Abstract
The toxic effects of anionic dyes such as tartrazine and sunset yellow on humans and the aquatic environment are of serious concern. The need for the removal of these dyes from wastewaters led to the use of adsorption techniques as a cheap and efficient treatment method. Thus, this research was based on the preparation of a low-cost activated carbon derived from cassava sievate designated as CS, which was utilized in the adsorption of sunset yellow and tartrazine from simulated wastewater. The sorption process was carried out under varying process factors in a batch mode. Adsorbent characterization displayed the presence of surface functional groups by the FT-IR and a porous structure as revealed by scanning electron microscopy. Optimum dye uptake was recorded at pH (1.0–2.0), temperature (30–40 °C), CS dosage (0.1 g), and dye concentration (150 mg/L). A maximum CS monolayer uptake of 20.83 and 0.091 mg/g was recorded for tartrazine and sunset yellow dyes, respectively. The pseudo-second-order (R2 > 0.99) and Freundlich (R2 > 0.92) models were most fitted to the kinetics and isotherm data of the uptake of the dyes on CS. The adsorption equilibrium attainment was reached was within 90 min of dye sequestration. The experimental results revealed that both sunset yellow and tartrazine dyes were considerably adsorbed onto the environmentally compatible and low-cost activated carbon derived from cassava sievate.
Similar content being viewed by others
Introduction
A great number of production processes employ a category of manufactured chemical dyes for various applications (Lata et al. 2008). Continuous use of these dyes in the food, cosmetic, leather, textiles, rubber, printing, plastics, pharmaceuticals industries aid in the impartation of color on their products. The resultant effect of these dye applications is the generation of a huge amount of wastewater containing dyes in very high concentrations. An estimated amount of nearly 800,000 tons of manufactured dyes are produced annually and about 50% of these are azo dyes (Greluk and Hubicki 2011). Sunset yellow and tartrazine are invaluable dyes applied at very low concentrations for the synthesis of medicines especially for the shells of drugs capsules, syrups, maquillage, and food additives. However, when present in high concentration, they are extremely toxic and are highly soluble in water, which renders them difficult to detect, that is, their existence in industrial wastewater (Dawodu and Akpomie 2016). Increased concentrations of sunset yellow and tartrazine in humans could endanger the torso to grave conditions, such as infertility, thyroid cancer, asthma, migraines, eczema, lupus, and hyperactivity (Caliman et al. 2008). It is therefore vital that concentrations of such dyes are lowered or removed from effluents before being discharged into water bodies.
Several modes of treatment for dye/color expulsion are used in order to reduce their effect on the environment and these include; Microbial decomposition, photo-catalytic reduction, coagulation/flocculation, ion exchange, reverse osmosis, and adsorption technique. Adsorption as a technique has been validated to be most helpful and reliable among these different methods of dye removal and has equally been observed to be more economical than others (Okeola et al. 2017). Activated carbon, a structurally homogeneous material has been rated as an excellent adsorbent material for adsorbate removal. It has been found to display a very high efficiency for the adsorption of bigger-sized compounds such as dyes (Zhang et al. 2012). The disadvantage in its use is the high price and this curtails its application in an emerging economy, thereby endorsing the use of low-cost activated carbons derived from agricultural materials as well as low-cost biomass materials for pollutants adsorption. A few examples include corncob, coconut husk, wheat bean, banana peel, and walnut shell-based activated carbons (Ahmad et al. 2009; Arami et al. 2005; Sulak et al. 2007; Ani et al. 2020).
A though literature search revealed that there is presently no research on the use of cassava sievate activated carbon for the adsorption of tartrazine and sunset yellow dyes. Therefore, this work reports for the first time the application of a low-cost activated carbon derived from cassava sievate for the sequestration of the two anionic dyes from simulated wastewater. The prepared carbon was characterized to provide information on the surface properties and then the effect of operating factors on the uptake of the dyes was investigated. The kinetics and isotherm modeling of the removal process was also performed.
