Abstract
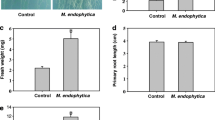
Azospirillum sp. is one of the most studied genera of plant growth-promoting rhizobacteria (PGPR). The ability of Azospirillum sp. to promote plant growth has been associated with its ability to produce several phytohormones, such as auxins, gibberellins and cytokinins, but mainly indole-3-acetic acid (IAA). It has been propoosed that the production of IAA explains the positive effects of co-inoculation with Azospirillum sp. on the rhizobia-legume symbiosis. In this study, we constructed an IAA-deficient mutant of A. brasilense Az39 (ipdC −) by using a restriction-free cloning method. We inoculated soybean seeds with 1·106 cfu·seed−1 of Bradyrhizobium japonicum E109 and co-inoculating leaves at the V3 stage with 1·108 cfu.plant−1 of A. brasilense Az39 wt or ipdC − or inoculated leaves with 20 μg.plant−1 synthetic IAA. The results confirmed soybean growth promotion as there was increased total plant and root length, aerial and root dry weight, number of nodules on the primary root, and an increase in the symbiosis established with B. japonicum E109. Nodule weight also increased after foliar co-inoculation with the IAA- producer A. brasilense Az39. The exogenous application of IAA decreased aerial and root length, as well as the number of nodules on primary roots in comparison with the Az39 wt strain. These results allow us to propose a biological model of response to foliar co-inoculation of soybean with IAA-producing rhizobacteria. This model clearly shows that both the presence of microorganism as part of the colonization process and the production of IAA in situ are co-responsible, via plant signaling molecules, for the positive effects on plant growth and symbiosis establishment.





Similar content being viewed by others
References
Bashan Y, de Bashan L (2010) How the plant growth-promoting bacterium Azospirillum promotes plant growth. A critical assessment. Adv Agron 108:77–136. https://doi.org/10.1016/S0065-2113(10)08002-8
Bottini R, Fulchieri M, Pearce D, Pharis R (1989) Identification of gibberellins A1, A3, and Iso-A3 in cultures of a. Lipoferum. Plant Physiol 90(1):45–47. https://doi.org/10.1104/pp.90.1.45
Burdmann S, Kigel J, Okon Y (1997) Effects of Azospirillum brasilense on nodulation and growth of common bean (Phaseolus vulgaris L.) Soil Biol Biochem 29(5-6):923–929. https://doi.org/10.1016/S0038-0717(96)00222-2
Cacciari I, Lippi D, Pietrosanti T, Pietrosanti W (1989) Phytohormone-like substances produced by single and mixed diazotrophic cultures of Azospirillum and Arthrobacter. Plant Soil 115(1):151–153. https://doi.org/10.1007/BF02220706
Campo RJ, Araujo RS, Hungria M (2009) Nitrogen fixation with the soybean crop in Brazil: compatibility between seed treatment with fungicides and bradyrhizobial inoculants. Symbiosis 48(1):154–163. https://doi.org/10.1007/BF03179994
Carreño-Lopez R, Campos-Reales N, Elmerich C, Baca B (2000) Physiological evidence for differently regulated tryptophan-dependent pathways for indole-3-acetic acid synthesis in Azospirillum brasilense. Mol Gen Genet 2 64(4):521–530
Cassán F, Díaz-Zorita M (2016) Azospirillum sp. in current agriculture: From the laboratory to the field. Soil Biol Biochem 103:117–130. https://doi.org/10.1016/j.soilbio.2016.08.020
Cassán F, Maiale S, Masciarelli O, Vidal A, Luna V, Ruiz O (2009) Cadaverine production by Azospirillum brasilense and its possible role in plant growth promotion and osmotic stress mitigation. Eur J Soil Biol 45(1):12–19. https://doi.org/10.1016/j.ejsobi.2008.08.003
Cassán F, Penna C, Creus C, Radovancich D, Monteleone E. et al. (2013) Inoculantes forulados con Azospirillum sp. In: Albanesi, A; Benintende, S; Cassán F. y Perticari A. (Eds). Manual de procedimientos microbiológicos para la evaluación de inoculantes. pp 25–34. Editorial: Asociación Argentina de Microbiología. ISBN: 978-987-26716-4-8
