Abstract
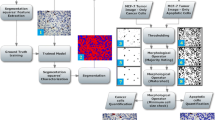
Due to the increased number of applications of both microscopic imaging and image analysis including biomedical studies, the design of specialized algorithms and tools to facilitate quantitative assessment of objects in the image content is of urgent need. Recently, a number of approaches ranging from object counting by machine learning methods to statistical image analysis have been suggested and successfully implemented to resolve the cell quantification problem. Here, we revisit the above problem considering samples where objects presented in the same images have to be explicitly distinguished and quantified without involving any dedicated experimental setting like differential fluorescent staining. We consider several possible classification criteria and show explicitly how their combination in a single algorithm can be used to improve results in complex images where single criteria-based rules inevitably fail. Finally, we suggest a possible approach for the analysis of non-homogeneous images based on combining object selection results for different threshold values thus enhancing the algorithm from multi-criteria to multi-threshold analysis. To demonstrate the performance of the suggested solutions, we show several prominent examples of complex structures ranging from images containing both live and apoptotic cells as well as containing mixtures of globular and fibrous forms of heat-shock protein IbpA.






Similar content being viewed by others
References
Atale, N., Gupta, S., Yadav, U. C. S., & Rani, V. (2014). Cell-death assessment by fluorescent and nonfluorescent cytosolic and nuclear staining techniques. Journal of Microscopy, 255(1), 7–19. https://doi.org/10.1111/jmi.12133.
Netuschil, L., Auschill, T. M., Sculean, A., & Arweiler, N. B. (2014). Confusion over live/dead stainings for the detection of vital microorganisms in oral biofilms - which stain is suitable?. BMC Oral Health: 14. https://doi.org/10.1186/1472-6831-14-2
van der Pol, E., Hoekstra, A. G., Sturk, A., Otto, C., van Leeuwen, T. G., & Nieuwland, R. (2010). Optical and non-optical methods for detection and characterization of microparticles and exosomes. Journal of Thrombosis and Haemostasis, 8(12), 2596–2607. https://doi.org/10.1111/j.1538-7836.2010.04074.x.
Kayumov, A. R., Khakimullina, E. N., Sharafutdinov, I. S., Trizna, E. Y., Latypova, L. Z., Lien, H. T., Margulis, A. B., Bogachev, M. I., & Kurbangalieva, A. R. (2015). Inhibition of biofilm formation in Bacillus subtilis by new halogenated furanones. The Journal of Antibiotics, 68(5), 297–301. https://doi.org/10.1038/ja.2014.143.
Kayumov, A. R., Nureeva, A. A., Trizna, E. Y., Gazizova, G. R., Bogachev, M. I., Shtyrlin, N. V., Pugachev, M. V., Sapozhnikov, S. V., & Shtyrlin, Y. G. (2015). New derivatives of pyridoxine exhibit high antibacterial activity against biofilm-embedded staphylococcus cells. BioMed Research International, 2015. https://doi.org/10.1155/2015/890968.
Trizna, E., Latypova, L., Kurbangalieva, A., Bogachev, M. I., & Kayumov, A. (2016). 2(5H)-Furanone derivatives as inhibitors of staphylococcal biofilms. BioNanoScience, 6(4), 423–426.
Baidamshina, D. R., Trizna, E. Y., Holyavka, M. G., Bogachev, M. I., Artyukhov, V. G., Akhatova, F. S., et al. (2017). Targeting microbial biofilms using Ficin, a nonspecific plant protease. Scientific Reports, 7. https://doi.org/10.1038/srep46068.
Sharafutdinov, I., Pavlova, A., Khabibrakhmanova, A., Kurbangalieva, A., & Kayumov, A. (2017). The antimicrobial effect of the 5-((-)bornyloxy)-2(5H)-furanone derivative on gram-positive bacteria. The FEBS Journal, 284, 143.
Trizna, E. Y., Khakimullina, E. N., Latypova, L. Z., Kurbangalieva, A. R., Sharafutdinov, I. S., Evtyugin, V. G., Babynin, E. V., Bogachev, M. I., & Kayumov, A. R. (2015). Thio derivatives of 2(5H)-Furanone as inhibitors against Bacillus subtilis biofilms. Acta Naturae, 7(2), 102–107.
