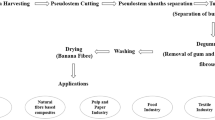
Abstract
Purpose
Hide is a by-product of meat production and is mostly used for leather production. Collagen is the main protein in mammalian skin, connective tissue and cartilage and presents an opportunity for value addition to waste hide off-cuttings by extracting collagen. Three different extraction methods were applied to five different hide sources. The hide sources differed with respect to the animal’s age, sex, diet and environment and influenced collagen extractables yield, and therefore the economic benefit of extraction.
Methods
Acid-solubilisation (AS), acid-enzyme solubilisation (AES1) and modified acid-enzyme solubilisation (AES2) were used to extract collagen from bull, calf, cow, face-pieces and ox-hides.
Results
The highest dry collagen content was from cow hides using the AES2 method (75.13%), followed closely by bull hides at 74.45%. On the other hand, the lowest collagen content was from cow hides (3.80%) with the AS extraction method and the AS method proved to be inefficient for collagen extraction from bull, cow, face-piece and ox-hide sources. Analysis concluded that all the samples were of Type I collagen with α, β, and γ chains.
Conclusions
Waste bovine hide off-cuttings can be used to extract high value product of collagen. AES2 proved to be the most preferable method of extraction out of the three methods applied and considering leather to collagen revenue, these waste bovine hide off-cuttings could potentially result in substantial revenue.
Graphic Abstract



source and extraction method on amount of material extracted: extractables (%) dry product yield. Details This graph was created on Excel 2016


Similar content being viewed by others
References
Lee, C.H., Singla, A., Lee, Y.: Biomedical applications of collagen. Int. J. Pharm. 221(1–2), 1–22 (2001)
Shoulders, M.D., Raines, R.T.: Collagen structure and stability. Annu. Rev. Biochem. 78, 929–958 (2009)
Gelse, K., Pöschl, E., Aigner, T.: Collagens—structure, function, and biosynthesis. Adv. Drug. Deliv. Rev. 55(12), 1531–1546 (2003)
Peng, Y., et al.: Evaluation for collagen products for cosmetic application. Int. J. Cosmet. Sci. 26(6), 313–313 (2004)
Fishman, H.M.: Collagen in cosmetics. Household Pers. Prod. Ind. 46(8) (2009)
Simelane, S., Ustunol, Z.: Mechanical properties of heat-cured whey protein-based edible films compared with collagen casings under sausage manufacturing conditions. J. Food Sci. 70(2), E131–E134 (2005)
Mbg, P.M.C., et al.: An oral supplementation based on hydrolyzed collagen and vitamins improves skin elasticity and dermis echogenicity: a clinical placebo-controlled study. Clin. Pharmacol. Biopharm. 4(142), 2 (2015)
O'Sullivan, A., et al.: Extraction of collagen from fish skins and its use in the manufacture of biopolymer films. J. Aquat. Food Prod. Technol. 15(3), 21–32 (2006)
Meena, C., Mengi, S., Deshpande, S.: Biomedical and industrial applications of collagen. In: Proceedings of the Indian Academy of Sciences-Chemical Sciences. Springer, New York (1999)
Pereira, R.F., Bártolo, P.J.: 3D bioprinting of photocrosslinkable hydrogel constructs. J. Appl. Polym. Sci. 132(48), 17 (2015)
Funk, J.: 3D Bio-Printing; becoming economically feasible. Retrieved July 10, 2016, from https://www.slideshare.net/Funk98/3d-bioprinting-becoming-economically-feasible (2013)
Klein, G.T., Lu, Y., Wang, M.Y.: 3D printing and neurosurgery—ready for prime time? World Neurosurg. 80(3–4), 233–235 (2013)
Inzana, J.A., et al.: 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials 35(13), 4026–4034 (2014)
Mironov, V., Prestwich, G., Forgacs, G.: Bioprinting living structures. J. Mater. Chem. 17(20), 2054–2060 (2007)
Lee, Y.-B., et al.: Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp Neurol 223(2), 645–652 (2010)
Doyle, K.: Bioprinting: From Patches to Parts. (2014)
SouthernLights Biomaterials: Biomaterials for regenerative medicine. Retrieved October 12, 2015, from https://www.slbio.com/ (2015)
Neves, N.M.: Biomaterials from nature for advanced devices and therapies. Wiley, New Jersey (2016)
