Abstract
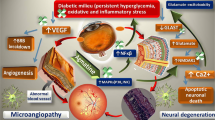
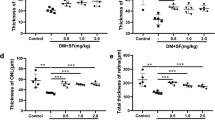
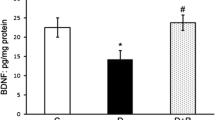
Neurodegeneration in diabetic retina has been widely considered as initiating factor that may lead to vascular damage, the classical hallmark of diabetic retinopathy. Diabetes induced altered glutamate metabolism in the retina, especially through glutamate excitotoxicity might play a major role in the neurodegeneration. Increased level of branched chain amino acids (BCAAs) measured in diabetic retina might cause an increase in the neurotoxic level of glutamate by transamination of citric acid cycle intermediates. In order to analyze the transamination of BCAAs and their influence on neurodegenerative factors, we treated streptozotocin-induced diabetic rats with gabapentin, a leucine analogue and an inhibitor of branched chain amino transferase (BCATc). Interestingly, gabapentin lowered the retinal level of BCAAs in diabetic rats. Furthermore, gabapentin treatments ameliorated the reduced antioxidant glutathione level and increased malondialdehyde (MDA), the marker of lipid peroxidation in diabetic rat retinas. In addition, gabapentin also reduced the expression of proapoptotic caspase-3, a marker of apoptosis and increased anti-apoptotic marker Bcl-2 in diabetic retinas. Thus, these results suggest that gabapentin stimulates glutamate disposal, and ameliorates apoptosis and oxidative stress in diabetic rat retina. The influence of gabapentin may be due to its capacity to increase the ratio of BCKA to BCAA which in turn would reduce glutamate excitotoxicity in diabetic retina.





Similar content being viewed by others
References
Al-Dosari DI, Ahmed MM, Al-Rejaie SS, Alhomida AS, Ola MS (2017) Flavonoid naringenin attenuates oxidative stress, apoptosis and improves neurotrophic effects in the diabetic rat retina. Nutrients 9:E1161. https://doi.org/10.3390/nu9101161
Ambati J, Chalam KV, Chawla DK, D'Angio CT, Guillet EG, Rose SJ, Vanderlinde RE, Ambati BK (1997) Elevated gamma-aminobutyric acid, glutamate, and vascular endothelial growth factor levels in the vitreous of patients with proliferative diabetic retinopathy. Arch Ophthalmol 115:1161–1166
Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, Kester M, Kimball SR, Krady JK, KF LN, Norbury CC, Quinn PG, Sandirasegarane L, Simpson IA, JDRF Diabetic Retinopathy Center Group (2006) Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes 55:2401–2411
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW (1998) Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest 102:783–791
Baydas G, Sonkaya E, Tuzcu M, Yasar A, Donder E (2005) Novel role for gabapentin in neuroprotection of central nervous system in streptozotocine-induced diabetic rats. Acta Pharmacol Sin 26:417–422
Bearse MA Jr, Han Y, Schneck ME, Barez S, Jacobsen C, Adams AJ (2004) Local Multifocal Oscillatory Potential Abnormalities in Diabetes and Early Diabetic Retinopathy. Invest Ophthalmol Vis Sci 45:3259–3265
Bengtsson B, Heijl A, Agardh E (2005) Visual fields correlate better than visual acuity to severity of diabetic retinopathy. Diabetologia 48:2494–2500
Bresnick GH, Palta M (1987) Predicting progression to severe proliferative diabetic retinopathy. Arch Ophthalmol 105:810–814
Dong Z, Saikumar P, Weinberg JM, Venkatachalam MA (2006) Calcium in cell injury and death. Annu Rev Pathol 1:405–434
Feng Y, Wang Y, Stock O, Pfister F, Tanimoto N, Seeliger MW, Hillebrands JL, Hoffmann S, Wolburg H, Gretz N, Hammes HP (2009) Vasoregression linked to neuronal damage in the rat with defect of polycystin-2. PLoS One 4:e7328. https://doi.org/10.1371/journal.pone.0007328
