Abstract
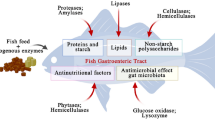
The present study aimed at re-cycling of an aquatic weed (water lettuce, Pistia sp.) as a component in formulation of low cost non-conventional carp diet. Dried Pistia leaves (PL) was bio-processed through solid state fermentation (SSF) for 15 days at 32 ± 1 °C by a tannase producing yeast, Pichia kudriavzevii (GU939629) isolated from the gut of a freshwater carp, Cirrhinus cirrhosus. SSF of PL was effective in reducing the contents of crude fibre and antinutritional factors (e.g., tannin, phytic acid and trypsin inhibitor) significantly (P < 0.05), whereas enhancing crude protein, lipid, ash, total free amino acids and fatty acids. Six isonitrogenous (35%) and isocaloric (17 kJ g−1) experimental diets were prepared incorporating raw (R1–R3) and fermented (F1–F3) PL at 10, 20 and 30% levels by weight replacing fishmeal (FM) and other feed ingredients into a FM-based reference diet and fed to rohu, Labeo rohita fingerlings (initial weight 3.39 ± 0.06 g) for 80 days. In general, fish fed diets with SSF-processed PL resulted in significantly better growth, nutrient digestibility, carcass composition and digestive enzyme activity compared to the diets with raw PL. Fish fed the diet F2 containing 20% fermented PL had the highest live weight gain (106.3%), specific growth rate (SGR, % day−1), protein efficiency ratio and apparent net protein utilization. The highest protein gain (40.84%) and lipid accumulation in the carcass were also recorded in the fish reared on the diet F2. The present study might suggest incorporation of bio-processed PL up to 20% level (12.5% of FM replacement) without interfering growth, feed utilization efficiency and body composition in L. rohita fingerlings.
Similar content being viewed by others
References
Naylor, R.L., Goldburg, R.J., Primavera, J.H., Kautsky, N., Beveridge, M.C.M., Clay, J., Folke, C., Lubchenco, J., Mooney, H., Troell, M.: Effect of aquaculture on world fish supplies. Nature 405, 1017–1024 (2000)
Ray, A.K., Das, I.: Utilization of diets containing composted aquatic weed (Salvinia cuculata) by the Indian major carp, rohu, (Labeo rohita Ham.) fingerlings. Bioresour. Technol. 40, 67–72 (1992)
Ray, A.K., Das, I.: Apparent digestibility of some aquatic macrophytes in rohu, Labeo rohita (Ham.) fingerlings. J. Aquac. Trop. 9, 335–342 (1994)
Ray, A.K., Das, I.: Evaluation of dried aquatic weed, Pistia stratiotes, meal as a feedstuff in pelleted feed for rohu, Labeo rohita, fingerlings. J. Appl. Aquac. 5(4), 35–44 (1996)
Bairagi, A., Ghosh, K.S., Sen, S.K., Ray, A.K.: Duckweed (Lemna polyrhiza) leaf meal as a source of feedstuff in formulated diets for rohu, Labeo rohita (Ham.) fingerlings after fermentation with a fish intestinal bacterium. Bioresour. Technol. 85, 17–24 (2002)
Bairagi, A., Ghosh, K.S., Sen, S.K., Ray, A.K.: Evaluation of nutritive value of Leucaena leucocephala leaf meal inoculated with fish intestinal bacteria Bacillus subtilis and Bacillus circulans in formulated diets for rohu, Labeo rohita (Ham.) fingerlings. Aquac. Res. 35, 436–446 (2004)
Gomes, E.F., Rema, P., Kaushik, S.J.: Replacement of fish meal by plant proteins in the diets of rainbow trout (Oncorhynchus mykiss). Aquaculture 130, 177–186 (1995)
Kalita, P., Mukhopadhyay, P.K., Mukherjee, A.K.: Evaluation of the nutritional quality of four unexplored aquatic weeds from northeast India for the formulation of cost-effective fish feeds. Food Chem. 103, 204–209 (2007)
