Abstract
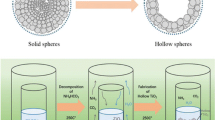
Ternary nitrides are now being considered as one of the emerging advanced functional materials owing to characteristic features and remarkable physicochemical properties that have rationale to substitute precious metals in many applied areas. The present work is focused on Ni3ZnN hollow microspheres for the first time, synthesized by nitridation of the NiO/ZnO precursors in ammonia atmosphere. The precursors composed of 1 μm spheres were made through coordination of terephthalic acid (H2BDC) and Zn2+/Ni2+ cations by solvothermal treatment. The prepared bimetallic nitride is a new single crystalline ternary nitride that belongs to cubic crystal phase (space group Pm-3m, No. 221). Ni3ZnN hollow microspheres were applied as electrocatalyst for proficiently catalyzing the oxygen reduction reactions (ORR), and exhibited very good catalytic activity with the onset potential of 0.81 V versus RHE in alkaline media. After 2000 cycles, Ni3ZnN hollow microspheres show only a 14 mV negative shift in its half-wave potential, suggesting high stability for the ORR. Our current results can show that new ternary nitride is a potential electrocatalytic material for renewable energy resource.

A new ternary metal nitride (Ni3ZnN) has been successfully synthesized through a direct nitridation of the NiO/ZnO hollow microsphere precursors. The prepared bimetallic nitride is a new single crystalline ternary nitride that belongs to cubic crystal phase (space group Pm-3m, No.221). Ni3ZnN were applied as electrocatalyst for proficiently catalyzing the oxygen reduction reaction, and exhibited very good catalytic activity and stability




Similar content being viewed by others
References
L. Han, S. Dong, E. Wang, Adv. Mater. 28, 9266–9291 (2016)
S. Wang, X. Wang, Small 11, 3097–3112 (2015)
X. Ge, A. Sumboja, D. Wuu, T. An, B. Li, F.W.T. Goh, et al., ACS Catal. 5, 4643–4667 (2015)
Y. Nie, L. Li, Z. Wei, Chem. Soc. Rev. 44, 2168–2201 (2015)
Z.-J. Jiang, Z. Jiang, J. Mater. Chem. A 2, 14071–14081 (2014)
J. Wang, H.L. Xin, J. Zhu, S. Liu, Z. Wu, D. Wang, J. Mater. Chem. A 3, 1601–1608 (2015)
W. Zeng, L. Wang, H. Shi, G. Zhang, K. Zhang, H. Zhang, et al., J. Mater. Chem. A 4, 8233–8241 (2016)
H.L. Rongrong Chen, D. Chu, G. Wang, J. Phys. Chem. C 113, 20689–20697 (2009)
H. Yang, J. Liu, J. Wang, C.K. Poh, W. Zhou, J. Lin, et al., Electrochim. Acta 216, 246–252 (2016)
C. Pozo-Gonzalo, O. Kartachova, A.A.J. Torriero, P.C. Howlett, A.M. Glushenkov, D.M. Fabijanic, et al., Electrochim. Acta 103, 151–160 (2013)
D.H. Zhongwei Chen, A. Yu, L. Zhang, J. Zhang, Energy Environ. Sci. 4, 3167–3192 (2011)
B. Cao, J.C. Neuefeind, R.R. Adzic, Inorg. Chem. 54, 2128–2136 (2015)
H. Yan, C. Tian, L. Wang, et al., Angew. Chem. Int. Ed. 54, 6325–6329 (2015)
Y. Wang, R. Ohnishi, E. Yoo, et al., J. Mater. Chem. 22, 15549–15555 (2012)
L. Liu, X. Yang, N. Ma, Small 12, 1295–1301 (2016)
L. An, W. Huang, N. Zhang, J. Mater. Chem. A 2, 62–65 (2014)
L. Zhao, L. Wang, P. Yu, Chem. Commun. 51, 12399–12402 (2015)
B. Cao, J.C. Neuefeind, R.R. Adzic, P.G. Khalifah, Inorg. Chem. 54, 2128–2136 (2015)
G.C. Li, P.F. Liu, R. Liu, M. Liu, K. Tao, S.R. Zhu, et al., Dalton Trans. 45, 13311–13316 (2016)
M. Uehara, A. Uehara, K. Kozawa, T. Yamazaki, Y. Kimishima, C. Physica, Superconductivity 470, S688–S690 (2010)
C. Li, W.G. Chen, F. Wang, S.F. Li, Q. Sun, S. Wang, et al., J. Appl. Phys. 105, 123921–123930 (2009)
J. Fang, Y.F. Yuan, L.K. Wang, H.L. Ni, H.L. Zhu, J.S. Gui, et al., Mater. Lett. 111, 1–4 (2013)
Y. Wang, C. Xie, D. Liu, X. Huang, J. Huo, S. Wang, ACS Appl. Mater. Interfaces 8, 18652–18657 (2016)
P.S. Ying Xiao, M. Cao, ACS Nano 8, 7846–7857 (2014)
Y. Yu, W. Gao, Z. Shen, Q. Zheng, H. Wu, X. Wang, et al., J. Mater. Chem. A 3, 16633–16641 (2015)
M. Mapa, C.S. Gopinath, Chem. Mater. 21, 351–359 (2009)
J. Li, Q. Wang, K. Liu, J. Jiang, D. Qian, J. Li, et al., Mater. Lett. 186, 189–192 (2017)
Acknowledgements
M. Yang would like to thank the National “Thousand Youth Talents” program of China and Ningbo 3315 program. Erum Pervaiz would like to thank the Talented Young Scientist Program (TYSP) by the Ministry of Science and Technology (MoST) China.
Funding
This work is supported by National Natural Science Foundation of China through grant 21471147, the National Key Research and Development Program of China through grant 2016YFB0101205, 2016YFB0101200, and Liaoning Provincial Natural Science Foundation through grant 2014020087.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
He, B., Yuan, Y., Wang, J. et al. Hierarchical Ni3ZnN Hollow Microspheres as Stable Non-Noble Metal Electrocatalysts for Oxygen Reduction Reactions. Electrocatalysis 9, 452–458 (2018). https://doi.org/10.1007/s12678-018-0461-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-018-0461-7