Abstract
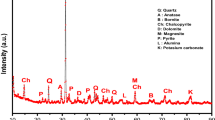
Pre-concentration stages are generally employed to process base and precious metals where the feed grades are inferior. The pre-concentration could remove major gangue minerals and enrich the desired metal content. Further, it improves the efficiency of subsequent concentration stages. In the present investigation, pre-concentration studies were carried out on the mixed copper ore from Malanjkhand, India, containing 0.48% Cu and 74.12% SiO2 contents. Mineralogical characterization revealed that chalcopyrite is the major copper contributing mineral along with a minor amount of malachite. The major gangue minerals present in the ore are quartz, feldspar and mica. Liberation studies of the feed material indicate that around 75% of gangue minerals (mainly quartz) are liberated at 150 µm. Pre-concentration studies were carried out using a Wilfley shaking table, wherein deck angle and wash water rate were varied to achieve the maximum recovery and grade. Optimum results were achieved at a wash water rate of 3.5 lpm and deck angle of 4.9°, wherein the silica rejection and separation efficiency of 52.19% and 53.68% were achieved. The final product assays 2.79% Cu grade and 66.32% copper recovery. The investigation results indicate that the shaking table could be used to pre-concentrate Malanjkhand copper ore.








Similar content being viewed by others
References
British Geological Survey, 2007, Copper-Natural Environment Research Council (2007).
Copper Alliance, 2019. Global 2018 Semis End Use Data Set. https://copperalliance.org/trends-and-innovations/data-set/. downloaded on 14 Jan 2019.
Hertwich E G, Gibon T, Bouman E A, Arvesen A, Suh S, Heath G A, Bergesen J D, Ramirez A, Vega M I, and Shi L, Proc Natl Acad Sci 112 (2015) 6277.
Vidal O, Le Boulzec H and François C, In EPJ Web of Conferences, EDP Sciences, vol. 189 (2018) p 18.
Kleijn R, Van der Voet E, Kramer G J, Van Oers L and Van der Giesen C, Energy 36 (2011) 5640.
Henckens M L C M and Worrell E, J Clean Prod 264 (2020) 121460.
US Geological Survey, 2018. Copper. Mineral Commodity Summaries (2018).
Mudd G M, Resour Policy 35 (2010) 98.
Wills B A, and Finch J A, Wills’ Mineral Processing Technology—An Introduction to the practical Aspects of the ore Treatment and Mineral Recovery, 8th edition, Elsevier Science and Technology Books, Amsterdam (2016), p 5.
Fuerstenau M C, Jameson G J, and Yoon R H, Littleton: Society for Mining, Metallurgy and Exploration, Inc. (2007).
Ackerman P K, Harris G H, Klimpel R R, and Aplan F F, Int J Miner Process 987 (1987) 105.
Senior G D, Guy P J, and Bruckard W J, Int J Miner Process 81 (2006) 15.
Hangone G, Bradshaw D, and Ekmekci Z, J S Afr Inst Min Metall 106 (2005) 199.
Castro S, Goldfarb J, and Laskowski J, Int J Miner Process 1 (1974) 141.
Castro S, Soto H, Goldfarb J, and Laskowski J, Int J Miner Process 1 (1974) 151.
Phetla T P, and Muzenda E, World Academy of Science, Engineering and Technology (2010), p 70.
Wang Y, Wen S, Liu D, Cao Q, Feng Q, and Lv C, Adv Mater Res 807–809 (2013) 2279.
Lee K, Archibald D, McLean J, and Reuter M A, Miner Eng 22 (2009) 395.
Arias N R, Sandoval C A, and Santamaria I, Rev Ion 31 (2018) 89. https://doi.org/10.18273/revion.v31n1-2018014.
Jena S S, Pattanaik A, and Venugopal R, J Mines Met Fuels 67 (2019) 326.
Xiong F, Li Y, Zhang Z, and Lan Y, Adv Mater Res 803 (2013) 131.
Xiong F, Li Y, Zhang Z, Du G, and Lan Y, Adv Mater Res 868 (2014) 417.
Tijsseling L T, Dehaine Q, Rollinson G K, and Galss H J, Miner Eng 138 (2019) 246.
Svoboda J, Guest R N, and Venter W J C, J S Afr Inst Min Metall 88 (1988) 9.
Guest R N, Svoboda J, and Venter W J C, J S Afr Inst Min Metall 88 (1988) 21.
Katwika C N, Kime M B, Kalenga N M, Mbuya B I, and Mwilen T R, Miner Process Extr Metall (2018). https://doi.org/10.1080/08827508.2018.1481057.
Ramakokovhu M M, Kasaini H, and Mbaya R K K, Int J Mater Metall Eng 6 (2012) 8.
Verlinden P, and Cuypers L, VROMANT S.A. (1956), p 89.
Chadwock, J. Int Min, (2008) 8.
Lutandula M S and Maloba B, J Environ Chem Eng 1 (2013) 1085.
Jena S S, Mandre N R, and Venugopal R, Trans Ind Inst Met 72 (2019) 245.
Sivamohan R, and Forssberg E, Int J Miner Process 15 (1985) 281.
Stewart R, J Sediment Res 56 (1986) 555.
McClenaghan M B, Geochem Explor Environ Anal 11 (2011) 265.
Tripathy S K, Singh V, and Ramamurthy R, Int J Min Eng Mineral Process 1 (2012) .
Tripathy S K, Ramamurthy R, and Singh V, J Miner Mater Charact Eng 10 (2011) 13.
Blankson G, Wood A, Quast K, Zanin M, Mensah A, and Skinner W, Powder Technol (2018). https://doi.org/10.1016/j.powtec.2018.12.005.
Junior J A, and Baldo J B, New J Glass Ceram 4 (2014) 29.
Vinhal J T, Coasta R H, Junior A B B, and Espinosa D C R, Energy Technology 2020: Recycling, Carbon Dioxide Management, and Other Technologies, The Minerals, Metals & Materials Series (2020), p 347. https://doi.org/10.1007/978-3-030-36830-2_33.
Mitchell T K, Nguyen A V, and Evans G M, Adv Colloid Interface Sci 114 (2005) 227.
Acknowledgements
The author is greatly thankful to Central Research Facility, IIT (ISM) Dhanbad, Indian Bureau of Mines-Nagpur and Material Research Centre-MNIT, Jaipur, for characterization studies. My sincere acknowledgement to Director, CSIR-IMMT for his permission to do experimental work. I also acknowledge the help of Dr. P. K. Sahu [Department of Applied geology, IIT(ISM) Dhanbad]. The help of Ms. Monisha Mondal and Miss. Aryasuta Nayak is highly appreciated. The author also gratefully acknowledge the reviewer, for his detailed review and comments, which ultimately helped in considerably improvement of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jena, S.S., Angadi, S.I., Mandre, N.R. et al. An Investigation into Pre-concentration of Low-Grade Silica-Rich Malanjkhand Copper Ore by Wilfley Table. Trans Indian Inst Met 74, 571–581 (2021). https://doi.org/10.1007/s12666-020-02160-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-020-02160-y