Abstract
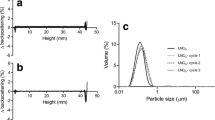
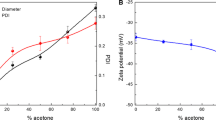
Oil-in-water nanoemulsions are promising colloidal dispersions for the delivery of hydrophobic drugs. The use of polysorbate 80 as stabilizing agent in these formulations can improve the delivery of the drug to the brain, thanks to a tropism for apolipoproteins. The aim of this study was to formulate injectable nanoemulsions stabilized by polysorbate 80, which meet United States Pharmacopoeia particle size requirements for parenteral emulsions. Nanoemulsions were prepared by high pressure homogenization and characterized in terms of mean hydrodynamic diameter, Gaussian distribution width, and volume-weighted percentage of fat droplets greater than 1.79 µm (PFAT1.79) or 5 µm (PFAT5). The effect of autoclaving, filtration and loading with nile red (a lipophilic fluorescent dye) on the nanoemulsions was evaluated. Real-time and accelerated stability tests were also performed. To satisfy Unites States Pharmacopoeia particle size specifications, nanoemulsions required six homogenization cycles. PFAT5 and PFAT1.79 were the particle size parameters more sensitive to discriminate the effect of homogenization, autoclaving, filtration and loading as well as globule size evolution during real-time stability tests. Results from accelerated stability studies correlated with the PFAT5 values measured over time. Overall, the study demonstrates that all nanoemulsions studied (autoclaved, filtered or loaded) satisfies United States Pharmacopoeia particle size requirements up to 90 days, maintaining PFAT5 to values lower than 0.05% v/v.








Similar content being viewed by others
References
Aggarwal P, Hall JB, McLeland CB et al (2009) Nanoparticle interaction with plasma proteins as it relates to particle biodistribution, biocompatibility and therapeutic efficacy. Adv Drug Deliv Rev 61:428–437. https://doi.org/10.1016/J.ADDR.2009.03.009
Allegra JR, Hawley SA (1972) Attenuation of sound in suspensions and emulsions: theory and experiments. J Acoust Soc Am 51:1545–1564. https://doi.org/10.1121/1.1912999
Bhattacharjee S (2016) DLS and zeta potential – What they are and what they are not? J Control Release 235:337–351. https://doi.org/10.1016/J.JCONREL.2016.06.017
Blasi P, Giovagnoli S, Schoubben A et al (2007) Solid lipid nanoparticles for targeted brain drug delivery. Adv Drug Deliv Rev 59:454–477. https://doi.org/10.1016/j.addr.2007.04.011
Blasi P, Giovagnoli S, Schoubben A et al (2011) Lipid nanoparticles for brain targeting I. Formulation optimization. Int J Pharm 419:287–295. https://doi.org/10.1016/J.IJPHARM.2011.07.035
Bonacucina G, Perinelli DR, Cespi M et al (2016) Acoustic spectroscopy: a powerful analytical method for the pharmaceutical field? Int J Pharm 503:174–195. https://doi.org/10.1016/j.ijpharm.2016.03.009
Calder PC, Jensen GL, Koletzko BV et al (2010) Lipid emulsions in parenteral nutrition of intensive care patients: current thinking and future directions. Intensive Care Med 36:735–749. https://doi.org/10.1007/s00134-009-1744-5
Chansiri G, Lyons RT, Patel MV, Hem SL (1999) Effect of surface charge on the stability of oil/water emulsions during steam sterilization. J Pharm Sci 88:454–458. https://doi.org/10.1021/js980293i
Chapter < 729>: globule size distribution in lipid injectable emulsion (2013) In: United States Pharmacopoeia. 36th ed
Chen H, Jin X, Li Y, Tian J (2016) Investigation into the physical stability of a eugenol nanoemulsion in the presence of a high content of triglyceride. RSC Adv 6:91060–91067. https://doi.org/10.1039/C6RA16270C
Dal Magro R, Albertini B, Beretta S et al (2018) Artificial apolipoprotein corona enables nanoparticle brain targeting. Nanomedicine 14:429–438. https://doi.org/10.1016/j.nano.2017.11.008
