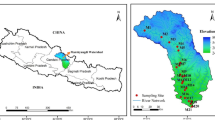
Abstract
Integration of the information from hydrochemical data, statistical analysis and modeling technique is crucial for extending our knowledge of hydrochemistry of karst aquifers. In this paper, conventional graphical methods, statistical analysis and PHREEQC inverse modeling were used to investigate the hydrogeochemical characteristics and evolution processes of karst groundwater in Jinci spring area. The results showed that the dominant hydrochemical types of karst groundwater determined by Piper diagram were Ca·Mg–SO4·HCO3, Ca·Mg–SO4, Ca–SO4, Ca–SO4·HCO3 and Ca–HCO3, and the main components of karst groundwater were dominated by rock–water interactions based on statistical analysis. Ca2+, Mg2+ and HCO3− originated mainly from the dissolution of calcite and dolomite; SO42− came mostly from gypsum dissolution and pyrite oxidation; and the source of Na+ and Cl− could either be major from halite dissolution or cation exchange. Moreover, PHREEQC inverse modeling indicated that calcite, gypsum and CO2 (g) were dissolved, and Ca–Mg exchange prevailed along the flow path (from recharge area to runoff area); the dissolution of gypsum, dolomite and CO2 (g), and halite precipitation, and Na–Mg and Ca–Mg exchange occurred along runoff area to discharge area; and there were the dissolution of carbonate, gypsum, halite and CO2 (g), Na–Mg and Ca–Mg exchange from deep buried area to discharge area.







Similar content being viewed by others
References
Chapelle FH (1983) Groundwater geochemistry and calcite cementation of the Aquia aquifer in southern Maryland. Water Resour Res 19:545–558
Cowell DW, Ford DC (1980) Hydrochemistry of a dolomite karst: the Bruce Peninsula of Ontario. Can J Earth Sci 17:520–526
Gibbs RJ (1970) Mechanisms controlling world water chemistry. Science 170:1088–1090
Han X, Lu R, Li Q (1993) Karst groundwater system: a study on big spring in Shanxi, 1st edn. Geological Publishing House, Beijing (in Chinese)
Hanshaw BB, Back W (1979) Major geochemical processes in the evolution of carbonate-aquifer systems. J Hydrol 43:287–312
Hanshaw BB, Back W (1985) Deciphering hydrological systems by means of geochemical processes. Hydrol Sci J 30:257–271
Hao Y, Zhu Y, Zhao Y et al (2009) The role of climate and human influences in the dry-up of the Jinci springs, China. J Am Water Resour As 45:1228–1237
Hassen I, Hamzaoui-Azaza F, Bouhlila R (2016) Application of multivariate statistical analysis and hydrochemical and isotopic investigations for evaluation of groundwater quality and its suitability for drinking and agriculture purposes: case of Oum Ali-Thelepte aquifer, central Tunisia. Environ Monit Assess 188:135
Jia Z, Zang H, Hobbs P et al (2017) Application of inverse modeling in a study of the hydrogeochemical evolution of karst groundwater in the Jinci spring region, northern China. Environ Earth Sci 76:312
Kenoyer GJ, Bowser CJ (1992) Groundwater chemical evolution in a sandy silicate aquifer in northern Wisconsin: 1. patterns and rates of change. Water Resour Res 28:579–589
Li X, Shu L, Liu L, Yin D, Wen J (2012) Sensitivity analysis of groundwater level in Jinci spring basin (China) based on artificial neural network modeling. Hydrogeol J 20:727–738
Li Z, Wang G, Wang X et al (2018) Groundwater quality and associated hydrogeochemical processes in Northwest Namibia. J Geochem Explor 186:202–214
Lipson DS, Mccray JE, Thyne GD (2007) Using PHREEQC to simulate solute transport in fractured bedrock. Groundwater 45:468–472
Long DT, Saleem ZA (1974) Hydrogeochemistry of carbonate groundwaters of an urban area. Water Resour Res 10:1229–1238
López-Chicano M, Bouamama M, Vallejos A, Pulido-Bosch A (2001) Factors which determine the hydrogeochemical behaviour of karstic springs. A case study from the Betic Cordilleras, Spain. Appl Geochem 16:1179–1192
Lv C, Ling M, Wu Z et al (2018) Analysis of groundwater variation in the Jinci Spring area, Shanxi province (China), under the influence of human activity. Environ Geochem Health 41:921–928
Mallick J, Singh CK, AlMesfer MK et al (2018) Hydro-geochemical assessment of groundwater quality in Aseer Region, Saudi Arabia. Water 10:1847
Martínez DE, Quiroz Londoño OM, Massone HE, Palacio Buitrago P, Lima L (2012) Hydrogeochemistry of fluoride in the Quequen river basin: natural pollutants distribution in the argentine pampa. Environ Earth Sci 65:411–420
Merkel BJ, Friedrich BP (2005) Groundwater geochemistry—a practical guide to modeling of natural and contaminated aquatic systems. Springer, Berlin