Khan, A., Ghosh, K.: Evaluation of phytase production by fish gut bacterium, Bacillus subtilis for processing of ipomea aquatica leaves as probable aquafeed ingredient. J. Aquat. Food Prod. Technol. 22(5), 508–519 (2013)
Mandal, S., Ghosh, K.: Optimization of tannase production and improvement of nutritional quality of two potential low-priced plant feedstuffs under solid state fermentation by Pichia kudriavzevii isolated from fish gut. Food Biotechnol. 27, 86–103 (2013)
Saha, S., Ray, A.K.: Evaluation of nutritive value of water hyacinth (Eichhornia crassipes) leaf meal in compound diets for rohu, Labeo rohita (Hamilton, 1822) fingerlings after fermentation with two bacterial strains isolated from fish gut. Turk J. Fish. Aquat. Sci. 11, 199–207 (2011)
Lech, G.P., Reigh, R.C.: Plant products affect growth and digestive efficiency of cultured Florida pompano (Trachinotus carolinus) fed compounded diets. PLoS ONE 7(4), e34981 (2012)
Chowdhary, S., Srivastava, P.P., Jena, J.K., Raizada, S., Yadav, J.K., Dayal, R., Mishra, S.: Growth responses on major replacement of animal protein with plant protein and graded levels of dietary supplement amino sugar, glucosamine in threatened Magur (Clarias batrachus, Linnaeus, 1758) fry. J. Appl. Biol. Biotechnol. 4(4), 42–50 (2016)
So, O., Si, O.: The effect of replacing Fish meal with 10% of groundnut cake in the diets of H. longifilis on its growth, food conversion and survival. J. Appl. Sci. Environ. Manag. 11(3), 87–90 (2007)
Francis, G., Makkar, H.P.S., Becker, K.: Anti-nutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 199, 197–227 (2001)
Khan, I.A., Maqbool, A.: Effects of dietary protein levels on the growth, feed utilization and haemato-biochemical parameters of freshwater fish, Cyprinus carpio Var. specularis. Fish Aquac J. 8(1), 187 (2017). https://doi.org/10.4172/2150-3508.10001
Mandal, S., Ghosh, K.: Development of plant derived low-cost fish feed through overcoming adverse effects of plant anti-nutrients. Fish. Chim. 29(1), 156–161 (2009)
Mandal, S., Ghosh, K.: Accumulation of tannin in different tissues of Indian major carps & exotic carps. Aquac. Res. 41, 945–948 (2010)
Hernes, P.J., Hedges, J.I.: Determination of condensed tannin monomers in plant tissues, soils, and sediments by capillary gas chromatography of acid hydrolysis extracts. Anal. Chem. 72, 5115–5124 (2000)
Olvera, N.M.A., Martinez, P., Galvan, C.R., Chavez, S.C.: The use of seed of the leguminous plant Sesbania grandijlora as a partial replacement for fish meal in the diets for tilapia (Oreochromis mossambicus). Aquaculture 71, 51–60 (1988)
Al-Owafeir, M.: The effects of dietary saponin and tannin on growth performance and digestion in Oreochromis niloticus and Clarias gariepinus. In: Ph. D. Thesis, pp. 220. Institute of Aquaculture, University of Stirling (1999)
Becker, K., Makkar, H.P.S.: Effects of dietary tannic acid and quebracho tannin on growth performance and metabolic rates of common carp (Cyprinus carpio L.). Aquaculture 175, 327–335 (1999)
Hossain, M.A., Jauncey, K.: Nutritional evaluation of some Bangladeshi oilseed meals as partial substitutes for fish meal in the diet of common carp, Cyprinus carpio L.. Aquac. Fish. Manag. 20, 255–268 (1989)
Krogdahl, A.: Alternative protein sources from plants contain anti-nutrients affecting digestion in salmonids. In: Takeda, M., Watanabe, T. (eds.) The Current Status of Fish Nutrition in Aqua-culture. Proceedings of the Third International Symposium on Feeding and Nutrition in Fish, pp. 253–261. Tokyo University of Fisheries, Tokyo (1989)