The Economist: Biomedical technology: tiny forms of scaffolding, combining biological and synthetic elements, have a wide range of medical uses. Retrieved October 28, 2016, from https://www.economist.com/news/technology-quarterly/21573056-biomedical-technology-tiny-forms-scaffolding-combining-biological-and-synthetic (2013)
Poland, D.E., Kaufman, H.E.: Clinical uses of collagen shields. J. Cataract Refract. Surg. 14(5), 489–491 (1988)
Global Industry Analysts, I: Collagen and HA-based biomaterials - a global strategic business report. https://www.strategyr.com/pressMCP-1209.asp (2015). Accessed 10 June 2016
Willoughby, C., Batterbury, M., Kaye, S.: Collagen corneal shields. Surv. Ophthalmol. 47(2), 174–182 (2002)
Geiger, M., Li, R., Friess, W.: Collagen sponges for bone regeneration with rhBMP-2. Adv. Drug Deliv. Rev. 55(12), 1613–1629 (2003)
Lippiello, L., Hall, D., Mankin, H.J.: Collagen synthesis in normal and osteoarthritic human cartilage. J. Clin. Investig. 59(4), 593 (1977)
Cen, L., et al.: Collagen tissue engineering: development of novel biomaterials and applications. Pediatr. Res. 63(5), 492–496 (2008)
Ramshaw, J.A., Werkmeister, J.A., Glattauer, V.: Collagen-based biomaterials. Biotechnol. Genet. Eng. Rev. 13(1), 335–382 (1996)
Parenteau-Bareil, R., Gauvin, R., Berthod, F.: Collagen-based biomaterials for tissue engineering applications. Materials 3(3), 1863–1887 (2010)
Chen, Q., et al.: Collagen-based scaffolds for potential application of heart valve tissue engineering. J. Tissue Sci. Eng. S 11, 2 (2012)
Chen, Q., et al.: Collagen-based scaffolds for potential application of heart valve tissue engineering. J. Tissue Sci. Eng. 11, 3 (2013)
Lee, V., et al.: Design and fabrication of human skin by three-dimensional bioprinting. Tissue Eng. Part C 20(6), 473–484 (2013)
Takaoka, K., et al.: Ectopic bone induction on and in porous hydroxyapatite combined with collagen and bone morphogenetic protein. Clin. Orthop. Relat. Res. 234, 250–254 (1988)
Ahmed, E.M.: Hydrogel: preparation, characterization, and applications: a review. J. Adv. Res. 6(2), 105–121 (2015)
Drury, J.L., Mooney, D.J.: Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 24(24), 4337–4351 (2003)
Lam, C.X.F., et al.: Scaffold development using 3D printing with a starch-based polymer. Mater. Sci. Eng. C 20(1), 49–56 (2002)
Cui, X., et al.: Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Eng. Part A 18(11–12), 1304–1312 (2012)
Global Hides and Skins Market: Review of 2004–2007 and Prospects for 2008. Retrieved July 12, 2015, from https://www.fao.org/fileadmin/templates/est/comm_markets_monitoring/Hides_Skins/Documents/Market_review_hides_and_skins_-_2008.pdf (2008)
Aberoumand, A.: Comparative study between different methods of collagen extraction from fish and its properties. World Appl. Sci. J. 16(3), 316–319 (2012)
Zhang, Y., et al.: Isolation and partial characterization of pepsin-soluble collagen from the skin of grass carp (Ctenopharyngodon idella). Food Chem. 103(3), 906–912 (2007)
Schmidt, M., et al.: Collagen extraction process. Int. Food Res. J. 23(3), 913–922 (2016)
Organization, F.a.A.: Definition and classification of commodities Hides and skins 1994. Retrieved June 2, 2016, from https://www.fao.org/es/faodef/fdef19e.htm#top (2016)
Skierka, E., Sadowska, M.: The influence of different acids and pepsin on the extractability of collagen from the skin of Baltic cod (Gadus morhua). Food Chem. 105(3), 1302–1306 (2007)
Giménez, B., et al.: Use of lactic acid for extraction of fish skin gelatin. Food Hydrocoll. 19(6), 941–950 (2005)
Kittiphattanabawon, P., et al.: Characterisation of acid-soluble collagen from skin and bone of bigeye snapper (Priacanthus tayenus). Food Chem. 89(3), 363–372 (2005)
Wu, J., et al.: Extraction and isolation of type I, III and V collagens and their SDS-PAGE analyses. Trans. Tianjin Univ. 2, 008 (2011)
Carlson, C.: Determination of hydroxyproline content as a measure of fibrosis in nondystrophic and dystrophic skeletal muscle. (2011)
Neuman, R.E., Logan, M.A.: The determination of collagen and elastin in tissues. J. Biol. Chem. 186(2), 549–556 (1950)
Yamauchi, M., Woodley, D.T., Mechanic, G.L.: Aging and cross-linking of skin collagen. Biochem. Biophys. Res. Commun. 152(2), 898–903 (1988)
Abattoirs, A: Age of animals slaughtered. Retrieved February 12, 2017, from https://www.aussieabattoirs.com/facts/age-slaughtered (2017)
