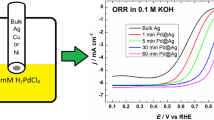
Abstract
Thin Pd coatings were obtained on glassy carbon electrodes by galvanically replacing the electron beam-deposited copper layers with palladium. The prepared electrodes were studied by scanning electron microscopy (SEM) and X-ray photoelectron spectroscopy (XPS). The electrodes were electrochemically tested for oxygen reduction reaction (ORR) employing the rotating disc electrode (RDE) method in alkaline solution. The remaining copper was detected for two of the thickest (5 and 10 nm) sacrificial Cu layers using energy-dispersive X-ray spectroscopy (EDX). SEM images of thin Pd films showed some larger particle growth for all the studied film thicknesses, but spherical particles such as those achieved for example by the electron beam evaporation method were not visible. Specific activity of the electrodes for O2 reduction did not depend on the thickness of the sacrificial copper layer. The O2 reduction reaction followed a four-electron pathway with a mechanism similar to that of commercial Pd/C, where the first electron transfer to the O2 molecule is the rate-determining step.

ᅟ





Similar content being viewed by others
References
E. Antolini, Energy. Environ. Sci. 2, 915 (2009)
M. Shao, J. Power Sources 196, 2433 (2011)
H. Erikson, A. Sarapuu, J. Solla-Gullon, K. Tammeveski, J. Electroanal. Chem. 780, 327 (2016)
M.H. Shao, Q.W. Chang, J.P. Dodelet, R. Chenitz, Chem. Rev. 116, 3594 (2016)
L. Jiang, A. Hsu, D. Chu, R. Chen, J. Electrochem. Soc. 156, B643 (2009)
K. Huang, Z. Liu, C. Lee, Electrochim. Acta 157, 78 (2015)
N. Arjona, M. Guerra-Balcazar, L. Ortiz-Frade, G. Osorio-Monreal, L. Alvarez-Contreras, J. Ledesma-Garcia, L.G. Arriaga, J. Mater. Chem. A 1, 15524 (2013)
C.L. Lee, H.P. Chiou, C.R. Liu, Int. J. Hydrog. Energy 37, 3993 (2012)
H. Erikson, A. Sarapuu, N. Alexeyeva, K. Tammeveski, J. Solla-Gullon, J.M. Feliu, Electrochim. Acta 59, 329 (2012)
O. Savadogo, K. Lee, K. Oishi, S. Mitsushima, N. Kamiya, K.I. Ota, Electrochem. Commun. 6, 105 (2004)
M.H. Seo, S.M. Choi, D.U. Lee, W.B. Kim, Z. Chen, J. Power Sources 300, 1 (2015)
W.P. Xiao, M.A.L. Cordeiro, M.X. Gong, L.L. Han, J. Wang, C. Bian, J. Zhu, H.L. Xin, D.L. Wang, J. Mater. Chem. A 5, 9867 (2017)
M.H. Shao, T. Huang, P. Liu, J. Zhang, K. Sasaki, M.B. Vukmirovic, R.R. Adzic, Langmuir 22, 10409 (2006)
M.H. Shao, K. Sasaki, R.R. Adzic, J. Am. Chem. Soc. 128, 3526 (2006)
M.H. Shao, P. Liu, J.L. Zhang, R. Adzic, J. Phys. Chem. B 111, 6772 (2007)
N.N. Kariuki, X.P. Wang, J.R. Mawdsley, M.S. Ferrandon, S.G. Niyogi, J.T. Vaughey, D.J. Myers, Chem. Mater. 22, 4144 (2010)
G.M. Jiang, X.W. Li, X.S. Lv, L. Chen, Sci. Bull. 61, 1248 (2016)
S. Kondo, M. Nakamura, N. Maki, N. Hoshi, J. Phys. Chem. C 113, 12625 (2009)
M. Shao, T. Yu, J.H. Odell, M. Jin, Y. Xia, Chem. Commun. 47, 6566 (2011)
H. Erikson, A. Sarapuu, K. Tammeveski, J. Solla-Gullon, J.M. Feliu, Electrochem. Commun. 13, 734 (2011)
H. Erikson, M. Lüsi, A. Sarapuu, K. Tammeveski, J. Solla-Gullon, J.M. Feliu, Electrochim. Acta 188, 301 (2016)
M. Lüsi, H. Erikson, A. Sarapuu, K. Tammeveski, J. Solla-Gullon, J.M. Feliu, Electrochem. Commun. 64, 9 (2016)
A. Zadick, L. Dubau, A. Zalineeva, C. Coutanceau, M. Chatenet, Electrochem. Commun. 48, 1 (2014)
T. Bhowmik, M.K. Kundu, S. Barman, Int. J. Hydrog. Energy 41, 14768 (2016)
J.L. Zhang, M.B. Vukmirovic, Y. Xu, M. Mavrikakis, R.R. Adzic, Angew. Chem. Int. Ed. 44, 2132 (2005)
V. Stamenkovic, B.S. Mun, K.J.J. Mayrhofer, P.N. Ross, N.M. Markovic, J. Rossmeisl, J. Greeley, J.K. Norskov, Angew. Chem. Int. Ed. 45, 2897 (2006)
S.M. Alia, K. Jensen, C. Contreras, F. Garzon, B. Pivovar, Y.S. Yan, ACS Catal. 3, 358 (2013)
N. Jung, Y. Sohn, J.H. Park, K.S. Nahm, P. Kim, S.J. Yoo, Appl. Catal. B 196, 199 (2016)