Cassán F, Vanderleyden J, Spaepen S (2014) Physiological and agronomical aspects of phytohormone production by model plant-growth-promoting rhizobacteria (PGPR) belonging to the genus Azospirillum. Plant Growth Regul 33(2):440–459. https://doi.org/10.1007/s00344-013-9362-4
Costacurta A, Keijers V, Vanderleyden J (1994) Molecular cloning and sequence analysis of an Azospirillum brasilense indole-3-pyruvate deccarboxylase. Mol Gen Genet 243(4):463–472
Crozier A, Arruda P, Jasmim JM, Monteiro AM, Sandberg G (1988) Analysis of indole-3-acetic acid and related indoles in culture medium from Azospirillum lipoferum and Azospirillum brasilense. Appl Environ Microbiol 54(11):2833–2837
Dart P (1977) Infection and development of leguminous nodules. In: Hardy RWI, Silver WS (eds) A tratise of dinitrogen fixation section III-biology. Wiley, New York, pp 367–372
Di Rienzo J.A.; Casanoves, F; Balzarini, M.G; Gonzalez, L; Tablada, M; Robledo, C.W. InfoStat versión 2014. Córdoba: Grupo InfoStact, FCA, Universidad Nacional de Córdoba, Argentina. URL http://www.infostat.com.ar
Dobbelaere S, Croonenborghs A, Thys A, Broek AV et al (1999) Phytostimulatory effect of Azospirillum brasilense wild type and mutant strains altered in IAA production on wheat. Plant Soil 212(2):153–162. https://doi.org/10.1023/A:1004658000815
Döbereiner J, Baldani VLD, Baldani JI (1995) Como isolar e identificar bactérias diazotróficas de plantas não-leguminosas. Brasília: EMBRAPA-SPI: EMBRAPA-CNPAB, Itaguaí, RJ, 60p
El-Saeid H, Abou-Hussein S, El-Tohamy W (2010) Growth characters, yield and endogenous hormones of cowpea plants in response to IAA application. Res J Agric Biol Sci 6:27–31
Fehr, WR, Caviness CE (1977) Stages of soybean development. Cooperative Extension Service, Agriculture and Home Economics Experiment Station. Iowa State University, Ames, Iowa
Frugier F, Kosuta S, Murray JD, Crespi M, Szczyglowski K (2008) Cytokinin: secret agent of symbiosis. Trends Plant Sci 13(3):115–120. https://doi.org/10.1016/j.tplants.2008.01.003
Gruodien J, Zvironaite V (1971) Effect of IAA on growth and synthesis of N compounds in Lucerne. Luk TSR Aukstuju Mosklo Darbai Biologia 17:77–87
Hirsch A, Fang Y, Asad S, Kapulnik Y (1997) The role of phytohormones in plant-microbe symbioses. Plant Soil 194(1/2):171–184. https://doi.org/10.1023/A:1004292020902
Hoagland D, Arnon D (1950) The Water-Culture Method for Growing Plants without Soil. California Agricultural Experiment Station. Circular 347:1–32
Horemans S, Koninck K, Neuray J, Hermans R et al (1986) Production of plant growth substances by Azospirillum sp. and other rhizophere bacteria. Symbiosis 2:341–346
Hubbell D, Tien T, Gaskins M, Lee J (1979) Physiological interaction in the Azospirillum-grass root association. In: Vose P, Ruschel A (eds) Associative N2-fixation. CRC Press, Boca Raton, pp 1–6
Hungria M, Nogueira M, Araujo R (2015) Soybean seed co-inoculation with Bradyrhizobium spp. and Azospirillum brasilense: a new biotechnological tool to improve yield and sustainability. Am J Plant Sci 6(06):811–817. https://doi.org/10.4236/ajps.2015.66087
Hussain K, Hussain M, Nawaz K, Majeed A et al (2011) Morphochemical response of Chaksu (Cassia absus L.) to different concentrations of indole acetic acid (IAA). Pak J Bot 43:1491–1493
Janzen R, Rood S, Dormar J, McGill W (1992) Azospirillum brasilense produces gibberellins in pure culture and chemically-medium and in co-culture on straw. Soil Biol Biochem 24(10):1061–1064. https://doi.org/10.1016/0038-0717(92)90036-W