Materials and methods
CS preparation, characterization, and sorption
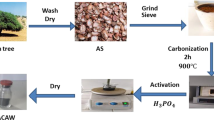
The adsorbent- cassava sievate were gotten from Egbelu Obube in Owerri North L.G.A., Imo State, Nigeria. The processed cassava sievates were dried under the sun for 5 days and were washed repeatedly with de-ionized water to wipe out all adhered particles and impurities. Thereafter, the washed substrate was kept under the sun to dry and the method of chemical activation and carbonization was used to produce the activated carbon. In this procedure, precisely 50 g of the dry mass of Cassava sievate was impregnated with 50 mL of 85% phosphoric acid in the ratio of 1:1. The carbonization process was carried out on the substrate using a muffle furnace at 550 °C for 1 h. The carbonized substrate was washed copiously with hot distilled water until a neutral pH was achieved and was dried in the oven at 100 0C for 4 h; thereafter, it was grounded and sieved with a sieve mesh of 180–200 mesh size. The sieved particles as-prepared activated carbon were designated as CS and used for the adsorption experiment without any further modification. FT-IR spectrophotometer (Shimadzu model) was applied in the assessment of the likely functional groups in the processed CS responsible for the uptake of the dyes while an assessment of its surface morphology was equally done with the use of Scanning electron microscope (SEM; Hitachi S4800 model).
The reagents employed were bought from Sigma-Aldrich and were of analytical grade, utilized with no further purification. A stock solution (1000 mg/L) of tartrazine and sunset yellow was prepared by dissolving the weighed amount of the dyes. The lower concentrations (30–150 mg/L) designed as working concentrations were all prepared from the 1000 mg/L using the dilution technique. The adsorption of the dyes on CS was done using the batch method as described in the supporting document. The kinetic and isotherm modeling of the adsorption process of sunset yellow and tartrazine was done using model equations (see Supporting document) (Chukwuemeka-Okorie et al. 2018; Ojo et al. 2019; Ravishankar et al. 2016).
Results and discussion
CS characterization
The SEM micrograph of CS is presented in Fig. 1. The specimen micrograph was taken at low accelerating voltage, 10 kV; and at this voltage, the surface microstructures of the sample are easily seen, as the penetration and diffusion area of incident electrons are shallow. As observed from the SEM image, the surface of the adsorbent is fine and highly porous. The working distance of 100 µm was used which gave the image a high resolution and a smaller depth of field. By decreasing the electron probe diameter on the specimen, the image was magnified to 500× and an objective lens aperture of 537 µm was used. From the SEM image of the adsorbent, pores of various sizes and varying layers could be observed. Aside from the rough and irregular structure of the adsorbent surface, micropores could be seen at that magnification and field depth. Therefore, the as-prepared CS consists of fine grains of irregular shapes and textures, with considerable layers of varying pore sizes to provide an excellent potential for adsorption of the dyes (Ezekoye et al. 2020).. The FT-IR spectrum of the adsorbent is shown in Fig. 2. From FT-IR spectrum, numerous bands were observed signifying that the sievate is composed of many functional groups, which would aid the sequestration of the dye molecules (Chukwuemeka-Okorie et al. 2018).
Effect of solution pH
The effect of pH on the uptake of tartrazine and sunset yellow onto CS is shown in Fig. 3. Although solution pH is known to influence significantly the uptake of pollutants on adsorbents, we observed from the results that the pH solution did not have much effect on the dyes uptake onto CS. However, a slight decrease in the uptake of both dyes on CS was achieved with an increase in pH. This is because as the pH of the adsorbate increases, the positively charged sites on the adsorbent get decreased (with a consequent increase in negative charges on the adsorbent) thereby causing less attraction of the anionic dyes for the surface of CS. This condition leads to electrostatic repulsion because it does not support the uptake of anionic dyes from the system (Ozcan et al. 2004). A decrease in sorption of both dyes is observed at high pH, this is because hydroxyl (OH−) ions compete effectively for the active site on the adsorbent with the anionic dye. The enhanced electrostatic force of attraction exists between the CS and anionic dyes at pH < 4 thereby increasing the removal of the dyes. At lower adsorbate pH, the adsorbent surface gets protonated, resulting in increasing dye adsorption due to electrostatic attraction. A similar result was recorded in the sorption of tartrazine onto sawdust biosorbent (Banerjee and Chattopadhyaya 2013).