Delmas T, Piraux H, Couffin A-C et al (2011) How to prepare and stabilize very small nanoemulsions. Langmuir 27:1683–1692. https://doi.org/10.1021/la104221q
Driscoll DF (2002) The significance of particle/globule-sizing measurements in the safe use of intravenous lipid emulsions. J Dispers Sci Technol 23:679–687. https://doi.org/10.1081/DIS-120015371
Driscoll DF (2006) Lipid injectable emulsions: pharmacopeial and safety issues. Pharm Res 23:1959–1969. https://doi.org/10.1007/s11095-006-9092-4
Dukhin AS, Goetz PJ (eds) (2010) Acoustic theory for particulates. In: Characterization of liquids, nano- and microparticulates, and porous bodies using ultrasound studies in interface science, chap 4, vol 24. Elsevier, Amsterdam, pp 127–185. https://doi.org/10.1016/S1383-7303(10)23004-1
Floyd AG (1999) Top ten considerations in the development of parenteral emulsions. Pharm Sci Technolo Today 2:134–143. https://doi.org/10.1016/S1461-5347(99)00141-8
Ganta S, Talekar M, Singh A et al (2014) Nanoemulsions in translational research—opportunities and challenges in targeted cancer therapy. AAPS Pharm Sci Tech 15:694–708. https://doi.org/10.1208/s12249-014-0088-9
Göppert TM, Müller RH (2005) Polysorbate-stabilized solid lipid nanoparticles as colloidal carriers for intravenous targeting of drugs to the brain: comparison of plasma protein adsorption patterns. J Drug Target 13:179–187. https://doi.org/10.1080/10611860500071292
Gupta A, Eral HB, Hatton TA, Doyle PS (2016) Nanoemulsions: formation, properties and applications. Soft Matter 12:2826–2841. https://doi.org/10.1039/C5SM02958A
Hippalgaonkar K, Majumdar S, Kansara V (2010) Injectable lipid emulsions—advancements, opportunities and challenges. AAPS PharmSciTech 11:1526–1540. https://doi.org/10.1208/s12249-010-9526-5
Hörmann K, Zimmer A (2016) Drug delivery and drug targeting with parenteral lipid nanoemulsions—a review. J Control Release 223:85–98. https://doi.org/10.1016/J.JCONREL.2015.12.016
Kandadi P, AfzalSyed M, Goparaboina S, Veerabrahma K (2012) Albumin coupled lipid nanoemulsions of diclofenac for targeted delivery to inflammation. Nanomed Nanotechnol Biol Med 8:1162–1171. https://doi.org/10.1016/J.NANO.2011.12.006
Khachane PV, Jain AS, Dhawan VV et al (2015) Cationic nanoemulsions as potential carriers for intracellular delivery. Saudi Pharm J 23:188–194. https://doi.org/10.1016/J.JSPS.2014.07.007
Laxmi M, Bhardwaj A, Mehta S, Mehta A (2015) Development and characterization of nanoemulsion as carrier for the enhancement of bioavailability of artemether. Artif Cells Nanomedicine Biotechnol 43:334–344. https://doi.org/10.3109/21691401.2014.887018
Lu W, Kelly AL, Miao S (2017) Bioaccessibility and cellular uptake of β-carotene encapsulated in model O/W emulsions: influence of initial droplet size and emulsifiers. Nanomater (Basel, Switzerland). https://doi.org/10.3390/nano7090282
Mao L, O’Kennedy BT, Roos YH et al (2012) Effect of monoglyceride self-assembled structure on emulsion properties and subsequent flavor release. Food Res Int 48:233–240. https://doi.org/10.1016/J.FOODRES.2012.04.002
McClements DJ (1995) Advances in the application of ultrasound in food analysis and processing. Trends Food Sci Technol 6:293–299. https://doi.org/10.1016/S0924-2244(00)89139-6
Narsimhan G, Goell P (2001) Drop coalescence during emulsion formation in a high-pressure homogenizer for tetradecane-in-water emulsion stabilized by sodium dodecyl sulfate. J Colloid Interface Sci 238:420–432. https://doi.org/10.1006/JCIS.2001.7548
McClements DJ (2012) Nanoemulsions versus microemulsions: terminology, differences, and similarities. Soft Matter 8:1719–1729
Nicoli DF, Wu JS, Chang YJ et al (1995) Wide dynamic range particle size: analysis by DLS-SPOS: a combination of two technologies. Am Lab 27:41–49