Maitra, S., Ray, A.K.: Inhibition of digestive enzymes in rohu, Labeo rohita (Hamilton) fingerlings by tannin, an in vitro study. Aquac. Res. 34, 93–95 (2003)
Mandal, S., Ghosh, K.: Inhibitory effect of Pistia tannin on digestive enzymes of Indian major carps, an in vitro study. Fish Physiol. Biochem. 36, 1171–1180 (2010)
Goel, G., Puniya, A.K., Aguilar, C.N., Sing, K.: Interaction of gut microflora with tannins in feeds. Naturwissenschaften 92, 497–503 (2005)
Soetan, K.O., Oyewole, O.E.: The need for adequate processing to reduce the anti-nutritional factors in plants used as human foods and animal feeds: a review. Afr. J. Food Sci. 3(9), 223–232 (2009)
Bhat, T.K., Singh, B., Sharma, O.P.: Microbial degradation of tannins—a current perspective. Biodegradation 9, 343–357 (1998)
Nelson, K.E., Pell, A.N., Schofield, P., Zinder, S.H.: Isolation and characterization of anaerobic ruminal bacterium capable of degrading hydrolyzable tannins. Appl. Environ. Microbiol. 61, 3293–3298 (1995)
Mandal, S., Ghosh, K.: Isolation of tannase-producing microbiota from the gastrointestinal tracts of some freshwater fish. J. Appl. Ichthyol. 29, 145–153 (2013). https://doi.org/10.1111/j.1439-0426.2012.02054.x
Talukdar, S., Ringo, E., Ghosh, K.: Extracellular tannase-producing bacteria detected in the digestive tracts of freshwater fishes (Actinopterygii: Cyprinidae and Cichlidae). Acta Ichtyol. Pisc. 46(3), 201–210 (2016)
Ghosh, K., Ray, A.K.: Tannins in plant feed ingredients: facts and probable consequences in fish nutrition. In: Petridis, G.K. (ed.) Tannins: Types, Foods Containing, and Nutrition, pp. 265–280. Nova Publishers, Hauppauge (2011)
Ghosh, K., Mandal, S.: Nutritional evaluation of groundnut oil cake in formulated diets for rohu, Labeo rohita (Hamilton) fingerlings after solid state fermentation with a tannase producing yeast, Pichia kudriavzevii (GU939629) isolated from fish gut. Aquac. Rep. 2, 82–90 (2015). https://doi.org/10.1016/j.aqrep.2015.08.006
Wee, K.L.: Use of non-conventional feedstuff of plant origin as fish feeds—is it practical and economically feasible? In: De Silva, S.S. (ed.) Fish Nutrition Research in Asia. Proceedings of the Fourth Asian Fish Nutrition Workshop. Asian Fisheries Society Special Publication 5, Asian Fisheries Society, Manila (1991)
Mondal, S., Roy, T., Ray, A.K.: Characterization and identification of enzyme producing bacteria isolated from the digestive tract of bata, Labeo bata. J. World Aquac. Soc. 41, 369–376 (2010)
Jhingran, V.G.: Fish and Fisheries of India, 3rd edn., pp. 335–337. Hindustan Publishing Corporation, Delhi (1997)
Mondal, K., Banerjee, D., Banerjee, R., Pati, B.: Production and characterization of tannase from Bacillus cereus KBR9. J. Gen. Appl. Microbiol. 47, 263–267 (2001)
Saha, S., Ghosh, K.: Evaluation of nutritive value of raw and fermented de-oiled physic nut, Jatropha curcas seed meal in the formulated diets for rohu, Labeo rohita (Hamilton) fingerlings. Proc. Zool. Soc. 66(1), 41–50 (2013)
Roy, T., Banerjee, G., Dan, S.K., Ghosh, P., Ray, A.K.: Improvement of nutritive value of sesame oilseed meal in formulated diets for rohu, Labeo rohita (Hamilton), fingerlings after fermentation with two phytase-producing bacterial strains isolated from fish gut. Aquac. Int. 22, 633–652 (2014)
Spyridakis, P., Metailler, R., Gabaudan, J., Riaza, A.: Studies on nutrient digestibility in European sea bass (Dicentrarchus labrax) 1. Methodological aspects concerning faeces collection. Aquaculture 77, 61–70 (1989)
