12):1634–1640
Austerlitz F, Mariette S, Machon N, Gouyon P-H, Godelle B (2000) Effects of colonization processes on genetic diversity: differences between annual plants and tree species. Genetics 154(3):1309–1321
Bachmann K (1994) Molecular markers in plant ecology. New Phytol 126(3):403–418. https://doi.org/10.1111/j.1469-8137.1994.tb04242.x
Bahulikar RA, Lagu MD, Kulkarni BG, Pandit SS, Suresh HS, Rao MKV, Ranjekar PK, Gupta VS (2004a) Genetic diversity among spatially isolated populations of Eurya nitida Korth. (Theaceae) based on inter-simple sequence repeats. Curr Sci 86(6):824–831
Bahulikar RA, Stanculescu D, Preston CA, Baldwin IT (2004b) ISSR and AFLP analysis of the temporal and spatial population structure of the post-fire annual, Nicotiana attenuata, in SW Utah. BMC Ecol 4:12. https://doi.org/10.1186/1472-6785-4-12
Chaudhary A, Bhandari A, Pandurangan A, Koul S (2015) Madhuca indica J. F. Gmel. (Sapotaceae): an overview. Int J Pharma Sci Let 5(2):539–545
Chen L, Chen F, He S, Ma L (2014) High genetic diversity and small genetic variation among populations of Magnolia wufengensis (Magnoliaceae), revealed by ISSR and SRAP markers Electron. J Biotechnol 17(6):268–274. https://doi.org/10.1016/j.ejbt.2014.08.003
Dai Z-C, Si C-C, Zhai D-L, Huang P, Qi S-S, Zhong Q-X, Hu X, Li H-M, Du D-L (2013) Human impacts on genetic diversity and differentiation in six natural populations of Madhuca hainanensis, an endemic and endangered timber species in China. Biochem Syst Ecol 50:212–219
Deshpande AU, Apte GS, Bahulikar RA, Lagu MD, Kulkarni BG, Suresh HS, Singh NP, Rao MKV, Gupta VS, Pant A, Ranjekar PK (2001) Genetic diversity across natural populations of three montane plant species from the Western Ghats, India revealed by intersimple sequence repeats. Mol Ecol 10(10):2397–2408. https://doi.org/10.1046/j.0962-1083.2001.01379.x
Gavankar R, Chemburkar M (2016) Genetic analysis of Madhuca longifolia (J. Koenig ex L.) JF Macbr. using RAPD markers. Int J Curr Microbiol App Sci 5(8):608–615
Ghadge SV, Raheman H (2005) Biodiesel production from mahua (Madhuca indica) oil having high free fatty acids. Biomass Bioenergy 28(6):601–605. https://doi.org/10.1016/j.biombioe.2004.11.009
Gienapp P, Teplitsky C, Alho J, Mills J, Merilä J (2008) Climate change and evolution: disentangling environmental and genetic responses. Mol Ecol 17(1):167–178
Hammer Ø, Harper DAT, Ryan PD (2001) Past: paleontological statistics software package for education and data analysis. Palaeontol Electronica 4(1):4–9
Heslop-Harrison J (1975) Incompatibility and the pollen-stigma interaction. Ann Rev Plant Physiol 26(1):403–425
Holsinger KE, Weir BS (2009) Genetics in geographically structured populations: defining, estimating and interpreting F(ST). Nat Rev Genet 10(9):639–650. https://doi.org/10.1038/nrg2611
Idris AE, Hamza NB, Yagoub SO, Ibrahim AIA, El-Amin HKA (2012) Maize (Zea mays L.) genotypes diversity study by utilization of Inter-Simple Sequence Repeat (ISSR) markers. Aust J Basic Appl Sci 6(10):42–47
Joshi SP, Ranjekar PK, Gupta VS (1999) Molecular markers in plant genome analysis. Curr Sci 77(2):230–241
Kulkarni PS, Sharanappa G, Ramesh MR (2013) Mahua (Madhuca indica) as a source of biodiesel in India. Int J Eng Res Appl 4(7):2319–2329
Morikawa M, Muto T, Santos-Guerrra A, Kondo K (2014) Identifying, discriminating and isolating cultivars of ‘Marguerites’ originated from Argyranthemum frutescens parentages and their intergeneric and interspecific hybridities by DNA markers amplified by RAPD (Random Amplified Polymorphic DNA) and ISSR (inter-simple sequence repeat). Chromosome Bot 9:97–112
Mukherjee AK, Ratha S, Dhar S, Debata AK, Acharya PK, Mandal S, Panda PC, Mahapatra AK (2010) Genetic relationships among 22 taxa of bamboo revealed by ISSR and EST-based random primers. Biochem Genet 48(11–12):1015–1025. https://doi.org/10.1007/s10528-010-9390-8
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8(19):4321–4325. https://doi.org/10.1093/nar/8.19.4321