Kloepper J, Schroth M (1978) Plant growth-promoting rhizobacteria on radishes. In: Proceedings of the 4th international conference on plant pathogenic bacteria Vol, 2, pp 879–882
Kolb W, Martin P (1985) Response of plant roots to inoculation with Azospirillum brasilense and to application of indole acetic acid. In: Azospirillum III. Springer Berlin Heidelberg, pp 215–221. https://doi.org/10.1007/978-3-642-70791-9_20
Lodeiro AR (2015) Interrogantes en la tecnología de la inoculación de semillas de soja con Bradyrhizobium spp. Rev Argent Microbiol 47(3):261–273. https://doi.org/10.1016/j.ram.2015.06.006
López-García S, Perticari A, Piccinetti C, Ventimiglia L et al (2009) In-furrow inoculation and selection for higher motility enhances the efficacy of Bradyrhizobium japonicum nodulation. Agron J 101(2):357–363. https://doi.org/10.2134/agronj2008.0155x
Mathesius U, Schlaman H, Spaink H, Of Sautter C et al (1998) Auxin transport inhibition precedes root nodule formation in white clover roots and is regulated by flavonoids and derivatives of chitin oligosaccharides. Plant J 14(1):23–34. https://doi.org/10.1046/j.1365-313X.1998.00090.x
Miles A, Misra S (1938) The estimation of the bactericidal power of blood. J Hyg 38(06):732–737. https://doi.org/10.1017/S002217240001158X
Muniralzaman M (2000) Effect of CyCoCel (CCC) on the growth and yield manipulations of vegetable soybean. ARG Traininy. Kasetsart university kamphaen sean. Nakhon phathom, Thailand, pp 1–16
Patten C, Glick B (1996) Bacterial biosynthesis of indole-3-acetic acid. Can J Microbiol 42(3):207–220. https://doi.org/10.1139/m96-032
Patten C, Glick B (2002) The role of Pseudomonas putida indoleacetic acid in the development of the host plant root system. Appl Environ Microbiol 68(8):3795–3801. https://doi.org/10.1128/AEM.68.8.3795-3801.2002
Perrig D, Boiero M, Masciarelli O, Penna C et al (2007) Plant-growth-promoting compounds produced by two agronomically important strains of Azospirillum brasilense, and implications for inoculant formulation. Appl Microbiol Biotechnol 75(5):1143–1150. https://doi.org/10.1007/s00253-007-0909-9
Piccoli P, Bottini R (1996) Gibberellins production in A. lipoferum cultures y enhanced by light. Biocell 20:185–190
Prinsen E, Costacurta A, Michiels K, Vanderleyden J et al (1993) Azospirillum brasilense indole-3-acetic acid biosynthesis: evidence for a non-tryptophan dependent pathway. Mol Plant-Microb Interact 6(5):609–615. https://doi.org/10.1094/MPMI-6-609
Remans R, Beebe S, Blair M, Manrique G, Tovar E, Rao I, Croonenborghs A, Torres-Gutierrez R, el Howeity M, Michiels J, Vanderleyden J (2008) Physiological and genetic analysis of root responsiveness to auxin-producing plant growth-promoting bacteria in common bean (Phaseolus vulgaris L.) Plant Soil 302(1-2):149–161. https://doi.org/10.1007/s11104-007-9462-7
Rivera D, Revale S, Molina R, Gualpa J et al (2014) Complete genome sequence of the model rhizosphere strain Azospirillum brasilense Az39, successfully applied in agriculture. Genome announcements 2(4):e00683–e00614
Rodríguez Cáceres E (1982) Improved medium for isolation of Azospirillum spp. Appl Environ Microbiol 44:990–991
Sadasivan L, Neyra C (1985) Floculation in Azospirillum brasilense and Azospirillum lipoferum: exopolysaccharides and cyst formation. J Bacteriol 163(2):716–723
Schmidt W, Martin P, Omay H, Bangerth F (1988) Influence of Azospirillum brasilense on nodulation of legumes. In: Kling-Müller W (ed) Azospirillum IV. Genetics, physiology, ecology. Springer, Heidelberg, pp 92–100
Simon R, Priefer U, Pühler A (1983) A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram negative bacteria. Biotechnology 1(9):784–790. https://doi.org/10.1038/nbt1183-784
Spaepen S, Vanderleyden J, Remans R (2007) Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol 31(4):425–448. https://doi.org/10.1111/j.1574-6976.2007.00072.x