X. Wang, L.J. Zhang, H.Y. Gong, Y.L. Zhu, H.H. Zhao, Y. Fu, Electrochim. Acta 212, 277 (2016)
A. Sarkar, A. Manthiram, J. Phys. Chem. C 114, 4725 (2010)
R.R. Adzic, J. Zhang, K. Sasaki, M.B. Vukmirovic, M. Shao, J.X. Wang, A.U. Nilekar, M. Mavrikakis, J.A. Valerio, F. Uribe, Top. Catal. 46, 249 (2007)
E.J. Coleman, A.C. Co, J. Catal. 316, 191 (2014)
J. Hu, K.A. Kuttiyiel, K. Sasaki, D. Su, T.H. Yang, G.G. Park, C.X. Zhang, G.Y. Chen, R.R. Adzic, Catalysts 5, 1321 (2015)
L.A. Porter, H.C. Choi, A.E. Ribbe, J.M. Buriak, Nano Lett. 2, 1067 (2002)
L.B. Sheridan, J. Czerwiniski, N. Jayaraju, D.K. Gebregziabiher, J.L. Stickney, D.B. Robinson, M.P. Soriaga, Electrocatalysis 3, 96 (2012)
M. Mohl, D. Dobo, A. Kukovecz, Z. Konya, K. Kordas, J.Q. Wei, R. Vajtai, P.M. Ajayan, J. Phys. Chem. C 115, 9403 (2011)
S. Baek, K.H. Kim, M.J. Kim, J.J. Kim, Appl. Catal., B 217, 313 (2017)
L.B. Sheridan, D.K. Gebregziabiher, J.L. Stickney, D.B. Robinson, Langmuir 29, 1592 (2013)
A. Papaderakis, I. Mintsouli, J. Georgieva, S. Sotiropoulos, Catalysts 7, 80 (2017)
S.M. Alia, Y.S. Yan, J. Electrochem. Soc. 162, F849 (2015)
R.Z. Yang, W.Y. Bian, P. Strasser, M.F. Toney, J. Power Sources 222, 169 (2013)
H. Erikson, A. Kasikov, C. Johans, K. Kontturi, K. Tammeveski, A. Sarapuu, J. Electroanal. Chem. 652, 1 (2011)
A. Sarapuu, A. Kasikov, N. Wong, C.A. Lucas, G. Sedghi, R.J. Nichols, K. Tammeveski, Electrochim. Acta 55, 6768 (2010)
L. Arroyo-Ramirez, D. Rodriguez, W. Otano, C.R. Cabrera, ACS Appl. Mater. Interfaces 4, 2018 (2012)
L. Arroyo-Ramírez, Y. Figueroa, D. Rodríguez, W. Otaño, C.R. Cabrera, ECS Trans. 28, 1 (2010)
K. Jukk, N. Alexeyeva, A. Sarapuu, P. Ritslaid, J. Kozlova, V. Sammelselg, K. Tammeveski, Int. J. Hydrog. Energy 38, 3614 (2013)
K. Jukk, N. Alexeyeva, P. Ritslaid, J. Kozlova, V. Sammelselg, K. Tammeveski, Electrocatalysis 4, 42 (2013)
B.G. Pollet, Electrocatalysis 5, 330 (2014)
H. Erikson, A. Sarapuu, J. Kozlova, L. Matisen, V. Sammelselg, K. Tammeveski, Electrocatalysis 6, 77 (2015)
O. Ghodbane, L. Roue, D. Belanger, Chem. Mater. 20, 3495 (2008)
M.S. Jin, H.Y. Liu, H. Zhang, Z.X. Xie, J.Y. Liu, Y.N. Xia, Nano Res. 4, 83 (2011)
A. Sarapuu, A. Kasikov, T. Laaksonen, K. Kontturi, K. Tammeveski, Electrochim. Acta 53, 5873 (2008)
M. Grden, M. Lukaszewski, G. Jerkiewicz, A. Czerwinski, Electrochim. Acta 53, 7583 (2008)
A.J. Bard, L.R. Faulkner, Electrochemical Methods, 2nd edn. (Wiley, New York, 2001)
D.R. Lide, CRC Handbook of Chemistry and Physics (CRC Press, Boca Raton, 2001)
R.E. Davis, G.L. Horvath, C.W. Tobias, Electrochim. Acta 12, 287 (1967)
J.S. Spendelow, A. Wieckowski, Phys. Chem. Chem. Phys. 9, 2654 (2007)
H. Erikson, M. Liik, A. Sarapuu, J. Kozlova, V. Sammelselg, K. Tammeveski, Electrochim. Acta 88, 513 (2013)
G.F. Alvarez, M. Mamlouk, S.M.S. Kumar, K. Scott, J. Appl. Electrochem. 41, 925 (2011)
L.M. Vracar, D.B. Sepa, A. Damjanovic, J. Electrochem. Soc. 136, 1973 (1989)
Acknowledgements
We would like to thank Prof. Sotiris Sotiropoulos from the Aristotle University of Thessaloniki for helpful comments.
Funding
This work was financially supported by institutional research funding (IUT20-16 and IUT2-24) of the Estonian Ministry of Education and Research. This research was also supported by the EU through the European Regional Development Fund (TK141 ‘Advanced materials and high-technology devices for energy recuperation systems’).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lüsi, M., Erikson, H., Merisalu, M. et al. Oxygen Electroreduction in Alkaline Solution on Pd Coatings Prepared by Galvanic Exchange of Copper. Electrocatalysis 9, 400–408 (2018). https://doi.org/10.1007/s12678-017-0445-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-017-0445-z