Nicoli DF, Hasapidis K, O’Hagan P et al (1998) High-resolution particle size analysis of mostly submicrometer dispersions and emulsions by simultaneous combination of dynamic light scattering and single-particle optical sensing. In: Provder T (ed) Particle size distribution III—assessment and characterization, chap 6. American Chemical Society, Washington, DC, pp 52–76
Niederquell A, Kuentz M (2013) Proposal of stability categories for nano-dispersions obtained from pharmaceutical self-emulsifying formulations. Int J Pharm 446:70–80. https://doi.org/10.1016/J.IJPHARM.2013.02.005
Peng J, Dong W, Li L et al (2015) Effect of high-pressure homogenization preparation on mean globule size and large-diameter tail of oil-in-water injectable emulsions. J Food Drug Anal 23:828–835. https://doi.org/10.1016/J.JFDA.2015.04.004
Perinelli DR, Cespi M, Pucciarelli S et al (2013) Effect of phosphate buffer on the micellisation process of Poloxamer 407: Microcalorimetry, acoustic spectroscopy and dynamic light scattering (DLS) studies. Colloids Surfaces A Physicochem Eng Asp 436:123–129. https://doi.org/10.1016/j.colsurfa.2013.06.002
Perinelli DR, Cespi M, Bonacucina G et al (2017) Heating treatments affect the thermal behaviour of doxorubicin loaded in PEGylated liposomes. Int J Pharm 534:81–88. https://doi.org/10.1016/J.IJPHARM.2017.09.069
Qian C, McClements DJ (2011) Formation of nanoemulsions stabilized by model food-grade emulsifiers using high-pressure homogenization: Factors affecting particle size. Food Hydrocoll 25:1000–1008. https://doi.org/10.1016/J.FOODHYD.2010.09.017
Schultz S, Wagner G, Urban K, Ulrich J (2004) High-pressure homogenization as a process for emulsion formation. Chem Eng Technol 27:361–368. https://doi.org/10.1002/ceat.200406111
Shaha RB, Zidan AS, Funck T et al (2007) Quality by design: Characterization of self-nano-emulsified drug delivery systems (SNEDDs) using ultrasonic resonator technology. Int J Pharm 341:189–194. https://doi.org/10.1016/J.IJPHARM.2007.04.009
Sharma S, Sahni JK, Ali J, Baboota S (2015) Effect of high-pressure homogenization on formulation of TPGS loaded nanoemulsion of rutin—pharmacodynamic and antioxidant studies. Drug Deliv 22:541–551. https://doi.org/10.3109/10717544.2014.893382
Singh Y, Meher GJ, Raval K et al (2017) Nanoemulsion: Concepts, development and applications in drug delivery. J Control Release 252:28–49. https://doi.org/10.1016/J.JCONREL.2017.03.008
Stang M, Schuchmann H, Schubert H (2001) Emulsification in high-pressure homogenizers. Eng Life Sci 1:151–157. https://doi.org/10.1002/1618-2863(200110)1:4%3C151::AID-ELSC151%3E3.0.CO;2-D
Stillhart C, Kuentz M (2012) Comparison of high-resolution ultrasonic resonator technology and Raman spectroscopy as novel process analytical tools for drug quantification in self-emulsifying drug delivery systems. J Pharm Biomed Anal 59:29–37. https://doi.org/10.1016/J.JPBA.2011.10.018
Stillhart C, Cavegn M, Kuentz M (2013) Study of drug supersaturation for rational early formulation screening of surfactant/co-solvent drug delivery systems. J Pharm Pharmacol 65:181–192. https://doi.org/10.1111/j.2042-7158.2012.01586.x
Sun W, Xie C, Wang H, Hu Y (2004) Specific role of polysorbate 80 coating on the targeting of nanoparticles to the brain. Biomaterials 25:3065–3071. https://doi.org/10.1016/J.BIOMATERIALS.2003.09.087
Taylor TM, Davidson PM, Bruce BD, Weiss J (2005) Ultrasonic spectroscopy and differential scanning calorimetry of liposomal-encapsulated nisin. J Agric Food Chem 53:8722–8728. https://doi.org/10.1021/jf050726k
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rosi Cappellani, M., Perinelli, D.R., Pescosolido, L. et al. Injectable nanoemulsions prepared by high pressure homogenization: processing, sterilization, and size evolution. Appl Nanosci 8, 1483–1491 (2018). https://doi.org/10.1007/s13204-018-0829-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-018-0829-2