AOAC (Association of Official Analytical Chemists): Official Methods of Analysis of the Official Association of Analytical Chemists, 18th edn. Association of the Official Analytical Chemists, Arlington (2005)
Maynard, L., Loosil, J., Hintz, H., Warner, R.: Animal Nutrition, 7th edn., pp. 13–14. McGraw-Hill, New York (1979)
Moore, S., Stein, W.W.: Photometric ninhydrin method for use in the chromatography of amino acids. J. Biol. Chem. 176, 367–388 (1948)
Cox, H.E., Pearson, D.: The Chemical Analysis of Foods. Chemical Publishing Co. Inc, New York (1962)
Schanderi, S.H.: Methods in Food Analysis, p. 709. Academic Press, New York (1970)
Vaintraub, I.A., Lapteva, N.A.: Colorimetric determination of phytate in unpurified extracts of seeds and the products of their processing. Anal. Biochem. 175, 227–230 (1988)
Smith, C., Van Megen, W., Twaalhoven, L., Hitchcock, C.: The determination of trypsin inhibitor levels in foodstuffs. J. Sci. Food. Agric. 3, 341–350 (1980)
Bolin, D.W., King, R.P., Klosterman, E.W.: A simplified method for the determination of chromic oxide (Cr2O3) when used as a index substance. Science 116, 634–635 (1952)
Steffens, W.: Principles of Fish Nutrition. pp.384. Ellis Horwood Ltd., Chichester (1989)
Cho, C.Y., Slinger, S.J., Bayley, H.S.: Bioenergetics of salmonid fishes: energy intake, expenditure and productivity. Comp. Biochem. Physiol. 73, 25–41 (1982)
APHA (American Public Health Association, American Water Works Association, Water Environment Federation): Standard Methods for the Examination of Water and Wastewater, 19th edn. American Public Health Association, New York (1995)
Lowry, O.H., Ronebrough, N.J., Farr, A.L., Randell, R.J.: Protein measurement with Folin phenol reagent. J. Biol. Chem. 193, 265–276 (1951)
Bernfeld, P.: Amylase (alpha) and (beta). In: Colowick, S.P., Kaplan, N.O. (eds.) Methods in Enzymology, vol. 1, pp. 149–150. Academic Press, New York (1955)
Bier, M.: Lipases. In: Colowick, S.P., Kaplan, N.O. (eds.) Methods in Enzymology, vol. 1. Academic Press, New York (1955)
Beveridge, M.C.M., Sikdar, P.K., Frerichs, G.N., Millar, S.: The ingestion of bacteria in suspension by the common carp Cyprinus carpio L. J. Fish Biol. 39, 825–831 (1991)
Rahmatullah, S.M., Beveridge, M.C.M.: Ingestion of bacteria in suspension Indian major carps (Catla catla, Labeo rohita) and Chinese carps (Hypophthalmichthys molitrix, Aristichthys nobilis). Hydrobiologica. 264, 79–84 (1993)
Zar, J.H.: Biostatistical Analysis, 4th edn. Pearson Education Singapore Pte. Ltd (Indian Branch), New Delhi (1999)
Kinnear, P.R., Gray, C.D.: In: SPSS for Windows Made Simple. Release 10. Psychology Press, Sussex (2000)
Ayoade, G.O., Sharma, B.M., Spridhar, M.K.C.: Trials of Pistia stratiotes L. as animal feed. J. Aquat. Plant. Manag. 20, 56–57 (1982)
Makkar, H.P.S., Becker, K.: Isolation of tannins from leaves of some trees and shrubs and their properties. J. Agric. Food Chem. 42, 73–734 (1994)
Mukhopadhyay, N., Ray, A.K.: Improvement of quality of sesame Sesamum indicum seed meal protein with supplemental amino acids in feeds for rohu Labeo rohita (Hamilton) fingerlings. Aquac. Res. 30, 549–557 (1999)
Mukhopadhyay, N., Ray, A.K.: Utilization of copra meal in the formulation of compound diets for rohu, Labeo rohita fingerlings. J. Appl. Ichthyol. 15, 127–131 (1999)
Pandey, A., Soccol, C.R., Rodriguez-Leon, J.A., Nigam, P.: History and Development of Solid State Fermentation. In: Pandey, A. (ed.) Biotechnology: Fundamentals and Applications, Asiatech Publishers, New Delhi (2001)