Volkov, V. (2015). Extraction of extended small-scale objects in digital images. The International Archives of the Photogrammetry, Remote Sensing and Spatial Information Sciences, XL-5/W6, 87–93.
Klinger-Strobel M, Suesse H, Fischer D, Pletz MW, Makarewicz O (2016) A novel computerized cell count algorithm for biofilm analysis. PLoS One: 11(5). https://doi.org/10.1371/journal.pone.0154937
Bogachev, M. I., Volkov, V. Y., Markelov, O. A., Trizna, E. Y., Baydamshina, D. R., Melnikov, V., Murtazina, R. R., Zelenikhin, P. V., Sharafutdinov, I. S., Kayumov, A. R. (2018). Fast and simple tool for the quantification of biofilm-embedded cells sub-populations from fluorescent microscopic images. PLoS One, 13(5). https://doi.org/10.1371/journal.pone.0193267
Heydorn, A., Nielsen, A. T., Hentzer, M., Sternberg, C., Givskov, M., Ersboll, B. K., & Molin, S. (2000). Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology-Sgm, 146, 2395–2407. https://doi.org/10.1099/00221287-146-10-2395.
Beyenal, H., Donovan, C., Lewandowski, Z., & Harkin, G. (2004). Three-dimensional biofilm structure quantification. Journal of Microbiology Methods, 59(3), 395–413. https://doi.org/10.1016/j.mimet.2004.08.003.
Merritt, J. H., Kadouri, D. E., O’Toole, G. A. (2005). Growing and analyzing static biofilms. Current Protocols in Microbiology: Chapter 1:Unit 1B.1. https://doi.org/10.1002/9780471729259.mc01b01s00
Ji, L., Piper, J., & Tang, J. Y. (1989). Erosion and dilation of binary images by arbitrary structuring elements using interval coding. Pattern Recognition Letters, 9(3), 201–209.
Adams, R., & Bischof, L. (1994). Seeded region growing. IEEE Transactions on Pattern Analysis and Machine Intelligence, 16(6), 641–647. https://doi.org/10.1109/34.295913.
Nattkemper, T. W., Twellmann, T., Ritter, H., & Schubert, W. (2003). Human vs. machine: Evaluation of fluorescence micrographs. Computers in Biology and Medicine, 33(1), 31–43.
Tchoukalova, Y. D., Harteneck, D. A., Karwoski, R. A., Tarara, J., & Jensen, M. D. (2003). A quick, reliable, and automated method for fat cell sizing. Journal of Lipid Research, 44(9), 1795–1801. https://doi.org/10.1194/jlr.D300001-JLR200.
Daims, H., Lucker, S., & Wagner, M. (2006). daime, a novel image analysis program for microbial ecology and biofilm research. Environmental Microbiology, 8(2), 200–213. https://doi.org/10.1111/j.1462-2920.2005.00880.x.
Vishnyakov, I. E., Bogachev, M. I., Salafutdinov, I., Borchsenius, S. N., & Kayumov, A. R. (2016). The temperature-dependent selectivity of potential interaction partners for the small heat shock protein IbpA from Acholeplasma laidlawii. BioNanoScience, 6(4), 437–442.
Kayumov, A. R., Bogachev, M. I., Manuvera, V. A., Lazarev, V. N., Sabantsev, A. V., Artamonova TO, Borchsenius, S. N., & Vishnyakov, I. E. (2017). Recombinant small heat shock protein from Acholeplasma laidlawii increases the Escherichia coli viability in thermal stress by selective protein rescue. Molecular Biology, 51(1), 112–121. https://doi.org/10.7868/S0026898417010086.
Funding
The conceptualization of the image analysis methodology and algorithm design has been performed in the framework of the basic state assignment by the Ministry of Science and Higher Education of the Russian Federation to St. Petersburg Electrotechnical University (project No. 2.5475.2017/6.7 to Mikhail I Bogachev). Preparation of experimental biological samples and their microscopic imaging have been supported by the Russian Science Foundation (project No. 17-74-20065 to Innokentii Vishnyakov).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bogachev, M.I., Volkov, V.Y., Kolaev, G. et al. Selection and Quantification of Objects in Microscopic Images: from Multi-Criteria to Multi-Threshold Analysis. BioNanoSci. 9, 59–65 (2019). https://doi.org/10.1007/s12668-018-0588-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12668-018-0588-2