Influence of dye concentration
The influence of dye concentration on the adsorption of sunset yellow and tartrazine dyes is shown in Fig. 4. The adsorption capacity of both dye ions onto CS increased with increased dye concentration, and this is attributable to an increased concentration gradient leading to enhanced driving force required to overcome all mass transfer resistances of the dyes between the aqueous and solid phases. The present finding is similar to the result earlier reported (Hamid et al. 2014). The dye concentration of 150 mg/L was utilized for the rest of this study and not 30 mg/L despite the high percentage adsorption recorded at 30 mg/L at a fixed adsorbent mass of 0.1 g. This was done to permit an effective study of the influence of dosage on the adsorption process.
Adsorption isotherm modeling
The equilibrium adsorption isotherm gives knowledge of the surface properties of sorbent materials, adsorption mechanism, and adsorbents affinity for the adsorbates (Das and Mondal 2011). The linear regression coefficients (R2) obtained from the isotherm models in the present study were used to determine the most suitable isotherm. The closest value to one gives an indication of the best fit model. The equilibrium model constants for the sorption of sunset yellow and tartrazine onto CS are shown in Table 1. Freundlich isotherm assumes that the adsorbent surface is heterogeneous and also an indication of the possibility of multilayer adsorption (Akpomie and Conradie 2020). The R2 values obtained from Freundlich isotherm for the studied dyes are higher than those of other models, indicating a good interpretation of the adsorption data. This suggests the heterogeneity of the CS surface and further explains the likelihood of multimolecular layer formation on the surface of the material (Akpomie et al. 2017). The values of n (between 1 and 10) obtained for the sorption of the dyes under study indicate favorable adsorption of the dye molecule on the adsorbent (Ezekoye et al. 2020). The Langmuir isotherm assumes a homogenous surface of the adsorbent and a formation of monolayer adsorption (Dawodu and Akpomie 2016). The R2 values obtained were lower than those of Freundlich for both dye molecules, indicating that Langmuir isotherm did not present a perfect description of the experimental adsorption data. The Temkin Isotherm uses the heat of adsorption to determine the mechanism and the likely interaction between the adsorbent and adsorbate. The R2 values obtained from Temkin isotherm for both dyes were lower than those of Frendulich and Langmuir isotherm models, indicating a poor description of the adsorption data for sunset yellow and tartrazine dyes. The Dubinin–Radushkevich model gives the description of the mechanism of adsorption through the values of its mean free energy. It was observed that the R2 values obtained from the Dubinin–Radushkevich model were lower compared to those of Freundlich isotherm. Table 2 shows the comparison of the adsorption uptake capacity of CS for tartrazine and sunset yellow with other materials. It is obvious that CS displayed a higher affinity for tartrazine than most of the materials used for tartrazine adsorption. However, that of sunset yellow was low than the materials used so far. This indicates the potential application of CS as a low-cost materials for the efficient decontamination of wastewater contaminated with tartrazine.
