Abstract
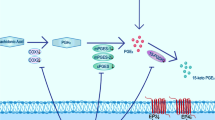
Mature brain-derived neurotrophic factor (mBDNF) has neuroprotection in cerebral ischemia. Conversely, the precursor of brain-derived neurotrophic factor (proBDNF) has the opposite function to its mature form, inducing apoptosis. However, whether the neuroprotection of Panax notoginsenoside Rb1 (PNS-Rb1) on ischemic stroke is due to, at least partially, its modulation of suppressing proBDNF/P75NTR/sortilin or upregulation of mBDNF is not clear. To test this hypothesis, rats induced by photothrombotic stroke were treated with PNS-Rb1 100 mg/kg or nimodipine 1 mg/kg twice a day until 3, 7, and 14 days. Our data indicate that PNS-Rb1 significantly reduced cerebral infarction rate, proBDNF/P75NTR/sortilin, and plasminogen activator inhibitor-1 (PAI-1) protein levels, and improved sensorimotor dysfunctions induced by ischemic stroke, upregulation of BDNF/TrkB levels, and its processing enzymes (tissue plasminogen activator, tPA) in a time-dependent manner. Taken together, our findings indicate that the improvement of sensorimotor dysfunctions by PNS-Rb1 following ischemic stroke is made, at least partially, by activating the BDNF/TrkB and inhibiting proBDNF/sortilin/P75NTR.







Similar content being viewed by others
References
Barker PA (2009) Whither proBDNF? Nat Neurosci 12(2):105–106. https://doi.org/10.1038/nn0209-105
Bouet V, Boulouard M, Toutain J, Divoux D, Bernaudin M, Schumann-Bard P, Freret T (2009) The adhesive removal test: a sensitive method to assess sensorimotor deficits in mice. Nat Protoc 4:1560–1564. https://doi.org/10.1038/nprot.2009.125
Cao W, Duan J, Wang X, Zhong X, Hu Z, Huang F et al (2014) Early enriched environment induces an increased conversion of proBDNF to BDNF in the adult rat's hippocampus. Behav Brain Res. 265:76-83. https://doi.org/10.1016/j.bbr.2014.02.022
Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M, Chopp M (2001) Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke 32:1005–1011
Choi Y, Kang SG, Kam KY (2015) Changes in the BDNF-immunopositive cell population of neocortical layers I and II/III after focal cerebral ischemia in rats. Brain Res 1605:76–82. https://doi.org/10.1016/j.brainres.2015.02.007
Coulson EJ, Reid K, Baca M, Shipham KA, Hulett SM, Kilpatrick TJ, Bartlett PF (2000) A new death domain of the p75 neurotrophin receptor that mediates rapid neuronal cell death. J Biol Chem 275:30537–30545
Coulson EJ, Reid K, Shipham KM, Morley S, Kilpatrick TJ et al (2004) The role of neurotransmission and the chopper domain in p75 neurotrophin receptor death signaling. Prog Brain Res 146:41–62
Donnan GA, Fisher M, Macleod M, Davis SM (2008) Stroke. Lancet (London, England) 371:1612–1623. https://doi.org/10.1016/S0140-6736(08)60694-7
Fan YJ, Wu LL, Li HY, Wang YJ, Zhou XF (2008) Differential effects of pro-BDNF on sensory neurons after sciatic nerve transection in neonatal rats. Eur J Neurosci 27:2380–2390. https://doi.org/10.1111/j.1460-9568.2008.06215.x
Hashimoto K (2013) Sigma-1 receptor chaperone and brain-derived neurotrophic factor: emerging links between cardiovascular disease and depression. Prog Neurobiol 100:15–29. https://doi.org/10.1016/j.pneurobio.2012.09.001
Hashimoto K (2016) Regulation of brain-derived neurotrophic factor (BDNF) and its precursor proBDNF in the brain by serotonin. Eur Arch Psychiatry Clin Neurosci 266:195–197. https://doi.org/10.1007/s00406-016-0682-9
Hempstead BL (2014) Deciphering proneurotrophin actions. Handb Exp Pharmacol 220:17–32. https://doi.org/10.1007/978-3-642-45106-5_2
