Abstract
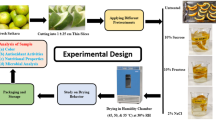
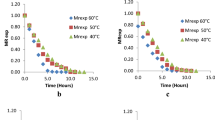
The aim of the present study was to assess the drying kinetic of tucum fruits (epicarp and mesocarp) Astrocaryum aculeatum Meyer at three different temperatures (50, 60, and 70 °C). The physicochemical characterization, water activity, moisture content, including β-carotene and vitamin C content in-natura and dried fruits were analyzed. The fruit fractions presented high β-carotene, protein and lipid levels. Fatty acid profile showed oleic acid as the major fatty acid. Different mathematical models were computed to assess the drying process. The Page model was observed to be the best to describe the drying kinetic with the highest correlation coefficient (R2) 0.99 and the least Chi squared (χ2) close to 105 at the studied temperatures. The drying process reduced water activity to desirable levels in all trials and β-carotene retentions after drying remained at satisfactory levels, fact that resulted in minimum value of 63% and approximately 94% in some cases. Vitamin C retention was comparatively more around 20–40% compared to control.


Similar content being viewed by others
References
Akpinar EK (2008) Mathematical modelling and experimental investigation on sun and solar drying of white mulberry. J Mech Sci Technol 22(8):1544–1553
AOAC—Association of Official Analytical Chemists (1998) Official method 942.15 acidity (titratable) of fruit products, 16th edn, AOAC, Washington, DC
AOAC—Association of Official Analytical Chemists (2001) Official methods 996.06 fat (total, saturated and unsaturated in foods) hydrolytic extraction gas chromatographic method. First Action 1996. Revised 2001
AOAC—Association of Official Analytical Chemists (2002) Official methods 967.21 vitamin C (ascorbic acid) in vitamin preparations and juices: 2,6-dichloroindophenol titrimetric method final action, 17th edn, AOAC, Washington, DC
Aral S, Beşe AV (2016) Convective drying of hawthorn fruit (Crataegus spp.): effect of experimental parameters on drying kinetics, color, shrinkage, and rehydration capacity. Food Chem 210:577–584
Barbosa-Canovas G, Fernandez-Molina JJ, Alzamora SM, Tapia MS, Lopez-Malo A, Welti Chanes J (2003) Handling and preservation of fruits and vegetables by combined methods for rural areas: technical manual. FAO, Roma
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917
Costa JR, da Leeuwen JV, Costa JA (2011) Tucumã of Amazonas Astrocaryum aculeatum G. Mey. In: Shanley P, Cymerys M, Serra M, Medina G (eds) Fruit trees and useful plants in Amazonian life. FAO, Rome, pp 205–2013
Couto MAL, Canniatti-Brazaca SG (2010) Quantification of vitamin C and antioxidant capacity of citrus varieties. Ciênc Technol Aliment 30(1):15–19
Didonet AA (2012) O mercado de um produto florestal não madeireiro e o resíduo sólido gerado pela sua comercialização: o caso do tucumã (Astrocaryum aculeatum G. Mey) nas feiras de Manaus. Dissertação (Mestrado em Ciências de Florestas Tropicais) Manaus–AM, Instituto Nacional de Pesquisas da Amazônia—INPA, pp 77
Didonet AA, Ferraz IDK (2014) Fruit trade of tucuma (Astrocaryum aculeatum g. Mey—arecaceae) at local market-places in Manaus (Amazonas, Brazil). Rev Bras Frutic 36(2):353–362
FAO (1986) Food and fruit bearing forest species, 3. Examples from Latin America, FAO forestry paper 44/3, Rome
Ferrer A, Remón S, Negueruela AI, Oria R (2005) Changes during the ripening of the very late season Spanish peach cultivar Calanda: feasibility of using CIELAB coordinates as maturity indices. Sci Hortic 105(4):435–446
Hall NT, Smoot JM, Knight RJ, Nagy S (1980) Protein and aminoacid compositions of ten tropical fruit by gas–liquid chromatography. J Agric Food Chem 28:1217–1221
Jobim ML, Santos RC, Alves CFS, Oliveira RM, Mostardeiro CP, Sagrillo MR, Souza Filho OC, Garcia LFM, Manica-Cattani MF, Ribeiro EE, Cruz IBM (2014) Antimicrobial activity of Amazon Astrocaryum aculeatum extracts and its association to oxidative metabolism. Microbiol Res 169:314–323
Kaleta A, Górnicki K (2010) Evaluation of drying models of apple (var. McIntosh) dried in a convective dryer. Int J Food Sci Technol 45(5):891–898
Kirmaci V, Usta H, Menlik T (2008) An experimental study on freeze-drying behavior of strawberries. Dry Technol 26:1570–1576