New Zealand, T.A.e.o.N.: Beef Farming. Retrieved May 12, 2017, from https://www.teara.govt.nz/en/beef-farming/page-7 (2017)
Peden, R.: Beef farming—beef finishing and dairy beef. Retrieved 12, January, 2018, from https://teara.govt.nz/en/beef-farming/page-7 (2008)
Kelly, D.: The difference between cows, oxen, and cattle. Retrieved August 3, 2015, from https://knowledgenuts.com/2013/11/20/the-difference-betweencows-oxen-and-cattle (2013)
Listrat, A., et al.: How muscle structure and composition influence meat and flesh quality. The Scientific World Journal, London (2016)
Kiew, P.L., Don, M.M.: The influence of acetic acid concentration on the extractability of collagen from the skin of hybrid Clarias sp. and its physicochemical properties: a preliminary study. Focus. Mod. Food Ind. 2(3), 123–128 (2013)
Leach, I.: hides and skins for the tanning industry. Food and Agricultural Organization of the United States. (1995)
Encyclopedia Britannica: Hydroxyproline. https://www.britannica.com/science/hydroxyproline (2018). Accessed 21 Jan 2019
Liu, D., et al.: Optimum condition of extracting collagen from chicken feet and its characetristics. Statistics 2(3), 35 (2001)
Nalinanon, S., et al.: Use of pepsin for collagen extraction from the skin of bigeye snapper (Priacanthus tayenus). Food Chem. 104(2), 593–601 (2007)
Sadowska, M., Kołodziejska, I., Niecikowska, C.: Isolation of collagen from the skins of Baltic cod (Gadus morhua). Food Chem. 81(2), 257–262 (2003)
Kim, S.-K., Venkatesan, J.: Introduction to Marine Glycobiology. Principles and Applications. Marine Glycobiology. Boca Raton: CRC Press (2016)
Heu, M.S., et al.: Characterization of acid-and pepsin-soluble collagens from flatfish skin. Food Sci. Biotechnol. 19(1), 27–33 (2010)
Singh, P., et al.: Isolation and characterisation of collagen extracted from the skin of striped catfish (Pangasianodon hypophthalmus). Food Chem. 124(1), 97–105 (2011)
Kiew, P.L., Mashitah, M.D.: Isolation and characterization of collagen from the skin of Malaysian catfish (Hybrid clarias sp.). J. Korean Soc. Appl. Biol. Chem. 56(4), 441–450 (2013)
Wang, L., et al.: Isolation and characterization of collagen from the skin of deep-sea redfish (Sebastes mentella). J. Food Sci. 72(8), E450–E455 (2007)
Wang, L., Yang, B., Du, X.: Extraction of acid-soluble collagen from grass carp (Ctenopharyngodon idella) skin. J. Food Process Eng. 32(5), 743–751 (2009)
Wang, L., et al.: Optimization of conditions for extraction of acid-soluble collagen from grass carp (Ctenopharyngodon idella) by response surface methodology. Innov. Food Sci. Emerg. Technol. 9(4), 604–607 (2008)
Bowes, J.H., Elliott, R., Moss, J.: The composition of collagen and acid-soluble collagen of bovine skin. Biochem. J. 61(1), 143 (1955)
Guzzi Plepis, A.M.D., Goissis, G., Das-Gupta, D.K.: Dielectric and pyroelectric characterization of anionic and native collagen. Polym. Eng. Sci. 36(24):2932–2938 (1996)
Kittiphattanabawon, P., et al.: Isolation and characterisation of collagen from the skin of brownbanded bamboo shark (Chiloscyllium punctatum). Food Chem. 119(4), 1519–1526 (2010)
Zhang, Z., Li, G., Shi, B.: Physicochemical properties of collagen, gelatin and collagen hydrolysate derived from bovine limed split wastes. J. Soc. Leather Technol. Chem. 90(1), 23 (2006)
Cheng, F.-Y., et al.: Effect of different acids on the extraction of pepsin-solubilised collagen containing melanin from silky fowl feet. Food Chem. 113(2), 563–567 (2009)
Lin, Y.K., Liu, D.C.: Comparison of physical–chemical properties of type I collagen from different species. Food Chem. 99(2), 244–251 (2006)
The European IPPC Bureau: Best available techiques (BAT) reference document for the tanning of hides and skins. 2013: Seville
Solutions, C.: Order collagen for research (2018)
Acknowledgements
We would like to acknowledge Wallace Corporation Ltd (New Zealand) for providing the raw materials.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Noorzai, S., Verbeek, C.J.R., Lay, M.C. et al. Collagen Extraction from Various Waste Bovine Hide Sources. Waste Biomass Valor 11, 5687–5698 (2020). https://doi.org/10.1007/s12649-019-00843-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-019-00843-2