Hermann DM, Chopp M (2012) Promoting brain remodelling and plasticity for stroke recovery: therapeutic promise and potential pitfalls of clinical translation. Lancet Neurol 11:369–380. https://doi.org/10.1016/S1474-4422(12)70039-X
Hou J, Xue J, Lee M, Yu J, Sung C (2014) Long-term administration of ginsenoside Rh1 enhances learning and memory by promoting cell survival in the mouse hippocampus. Int J Mol Med 33:234–240. https://doi.org/10.3892/ijmm.2013.1552
Huang XP, Ding H, Lu JD, Tang YH, Deng BX, Deng CQ (2015) Effects of the combination of the main active components of Astragalus and Panax notoginseng on inflammation and apoptosis of nerve cell after cerebral ischemia-reperfusion. Am J Chin Med 43:1419–1438. https://doi.org/10.1142/S0192415X15500809
Je HS, Yang F, Ji Y, Potluri S, Fu XQ, Luo ZG, Nagappan G, Chan JP, Hempstead B, Son YJ, Lu B (2013) ProBDNF and mature BDNF as punishment and reward signals for synapse elimination at mouse neuromuscular junctions. J Neurosci 33(24):9957–9962. https://doi.org/10.1523/JNEUROSCI.0163-13.2013
Lawrence DA, Strandberg L, Ericson J, Ny T (1990) Structure-function studies of the SERPIN plasminogen activator inhibitor type 1. Analysis of chimeric strained loop mutants. J Biol Chem 265:20293–20301
Lee CH, Kim JH (2014) A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J Ginseng Res 38:161–166. https://doi.org/10.1016/j.jgr.2014.03.001
Li JY, Liu J, Manaph NPA, Bobrovskaya L, Zhou XF (2017) ProBDNF inhibits proliferation, migration and differentiation of mouse neural stem cells. Brain Res. 1668:46–55. https://doi.org/10.1016/j.brainres.2017.05.013
Liepinsh E, Ilag LL, Otting G, Ibáñez CF (1997) NMR structure of the death domain of the p75 neurotrophin receptor. EMBO J 16(16):4999–5005
Liu XY, Zhou XY, Hou JC, Zhu H, Wang Z, Liu JX, Zheng YQ (2015) Ginsenoside Rd promotes neurogenesis in rat brain after transient focal cerebral ischemia via activation of PI3K/Akt pathway. Acta Pharmacol Sin 36:421–428. https://doi.org/10.1038/aps.2014.156
Masumu M, Hata R (2003) Recent advances in adenovirus-mediated gene therapy for cerebral ischemia. Curr Gene Ther 3:43–48
Nykjaer A, Lee R, Teng KK, Jansen P, Madsen P, Nielsen MS, Jacobsen C, Kliemannel M, Schwarz E, Willnow TE, Hempstead BL, Petersen CM (2004) Sortilin is essential for proNGF-induced neuronal cell death. Nature 427:843–848
Ong WY, Farooqui T, Koh HL, Farooqui AA, Ling EA (2015) Protective effects of ginseng on neurological disorders. Front Aging Neurosci 7:129. https://doi.org/10.3389/fnagi.2015.00129
Pang PT, Teng HK, Zaitsev E, Woo NT, Sakata K et al (2004) Cleavage of proBDNF by tPA/plasmin is essential for long-term hippocampal plasticity. Science 306(5695):487–491. https://doi.org/10.1126/science.1100135
Rahman M, Luo H, Sims NR, Bobrovskaya L, Zhou XF (2018) Investigation of mature BDNF and proBDNF signaling in a rat photothrombotic ischemic model. Neurochem Res 43(3):637–649. https://doi.org/10.1007/s11064-017-2464-9
Roza G s, Guerra MJ, Labandeira-Garcia JL (1997) An automated rotarod method for quantitative drug-free evaluation of overall motor deficits in rat models of parkinsonism. Brain Res Protocol 2:75–84
Seidah NG, Chretien M (1999) Proprotein and prohormone convertases: a family of subtilases generating diverse bioactive polypeptides. Brain Res 848(1–2):45–62
Sun Y, Lim Y, Li F, Liu S, Lu JJ, Haberberger R, Zhong JH, Zhou XF (2012) ProBDNF collapses neurite outgrowth of primary neurons by activating RhoA. PLoS One 7(4):e35883. https://doi.org/10.1371/journal.pone.0035883
Tachikawa KK, Harada K, Kashimoto T, Miyate Y et al (1999) Effects of ginseng saponins on responses induced by various receptor stimuli. Eur J Pharmacol 369:23–32