Kjeldahl JGCT (1883) Neue methode zur bestimmung des stickstoffs in organischen körpern. Fresenius J Anal Chem 22(1):366–382
Lorenzi H, Noblick L, Kahn F, Ferreira E (2010) Flora brasileira Lorenzi: Arecaceae (palmeiras). Instituto Plantarum, Nova Odessa, p 43
Mambrim MCT, Barrera-Arellano D (1997) Caracterización de aceites de frutos de palmeras de la región amazónica del Brasil. Grasas Aceites 48(3):154–158
Manzato L, Rabelo LCA, de Souza SM, da Silva CG, Sanches EA, Rabelo D, Mariuba LAM, Simonsen J (2017) New approach for extraction of cellulose from tucum’s endocarp and its structural characterization. J Mol Struct 1143:229–234
McGuire RG (1992) Reporting of objective color measurements. HortScience 27(12):1254–1255
McMinn WAM, Magee TRA (1999) Principles, methods and applications of the convective drying of foodstuffs. Food Bioprod Process 77(3):175–193
Moussa F, Kahn F (1997) Uso y potencial económico de dos palmas, Astrocaryum aculeatum Meyer y Astrocaryum vulgare Martius, em la Amazônia brasileña. Isntitut Français de Recherche Scientifique pour lê Développement em Coopération (ORSTOM). Brasília, DF, Brasil, pp 101–114
Nowak V, Du J, Charrondière UR (2016) Assessment of the nutritional composition of quinoa (Chenopodium quinoa Willd.). Food Chem 193:47–54
Onwude DI, Hashim N, Janius RB, Nawi NM, Abdan K (2016) Modeling the thin-layer drying of fruits and vegetables: a review. Compr Rev Food Sci Food Saf Banner 15(3):599–618
Rodriguez-Amaya DB (2001) A guide to carotenoid analysis in foods. ILSI Press, Washington, p 65
Rodriguez-Amaya DB, Kimura M, Amaya-Farfan J (2008) Fontes brasileiras de carotenóides: Tabela brasileira de composição de carotenóides em alimentos. Ministério de Meio Ambiente (MMA). http://www.mma.gov.br/estruturas/sbf_agrobio/_publicacao/89_publicacao09032009113306.pdf. Accessed 3th Aug 2017
Sagrillo MR, Garcia LFM, de Souza Filho OC, Duarte MMMF, Ribeiro EE, Cadoná FC, da Cruz IBM (2015) Tucuma fruit extracts (Astrocaryum aculeatum Meyer) decrease cytotoxic effects of hydrogen peroxide on human lymphocytes. Food Chem 173:741–748
Sampaio RM, Neto Monteles JP, Perez VH, Marcos SK, Boizan MA, da Silva LR (2017) Mathematical modeling of drying kinetics of persimmon fruits (Diospyros kaki cv. Fuyu). J Food Process Pres 41:1–7
Santos RD, Gagliardi ACM, Xavier HT et al (2013) I Diretriz sobre o consumo de gorduras e saúde cardiovascular. Arq Bras Cardiol 100(1):1–40
Schroth G, da Mota MSS, Lopes R, de Freitas AF (2004) Extractive use, management and in situ domestication of a weedy palm, Astrocaryum tucum, in the central Amazon. For Ecol Manag 202(1):161–179
Siqueira FC (2014) Obtenção da polpa de tucumã (Astrocaryum vulgare Mart.) desidratada visando a preservação dos carotenoides. Dissertação (Mestrado em Ciência e Tecnologia de Alimentos). Belém, PA, Instituto de Tecnologia, Universidade Federal do Pará—UFPA, pp 79
Vásquez-Parra JE, Ochoa-Martínez CI, Bustos-Parra M (2013) Effect of chemical and physical pretreatments on the convective drying of cape gooseberry fruits (Physalis peruviana). J Food Eng 119(3):648–654
Wilberg VC, Rodriguez-Amaya DB (1995) HPLC quantitation of major carotenoids of fresh and processed guava, mango and papaya. LWT Food Sci Technol 28(5):474–480
Yuyama LKO, Maeda RN, Pantoja L, Aguiar JPL, Marinho HA (2008) Processamento e avaliação da vida-de-prateleira do tucumã (Astrocaryum aculeatum Meyer) desidratado e pulverizado. Ciênc Technol Aliment 28(2):408–441
Yuyama LKO, Aguiar JPL, Filho DFS, Yuyama K, Varejão MJ, Fávaro DIT, Vasconcellos MBA, Pimentel SA, Caruso MSF (2011) Caracterização físico-química do suco de açaí de Euterpe precatoria Mart. oriundo de diferentes ecossistemas amazônicos. Acta Amaz 41(4):545–552
Acknowledgements
The authors are grateful to the following Brazilian agencies: State University of the Northern Rio de Janeiro (UENF) and Research Foundation of Rio de Janeiro State (FAPERJ) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, M.B., Perez, V.H., Pereira, N.R. et al. Drying kinetic of tucum fruits (Astrocaryum aculeatum Meyer): physicochemical and functional properties characterization. J Food Sci Technol 55, 1656–1666 (2018). https://doi.org/10.1007/s13197-018-3077-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-018-3077-2