Nagaraju J, Kathirvel M, Kumar RR, Siddiq EA, Hasnain SE (2002) Genetic analysis of traditional and evolved Basmati and non-Basmati rice varieties by using fluorescence-based ISSR-PCR and SSR markers. Proc Natl Acad Sci USA 99(9):5836–5841. https://doi.org/10.1073/pnas.042099099
Naik D, Singh D, Vartak V, Paranjpe S, Bhargava S (2009) Assessment of morphological and genetic diversity in Gmelina arborea Roxb. New Forest 38(1):99–115. https://doi.org/10.1007/s11056-009-9134-y
Nybom H (2004) Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol Ecol 13(5):1143–1155
Nybom H, Bartish IV (2000) Effects of life history traits and sampling strategies on genetic diversity estimates obtained with RAPD markers in plants. Perspect Plant Ecol Evol Syst 3(2):93–114
Nybom H, Weising K, Rotter B (2014) DNA fingerprinting in botany: past, present, future. Investig Genet 5:1
Palme AE, Semerikov V, Lascoux M (2003) Absence of geographical structure of chloroplast DNA variation in sallow. Salix caprea L. Heredity (Edinb) 91(5):465–474. https://doi.org/10.1038/sj.hdy.6800307
Patel M, Naik SN (2010) Flowers of Madhuca indica, present status and future prospective. Indian J Nat Prod Resour 1(4):438–443
Patel PK, Prajapati NK, Dubey BK (2012) Madhuca indica: a review of its medicinal property. Int J Pharm Sci Rev Res 3(5):1285–1293
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in excel. Population genetic software for teaching and research. Mol Ecol Notes 6(1):288–295
Peakall R, Smouse PE (2012) GenAlEx 65: genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 28(19):2537–2539. https://doi.org/10.1093/bioinformatics/bts460
Sarwat M, Das S, Srivastava PS (2011) Estimation of genetic diversity and evaluation of relatedness through molecular markers among medicinally important trees: terminalia arjuna, T. chebula and T. bellerica. Mol Biol Rep 38(8):5025–5036
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biol Evol 30(12):2725–2729. https://doi.org/10.1093/molbev/mst197
Wani MS, Wani A, Mughal A (2015) Estimation of divergence to genetic variation in half-sib families of Madhuca indica GMEL. Under greenhouse and open field environmental conditions. Indian J Agr Sci 141(1):35–40
Yeh FC, Yang RC, Boyle T, Ye ZH, Mao JX (1999) POPGENE, version 1.32: the user friendly software for population genetic analysis. Molecular Biology and Biotechnology Centre, University of Alberta, Edmonton, AB, Canada
Acknowledgements
We thank Lilesh Chavan, Nana Pawara and Giri Gurudas from BISLD, Nasik, India, for their field support. Authors also thank Dr. Monali Rahalkar (Agharkar Research Institute, Pune) for critical reading suggestions for improvement of the manuscript.
Funding
Received funding for Maharashtra Gene Bank project from Rajiv Gandhi Science and Technology Commission, Mumbai and IISER Pune, India.
Author information
Authors and Affiliations
Contributions
SDN, VKK, and SA gave technical guidance and were involved in documentation, identification, and collection of plant samples. SSJ and RAB executed the laboratory experiments and RAB interpreted and wrote the draft manuscript. All the authors scrutinized and reviewed the manuscript, and approved the final version.
Corresponding author
Ethics declarations
In present work, human participation was limited to identification of plants by local tribal people and, therefore, formal consent was not required.
Conflict of interest
The authors declare no conflict of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13205_2018_1168_MOESM2_ESM.jpg
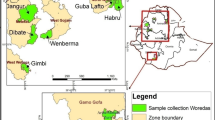
Supplementary material 2 (JPEG 132 kb) Fig. 1 Map of Maharashtra, India showing sampling locations of candidate trees of Madhuca indica
Rights and permissions
About this article
Cite this article
Nimbalkar, S.D., Jade, S.S., Kauthale, V.K. et al. Genetic diversity in the candidate trees of Madhuca indica J. F. Gmel. (Mahua) revealed by inter-simple sequence repeats (ISSRs). 3 Biotech 8, 143 (2018). https://doi.org/10.1007/s13205-018-1168-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1168-4