Effect of adsorbent dose
The size and amount of adsorbent are among the most critical parameters for rapid, efficient, and optimum dyes removal. The adsorbent dose determines the capacity of adsorbent for a given initial concentration of dye, and it is an important parameter in adsorption studies. The impact of dosage on the adsorption of sunset yellow and tartrazine onto CS is shown in Fig. 5. The uptake capacity of both dyes on the surface of CS decreased with increasing in material dose. With an increasing CS dosage from 0.1 to 0.5 g, a decrease in the uptake capacity from 22.2 to 4.49 mg/g for sunset yellow and from 29.4 to 5.78 mg/g for tartrazine was obtained. The decrease in the uptake capacity of CS for both anionic dyes with increasing CS dosage is ascribed to the decrease in the effective participation of the adsorption sites of CS as a result of aggregation/overlapping of the sites at higher doses (Mohammed et al. 2019; Mondal 2017). Therefore, at lower CS doses there is a more efficient use of the active sites of CS resulting in higher dye loading. It is important to keep in mind that as the material dosage increases, the concentration of both sunset yellow and tartrazine remains the same. This promotes less loading on the specific-active sites of the material and thus less utilization of the sites (Ezekoye et al. 2020). This accounted for the reason why we selected 0.1 g of CS for the adsorption study to ensure optimum use of the adsorption sites. A similar trend was also documented by other researchers in the removal of heavy metals (Abdus-Salam and Adekola 2018; Bayuo et al. 2019; Ozsin et al. 2019), and dyes (Terangpi and Chakraborty 2016; Uddin et al. 2017) from polluted water.
Effect of temperature
The influence of temperature was carried at temperature ranges of 27–70 °C. The graph in Fig. 6 indicates that the adsorption rate decreases with an increase in temperature from (22.46–21.96 mg/g) for sunset yellow dye. This could be attributed to the weakening of the bonds that bind the dyes to the active sites on the adsorbent as temperature increases (Sujana and Anand 2011; Bhaumik and Mondal 2014). This is because the increasing temperature is associated with an increase in kinetic energy, which leads to the dissociation of some physically adsorbed dye molecules from the surface of the adsorbent (Khazri et al. 2016; Akpomie and Conradie 2020). Related result has also been reported (Arami et al. 2005). A decrease in adsorption capacity was observed with an increase in temperature implying an exothermic nature of the reaction. Furthermore, an initial increase in amount adsorbed was observed for tartrazine dye with an initial increase in temperature from 27 to 40 0C. This correlates with the fact that the increase in temperature of solution aids in dilation of the pores on the adsorbent leading to an increased the rate of adsorption. But it was observed that with further increase in temperature, rate of adsorption decreased, which could still be attributed to the fact that an increase in temperature causes dissociation of the dyes from the surface of CS as a result of increasing kinetic energy (Akpomie and Conradie 2020).
Effect of contact time
The effect of contact time on uptake of dyes onto any material is vital in order to detect when equilibrium is attained (Akpomie et al. 2017). The effect of the contact time on the uptake of sunset yellow and tartrazine dyes onto CS was inferred as shown in Fig. 7. It can be seen that the rate of sequestration of the dyes from solution was originally quick and then slowly lessened until an equilibrium was attained, after which there was no meaningful increase in the sorption rate. The fast adsorption rate observed at the initial stages is attributing to the vacant and abundant surface-active sites on the adsorbent, which becomes saturated as the time increases implying attainment of adsorption equilibrium (Gupta et al. 2010). It was observed that the same equilibrium times were observed for the two dyes. The equilibrium adsorption capacity of sunset yellow molecules is 22.38 mg/g at 90 min and 30 mg/g for tartrazine at 90 min. A contact time of 2 h was applied in our experiments to ensure equilibrium removal of both dye molecules on CS.