Spaepen S, Dobbelaere S, Croonenborghs A, Vanderleyden J (2008) Effects of Azospirillum brasilense indole-3-acetic acid production on inoculated wheat plants. Plant Soil 312(1–2):15–23. https://doi.org/10.1007/s11104-008-9560-1
Srinivasan P, Gopal K (1977) Effect of plantofix and NAA formulation on groundnut var TMU-7. Curr Sci 46:119–120
Srinivasan M, Holl F, Petersen D (1996) Influence of indoleacetic-acid-producing Bacillus isolates on the nodulation of Phaseolus vulgaris by Rhizobium etli under gnotobiotic conditions. Can J Microbiol 42(10):1006–1014. https://doi.org/10.1139/m96-129
Thimann K (1936) Auxins and the growth of roots. Am J Bot 23(8):561–569. https://doi.org/10.2307/2436087
Tien T, Gaskina M, Hubbell D (1979) Plant growth substances produced by Azospirillum brasilense and their effect on the growth of pearl millet (Pennisetum Americanum L). Appl Environ Microbiol 37(5):1016–1024
Torres D, Revale S, Obando M, Maroniche G et al (2015) Genome sequence of Bradyrhizobium japonicum E109, one of the most agronomically used nitrogen-fixing rhizobacteria in Argentina. Genome announcements 3(1):e01566–e01514
Unger T, Jacobovitch Y, Dantes A, Bernheim R (2010) Applications of the restriction free (RF) cloning procedure for molecular manipulations and protein expression. J Struct Biol 172(1):34–44. https://doi.org/10.1016/j.jsb.2010.06.016
Vicario J, Gallarato L, Paulucci N, Perrig D et al (2015) Co-inoculation of legumes with Azospirillum and symbiotic rhizobia. In: Cassán F, Okon Y, Creus C (eds) Handbook for Azospirillum. Springer, Cham
Vincent JM (1970) A manual for the practical study of the root-nodule bacteria. Blackwell Scientific Publications, Oxford
Yahalom E, Okon Y, Dovrat A (1990) Possible mode of action of Azospirillum brasilense strain cd on the root morphology and nodule formation in burr medic (Medicago polymorpha). Can J Microbiol 36(1):10–14. https://doi.org/10.1139/m90-003
Zilli JÉ, Ribeiro KG, Campo RJ, Hungria M (2009) Influence of fungicide seed treatment on soybean nodulation and grain yield. Revista Brasileira de Ciência do Solo 33(4):917–923. https://doi.org/10.1590/S0100-06832009000400016
Zimmer W, Bothe H (1988) The phytohormonal interactions between Azospirillum and wheat. Plant Soil 110(2):239–247. https://doi.org/10.1007/BF02226804
Acknowledgements
We thank Universidad Nacional de Río Cuarto, Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICET), Fondo Nacional de Ciencia y Tecnología (FONCyT) and Instituto Nacional de Tecnología Agropecuaria (INTA, Argentina). Fabricio Cassán is a Researcher of CONICET at the Universidad Nacional de Río Cuarto. Gaston Lopez is a postdoctoral researcher; José Gualpa and Romina Molina are PhD students at the Universidad Nacional de Río Cuarto and granted by CONICET.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors report no conflicts of interest.
Electronic supplementary material
ESM 1
Table S1 Primers used in this study. (DOCX 14 kb)
ESM 2
Fig. S1: Scanning electron microscopy (SEM) of soybean leaf showing A. brasilense Az39 colonizing the surface of leaves 48 h after inoculation. The white arrows indicate the presence of bacteria on the plant tissue. A and B represent different fractions of the leaf. Colonization ability was confirmed for both A. brasilense Az39 wt and the IAA–deficient mutant ipdC − (not shown). (DOCX 756 kb)
Rights and permissions
About this article
Cite this article
Puente, M.L., Gualpa, J.L., Lopez, G.A. et al. The benefits of foliar inoculation with Azospirillum brasilense in soybean are explained by an auxin signaling model. Symbiosis 76, 41–49 (2018). https://doi.org/10.1007/s13199-017-0536-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-017-0536-x