Tengerdy, R.P.: Solid substrate fermentation for enzyme production. In: Pandey, A. (ed.) Advances in Biotechnology, pp. 13–16. Educational Publishers and Distributors, New Delhi (1998)
El-Sayed, A.F.M., Mansour, C.R., Ezzat, A.A.: Effects of dietary protein levels on spawning performance of Nile tilapia (Oreochromis niloticus) brood stock reared at different water salinities. Aquaculture 220, 619–632 (2003)
Allan, G.L., Parkinson, S., Booth, M.A., Stone, D.A.J., Rowland, S.J., Frances, J., Warner-Smith, R.: Replacement of fishmeal in diets for Australian silver perch, Bidyanus bidyanus: digestibility of alternative ingredients. Aquaculture 186, 293–310 (2000)
Lee, S.M.: Apparent digestibility coefficients of various feed ingredients for juvenile and grower rockfish (Sebastes schlegeli). Aquaculture 207, 79–95 (2002)
Mukhopadhyay, N., Ray, A.K.: Effect of fermentation on the nutritive value of sesame seed meal in the diets for rohu, Labeo rohita (Hamilton), fingerlings. Aquac. Nutr. 5, 229–236 (1999)
Ramachandran, S., Bairagi, A., Ray, A.K.: Improvement of nutritive value of grass pea (Lathyrus sativus) seed meal in the formulated diets for rohu, Labeo rohita (Ham.) fingerlings after fermentation with a fish gut bacterium. Bioresour. Technol. 96, 1465–1472 (2005)
Krogdahl, A., Lea, T.B., Olli, J.L.: Soybean proteinase inhibitors affect intestinal trypsin activities and amino acid digestibilities in rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. 107, 215–219 (1994)
Santigosa, E., Sa´nchez, J., Me´dale, F., Kaushik, S., Pe´rez-Sa´nchez, J., Gallardo, M.A.: Modifications of digestive enzymes in trout (Oncorhynchus mykiss) and sea bream (Sparus aurata) in response to dietary fish meal replacement by plant protein sources. Aquaculture 282, 68–74 (2008)
Kumar, V., Makkar, H.P.S., Becker, K.: Detoxified Jatropha curcas kernel meal as a dietary protein source: growth performance, nutrient utilization and digestive enzymes in common carp (Cyprinus carpio L.) fingerlings. Aquac. Nutr. 17(3), 313–326 (2011)
Scalbert, A.: Antimicrobial properties of tannins. Phytochemistry. 30, 3875–3883 (1991)
McSweeney, C.S., Palmer, B., Bunch, R., Krause, D.O.: Microbial interactions with tannins: nutritional consequences for ruminants. Anim. Feed Sci. Technol. 91, 83–93 (2001)
Smith, A.H., Zoetendal, E., Mackie, R.I.: Bacterial mechanism to overcome inhibitory effects of dietary tannins. Microb. Ecol. 50, 197–205 (2005)
Phuong, N.T., Thanonkeo, P., Phong, H.X.: Screening useful isolated yeasts for ethanol fermentation at high temperature. Int. J. Appl. Sci. Technol. 2(4), 65–71 (2012)
Acknowledgements
The authors are grateful to the Head, Department of Zoology (DST-FIST and UGC-SAP-DRS sponsored), The University of Burdwan, West Bengal, India; The Department of Science and Technology (PURSE programme) and Science and Engineering Research Board (SERB Research Project No. SR/FT/LS-193/2009), New Delhi, India for providing research facilities. The first author is grateful to The University of Burdwan for awarding the university fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mandal, S., Ghosh, K. Utilization of Fermented Pistia Leaves in the Diet of Rohu, Labeo rohita (Hamilton): Effects on Growth, Digestibility and Whole Body Composition. Waste Biomass Valor 10, 3331–3342 (2019). https://doi.org/10.1007/s12649-018-0336-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-018-0336-4