Kinetic analysis
Kinetics provides valuable data for the description of the mechanism and rate of the adsorption process. In the present study, pseudo-first-order (PF), pseudo second-order (PS), liquid-film diffusion (FD), and intraparticle diffusion (ID) models were used to investigate the mechanism of adsorption. The kinetic rate constants are presented in Table 3. The regression (R2) values recorded for both dye molecules showed that PF model did not provide a perfect fit to the experimental data. The experimental value of equilibrium sorption capacity, qe(exp) showed great discrepancy from the calculated values, qe(cal) for PF model for both sunset yellow and tartrazine dyes. The PS model was also tested to evaluate the rate and mechanism of uptake. The linear regression coefficients obtained showed that the PS model presented the best fit to the experimental data. Also, the values of qe(exp) were closer to the values of qe(cal) compared to that of PF model indicating the best fit to removal data. This good fit provided by PS suggests that the mass transfer mechanisms (both internal and external) were important factors in uptake of both dyes on CS and that chemisorption may be the rate-determining step for the sorption process. (Das and Mondal 2011). Intraparticle diffusion model assumes that the dye molecules diffuse inward into the porous adsorbent. The values of the R2 recorded for the uptake of both dyes onto CS were low (R2 < 0.750), indicating that ID is not the dominant mechanism for the removal of the dye molecules onto CS. Besides, the ID model had intercept values indicating the involvement of surface sorption process (Yadav et al. 2014). The R2 values obtained for the FD model for both dyes were lower than those of the PS model, but the R2 value obtained for the sorption of tartrazine molecules was higher than that of the ID model. This implies that FD (surface phenomenon) played more roles in the uptake process of tartrazine molecules onto CS than the ID. Nevertheless, the best fit expressed by the PS models indicates chemisorption as the main process for the uptake of tartrazine and sunset yellow dye molecules onto CS (Okeola et al. 2017).
Conclusion
This study investigated the use of a low-cost material (CS), which is an activated carbon derived from cassava sievate biomass for the abstraction of tartrazine and sunset yellow anionic dyes from simulated dye polluted water. The FT-IR spectrum of CS displayed a potent surface functional group required for efficient uptake of tartrazine and sunset yellow dyes onto CS. Besides, the SEM morphology of CS exhibited a porous structure favorable for the dye uptake on CS adsorbent. The sequestration of sunset yellow and tartrazine dyes on CS attained equilibrium uptake at 90 min. Lower pH values (1.0–4.0) were found to be most favorable for the uptake of both dyes on CS. Besides the optimum adsorption of both tartrazine and sunset yellow was achieved at a high dye concentration of 150 mg/L. On the other hand, lower CS doses were more favorable for efficient uptake of both dyes. Thus, decreasing adsorption capacity of CS from 22.2 to 44.9 mg/g for sunset yellow and 29.4 to 5.78 mg/g with increasing CS dosage from 0.1 to 0.5 mg/g was exhibited. In addition, optimum solution temperatures of 30 and 40 °C were most favorable for the removal of sunset yellow and tartrazine anionic dyes, respectively. The Freundlich model was more suited in the isotherm modeling of the dyes uptake on CS. A maximum monolayer tartrazine uptake on CS of 20.83 mg/g was obtained, which was found to be significantly higher than must documented materials. However, a low monolayer uptake capacity of 0.091 mg/g was reported and was lower than most materials used in the adsorption of sunset yellow. The kinetics of dye's uptake on CS was well modeled by the pseudo-second-order kinetic model. The findings of this research showed the efficiency of CS in the decontamination of waters polluted with sunset yellow and tartrazine anionic dyes, most especially for the latter.