Morán-Ramírez J, Ledesma-Ruiz R, Mahlknecht J, Ramos-Leal JA (2016) Rock–water interactions and pollution processes in the volcanic aquifer system of Guadalajara, Mexico, using inverse geochemical modeling. Appl Geochem 68:79–94
Naik PK, Awasthi AK, Anand AVSS, Behera PN (2009) Hydrogeochemistry of the Koyna River basin, India. Environ Earth Sci 59:613–629
Parkhurst DL (1997) Geochemical mole-balance modeling with uncertain data. Water Resour Res 33:1957–1970
Pinault JL, Plagnes V, Aquilina L, Bakalowicz M (2001) Inverse modeling of the hydrological and the hydrochemical behavior of hydrosystems: characterization of karst system functioning. Water Resour Res 37:2191–2204
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analysis. Trans Am Geophys Union 25:914–923
Raju NJ, Patel P, Reddy BCSR, Suresh U, Reddy TVK (2016) Identifying source and evaluation of hydrogeochemical processes in the hard rock aquifer system: geostatistical analysis and geochemical modeling techniques. Environ Earth Sci 75:1157
Rao NS, Rao PS, Reddy GV et al (2012) Chemical characteristics of groundwater and assessment of groundwater quality in Varaha River Basin, Visakhapatnam District, Andhra Pradesh, India. Environ Monit Assess 184:5189–5214
Reddy AGS, Kumar KN (2010) Identification of the hydrogeochemical processes in groundwater using major ion chemistry: a case study of Penna-Chitravathi River basins in southern India. Environ Monit Assess 170:365–382
Scanlon BR (1990) Relationships between groundwater contamination and major-ion chemistry in a karst aquifer. J Hydrol 119:271–291
Scanlon BR (2010) Physical controls on hydrochemical variability in the Inner Bluegrass Karst Region of central Kentucky. Groundwater 27:639–646
Scanlon BR, Thrailkill J (1987) Chemical similarities among physically distinct spring types in a karst terrain. J Hydrol 89:259–279
Shanyengana ES, Seely MK, Sanderson RD (2004) Major-ion chemistry and ground-water salinization in ephemeral floodplains in some arid regions of Namibia. J Arid Environ 57:211–223
Sharif MU, Davis RK, Steele KF et al (2008) Inverse geochemical modeling of groundwater evolution with emphasis on arsenic in the Mississippi River Valley alluvial aquifer, Arkansas (USA). J Hydrol 350:41–55
Singh CK, Kumar A, Shashtri S et al (2017) Multivariate statistical analysis and geochemical modeling for geochemical assessment of groundwater of Delhi, India. J Geochem Explor 175:59–71
Uliana MM, Sharp J (2001) Tracing regional flow paths to major springs in Trans-Pecos Texas using geochemical data and geochemical models. Chem Geol 179:53–72
Varol S, Davraz A (2015) Evaluation of the groundwater quality with WQI (Water Quality Index) and multivariate analysis: a case study of the Tefenni plain (Burdur/Turkey). Environ Earth Sci 73:1725–1744
Weijden CHVD, Pacheco FAL (2006) Hydrogeochemistry in the Vouga River basin (central Portugal): pollution and chemical weathering. Appl Geochem 21:580–613
Xing L, Guo H, Zhan Y (2013) Groundwater hydrochemical characteristics and processes along flow paths in the north China plain. J Asian Earth Sci 70–71:250–264
Yang P, Cheng Q, Xie S et al (2017) Hydrogeochemistry and geothermometry of deep thermal water in the carbonate formation in the main urban area of Chongqing, China. J Hydrol 549:50–61
Yang Y, Guo T, Jiao W (2018) Destruction processes of mining on water environment in the mining area combining isotopic and hydrochemical tracer. Environ Pollut 237:356–365
Zang H, Zheng X, Jia Z, Chen J, Qin Z (2015) The impact of hydrogeochemical processes on karst groundwater quality in arid and semiarid area: a case study in the Liulin spring area, north China. Arab J Geosci 8:6507–6519
Zheng X, Zang H, Zhang Y et al (2018) A study of hydrogeochemical processes on karst groundwater using a mass balance model in the Liulin spring area, north China. Water 10:903
Acknowledgements
The work was supported by Institute of Karst Geology, Chinese Academy of Geological Sciences, China. The authors would like to thank Liang Yongping and Tang Chunlei for their help on hydrochemical sampling campaigns, data collections and technical support
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lu, S., Chen, J., Zheng, X. et al. Hydrogeochemical characteristics of karst groundwater in Jinci spring area, north China. Carbonates Evaporites 35, 68 (2020). https://doi.org/10.1007/s13146-020-00602-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s13146-020-00602-8