Tao J, Chen B, Gao Y, Yang S, Huang J, Jiang X, Wu Y, Peng J, Hong Z, Chen L (2014) Electroacupuncture enhances hippocampal NSCs proliferation in cerebral ischemia-reperfusion injured rats via activation of notch signaling pathway. Int J Neurosci 124:204–212. https://doi.org/10.3109/00207454.2013.840781
Teng HK, Teng KK, Lee R, Wright S, Tevar S, Almeida RD, Kermani P, Torkin R, Chen ZY, Lee FS, Kraemer RT, Nykjaer A, Hempstead BL (2005) ProBDNF induces neuronal apoptosis via activation of a receptor complex of p75NTR and sortilin. J Neurosci 25:5455–5463
Wong I, Liao H, Bai X, Zaknic A, Zhong J, Guan Y, Li HY, Wang YJ, Zhou XF (2010) ProBDNF inhibits infiltration of ED1+ macrophages after spinal cord injury. Brain Behav Immun 24:585–597. https://doi.org/10.1016/j.bbi.2010.01.001
Xu ZQ, Sun Y, Li HY, Lim Y, Zhong JH, Zhou XF (2011) Endogenous proBDNF is a negative regulator of migration of cerebellar granule cells in neonatal mice Eur J Neurosci 33:1376–1384 https://doi.org/10.1111/j.1460-9568.2011.07635.x
Yan YT, Li SD, Li C, Xiong YX, Lu XH, Zhou XF, Yang LQ, Pu LJ, Luo HY (2018) Panax notoginsenoside saponins Rb1 regulates the expressions of Akt/ mTOR/PTEN signals in the hippocampus after focal cerebral ischemia in rats. Behav Brain Res 345:83–92. https://doi.org/10.1016/j.bbr.2018.02.037
Yang J, Harte-Hargrove LC, Siao CJ, Marinic T, Clarke R, Ma Q, Jing D, LaFrancois JJ, Bath KG, Mark W, Ballon D, Lee FS, Scharfman HE, Hempstead BL (2014) proBDNF negatively regulates neuronal remodeling, synaptic transmission, and synaptic plasticity in hippocampus. Cell Rep 7:796–806. https://doi.org/10.1016/j.celrep.2014.03.040
Ye R, Han J, Kong X, Zhao L, Cao R, Rao Z, Zhao G (2008) Protective effects of ginsenoside Rd on PC12 cells against hydrogen peroxide. Biol Pharm Bull 31:1923–1927
Ye R, Li N, Han J, Kong X, Cao R, Rao Z, Zhao G (2009) Neuroprotective effects of ginsenoside Rd against oxygen-glucose deprivation in cultured hippocampal neurons. Neurosci Res 64:306–310. https://doi.org/10.1016/j.neures.2009.03.016
Author Contribution Statements
Study design: HY Luo and J Sun. Study conduct: CY Yang and JY Yang. Data collection: YX Xiong. Data analysis: H Su, S Hu, and XF Zhuang. Data interpretation: JQ Ma. Drafting manuscript: CY Yang and HY Luo. Revising manuscript content: XF Zhou. All authors have read and approved the final submitted manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (81760064), Yunnan Applied Basic Research Project(2019FA040), Joint Special Fund Project of Yunnan Provincial Science and Technology Department-Kunming Medical University (2017FE467-025, 160, 163, 2017FE468-135, U012017515, and 2018FE001(-162)), Scientific Research Fund of Yunnan Provincial Education Department (2015Y180), and Innovative Experimental Program for Undergraduates in Kunming Medical University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All experimental procedures involving animals were approved by the Animal Ethics Committee of Kunming Medical University and conducted between 7 a.m. and 7 p.m. in accordance with the guidelines of the National Health and Medical Research Council of China. Rats were acclimatized for a week before any procedures were initiated.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, CY., Yang, JY., Xiong, YX. et al. Panax notoginsenoside Rb1 Restores the Neurotrophic Imbalance Following Photothrombotic Stroke in Rats. Neurotox Res 36, 441–451 (2019). https://doi.org/10.1007/s12640-019-00058-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00058-2