References
Abdus-Salam N, Adekola SK (2018) Adsorption studies of zinc (II) on magnetite, baobab (Adansonia digitata) and magnetite–baobab composite. Appl Water Sci 8:222
Akpomie KG, Onoabedje EA, Alumona TN, Alum OL, Okagu OD, Ezeofor CC (2017) Attenuation of methylene blue from aqua-media on acid activated montmorillonite of Nigerian origin. J Environ Sci Manag 20:17–27
Akpomie KG, Conradie J (2020) Banana peel as a biosorbent for the decontamination of water pollutants. A review. Environ Chem Lett 18:1085–1112
Ani JU, Akpomie KG, Okoro UC, Aneke LE, Onukwuli OD, Ujam OT (2020) Potentials of activated carbon produced from biomass materials for sequestration of dyes, heavy metals, and crude oil components from aqueous environment. Appl Water Sci 10:69
Arami M, Limaea NY, Mahmoodi NM, Salman N, Tabrizi. (2005) Removal of dyes from coloured textile waste water by orange peel adsorbent: equilibrium and kinetic studies. J Colloid Interface Sci 288:371
Banerjee S, Chattopadhyaya MC (2017) Adsorption characteristics for the removal of a toxic dye, tartrazine from aqueous solution by a low-cost agricultural by-product. Arab J Chem 10:S1629–S1638
Bayuo J, Pelig-Ba KB, Abukari MS (2019) Adsorptive removal of chromium (VI) from aqueous solution unto groundnut shell. Appl Water Sci 9:107
Bhaumik R, Mondal NK (2014) Optimizing adsorption of fluoride from water by modified banana peel dust using response surface modelling approach. Appl Water Sci 6:115–135
Caliman FA, Apostol LC, Bulgariu D, Bulgariu L, Gavrilescus M (2009) Sorption of acid yellow 23 from aqueous solution onto soil. FINIDAD 544:465–473
Caliman FA, Apostol LC, Hilihor RM, Gavirilescus M (2008) Factors influencing the sorption of anionic azo dyes tartrazine from aqueous solution onto natural clay. Mater Process Innov 28:455–460
Chukwuemeka-okorie HO, Ekemezie PN, Akpomie KG, Olikagu CS (2018) Calcined corncob-kaolinite combo as new sorbent for sequestration of toxic metalions from polluted aqua media and desorption. Front Chem 6:1–13
Das B, Mondal NK (2011) Calcareous soil as a new adsorbent to remove lead from aqueous solution, equilibrium and thermodynamic study. Univ J Environ Res Technol 1(4):515–530
Dawodu FA, Akpomie KG (2016) Evaluating the potential of a Nigerian soil as an adsorbent for tartrazine dye: Isotherm, kinetic and thermodynamic studies. Alexandr Eng J 55(4):3211–3218
Dotto GL, Vieira MLG, Pinto LAA (2012) Kinetics and mechanism of tartrazine adsorption on chitin and chitosan. Ind Eng Chem Res 51:6599–6608
Ezekoye OM, Akpomie KG, Eze SI, Chukwujindu CN, Ani JU, Ujam OT (2020) Biosorptive interaction of alkaline modified Dialium guineense seed powders with ciprofloxacin in contaminated solution: central composite, kinetics, isotherm, thermodynamics, and desorption. Int J Phytorem 22(10):1028–1037
Ghaedi M (2012) Comparison of cadmium hydroxide nanowires and silver nanoparticles loaded on activated carbon as new adsorbents for efficient removal of Sunset Yellow: kinetics and equilibrium study. Spectrochim Acta Part A 94:346–351
Greluk M, Hubicki Z (2011) Efficient removal of Acid Orange 7 dye from water using the strongly basic anion exchange resin Amberlite IRA-95. Desalination 278:219
Gupta VK, Jain R, Shrivastava M, Nayak A (2010) Equilibrium and thermodynamic studies on the adsorption of the dye tartrazine onto waste coconut husks carbon and activated carbon. J Chem Eng Data 55:5083
Hamid D, Amineh M, Issa M (2014) Removal of anionic dye from wastewater by adsorption on modified rice husk and sugarcane bagasse. Int J Hybrid Inf Technol 7(2):309–320
Khazri H, Ghorbel-Abid I, Kalfat R, Trabelsi-Ayadi M (2016) Removal of ibuprofen, naproxen and carbamazepine in aqueous solution onto natural clay: equilibrium, kinetics, and thermodynamic study. Appl Water Sci 7:3031–3040
Konga Q, Liuc Q, Miaoc MS, Liua YZ, Chend QF, Zhaod C (2017) Kinetic and equilibrium studies of the biosorption of sunset yellow dye by alligator weed activated carbon. Desalinat Water Treat 66:281–290
Lata H, Garg VK, Gupta RK (2008) Adsorptive removal of basic dye by chemically activated Parthenium biomass: equilibrium and kinetic modeling. Desalination 219:250
Mittal A, Kurup L, Mittal J (2007) Freundlich and Langmuir adsorption isotherms and kinetic for the removal of tartrazine from aqueous solution using hen feathers. J Hazard Mater 146:243–250
Mittal J, Mittal L, Kurup L (2006) Adsorption isotherm kinetics and column operations for the removal of hazardous dye, tartrazine from aqueous solution using waste materials-Bottom ash and de-oiled soya, as adsorbents. J Hazard Mater B136:567–578
Mohammed AA, Al-Musawi TJ, Kareem SL, Zarrabi MA (2019) Simultaneous adsorption of tetracycline, amoxicillin, and ciprofloxacin by pistachio shell powder coated with zinc oxide nanoparticles. Arab J Chem 13(3):4629–4643
Mondal NK (2017) Natural banana (Musa acuminate) peel: an unconventional adsorbent for removal of fluoride from aqueous solution through batch study. Water Conserv Sci Eng 1:223–232
Ojo TA, Ojedokun AT, Bello OS (2019) Functionalization of powdered walnut shell with orthophosphoric acid for congo red dye removal. Part Sci Technol 37:74–85
Okeola FO, Odebunmi EO, Ameen OM, Amoloye MA, Lawal AA, Abdulmummeen AG (2017) Equilibrium kinetics and thermodynamic studies of the adsorption of tartrazine and sunset yellow. Arid Zone J Eng Technol Environ 13(2):268–280
Ozsin G, Kilic M, Apaydin-Varol E, Putun AE (2019) Chemically activated carbon production from agricultural waste of chickpea and its application for heavy metal adsorption: equilibrium, kinetic, and thermodynamic studies. Appl Water Sci 9:56
Ravishankar H, Wang J, Shu L, Jagatheesan V (2016) Removal of Pb(II) ion using polymer based grapheme oxide magnetic nano-sorbent. Process Saf Environ Prot 104:472–480
Salvi NA, Chattopadhyay S (2016) Biosorption of Azo dyes by spent Rhizopus arrhizus biomass. Appl Water Sci 7:3041–3054
Seey TL, Kassim MJNM (2012) Acidic and basic dyes removal by adsorption on chemically treated mangrove barks. Int J Appl Sci Technol 2:270–276
Sujana MG, Anand S (2011) Fluoride removal studies from contaminated ground water by using bauxite. Desalination 267(2–3):222–227
Sulak MT, Demirbas E, Kobya M (2007) Removal of Astrazon yellow 7GL from aqueous solutions by adsorption onto wheat bran. Biores Technol 98:2590
Terangpi P, Chakraborty S (2016) Adsorption kinetics and equilibrium studies for removal of acid azo dyes by aniline formaldehyde condensate. Appl Water Sci 7:3661–3671
Torres-Perez J, Huang Y, Hadi P, Mackey H (2018) Equilibrium, Kinetic and Optimization studies for the adsorption of tartrazine in water onto activated carbon from pecan nut shells. Water Air Soil Pollut 229:73
Uddin MT, Rahman MA, Rukanuzzaman M, Islam MA (2017) A potential low cost adsorbent for the removal of cationic dyes from aqueous solutions. Appl Water Sci 7:2831–2842
Wawrzkiewicz M (2011) Sorption of Sunset Yellow dye by weak base anion exchanger–kinetic and equilibrium studies. Environ Technol 32(4):455–465
Yadav SK, Singh DK, Sinha S (2014) Chemical carbonization of papaya seed originated charcoals for sorption of Pb(II) from aqueous solution. J Environ Chem Eng 2:9–19
Zhang W, Li H, Kan X, Dong L, Yan H, Jiang Z, Yang H, Li A, Cheng R (2012) Adsorption of anionic dyes from aqueous solutions using chemically modified straw. Biores Technol 117:40
Funding
The author(s) received no specific funding for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chukwuemeka-Okorie, H.O., Ekuma, F.K., Akpomie, K.G. et al. Adsorption of tartrazine and sunset yellow anionic dyes onto activated carbon derived from cassava sievate biomass. Appl Water Sci 11, 27 (2021). https://doi.org/10.1007/s13201-021-01357-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-021-01357-w