Frayser R, Buse MG (1978) Branched chain amino acid metabolism in the retina of diabetic rats. Diabetologia 14:171–176
Gardner TW, Davila JR (2017) The neurovascular unit and the pathophysiologic basis of diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 255:1–6
Goto M, Miyahara I, Hirotsu K, Conway M, Yennawar N, Islam MM, Hutson SM (2005) Structural determinants for branched-chain aminotransferase isozyme-specific inhibition by the anticonvulsant drug gabapentin. J Biol Chem 280:37246–37256
Gowda K, Zinnanti WJ, LaNoue KF (2011) The influence of diabetes on glutamate metabolism in retinas. J Neurochem 117:309–320
Hariharan M, Naga S, VanNoord T (1993) Systematic approach to the development of plasma amino acid analysis by high-performance liquid chromatography with ultraviolet detection with precolumn derivatization using phenyl isothiocyanate. J Chromatogr 621:15–22
Herranz JL, Sol JM, Hernandez G (2000) Spanish gabapentin work group. Gabapentin used in 559 patients with partial seizures: a multicenter observation study. Rev Neurol 30:1141–1145
Hutson SM, Berkich D, Drown P, Xu B, Aschner M, LaNoue KF (1998) Role of branched-chain aminotransferase isoenzymes and gabapentin in neurotransmitter metabolism. J Neurochem 71:863–874
Hutson SM, Lieth E, LaNoue KF (2001) Function of leucine in excitatory neurotransmitter metabolism in the central nervous system. J Nutr 131:846S–850S
Ishikawa S, Nakazawa M, Ishikawa A, Ishiguro S, Tamai M (1995) Alteration of glutamine concentration in the vitreous humor in patients with proliferative vitreoretinopathy. Curr Eye Res 14:191–197
Jakubowski W, Bartosz G (2000) 2,7-dichlorofluorescin oxidation and reactive oxygen species: what does it measure? Cell Biol Int 24:757–760
Kim YS, Chang HK, Lee JW, Sung YH, Kim SE, Shin MS, Yi JW, Park JH, Kim H, Kim CJ (2009) Protective effect of gabapentin on N-methyl-D-aspartate-induced excitotoxicity in rat hippocampal CA1 neurons. J Pharmacol Sci 109:144–147
Kim JY, Kim DG, Kim SH, Kwon OW, Kim SH, You YS (2016) Macular edema after gabapentin. Korean J Ophthalmol 2:153–155
Kusari J, Zhou S, Padillo E, Clarke KG, Gil DW (2007) Effect of memantine on neuroretinal function and retinal vascular changes of streptozotocin-induced diabetic rats. Invest Ophthalmol Vis Sci 48:5152–5159
Li Q, Puro DG (2002) Diabetes-induced dysfunction of the glutamate transporter in retinal Müller cells. Invest Ophthalmol Vis Sci 43:3109–3116
Lieth E, LaNoue KF, Antonetti DA, Ratz M (2000) Diabetes reduces glutamate oxidation and glutamine synthesis in the retina. The Penn State Retina Research Group. Exp Eye Res 70:723–730
Lieth E, LaNoue KF, Berkich DA, Xu B, Ratz M, Taylor C, Hutson SM (2001) Nitrogen shuttling between neurons and glial cells during glutamate synthesis. J Neurochem 76:1712–1723
Lipton SA, Rosenberg PA (1994) Excitatory amino acids as a final common pathway for neurologic disorders. N Engl J Med 330:613–622
Lowry OH, Rosebough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
McKenna MC, Sonnewald U, Huang X, Stevenson J, Johnsen SF, Sande LM, Zielke HR (1998) Alpha-ketoisocaproate alters the production of both lactate and aspartate from [U-13C]glutamate in astrocytes: a 13C NMR study. J Neurochem 70:1001–1008
Moran EP, Wang Z, Chen J, Sapieha P, Smith LE, Ma JX (2016) Neurovascular cross talk in diabetic retinopathy: pathophysiological roles and therapeutic implications. Am J Physiol Heart Circ Physiol 311:738–749
Niizuma K, Endo H, Chan PH (2009) Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J Neurochem 109:133–138
Ola MS, Alhomida AS (2014) Neurodegeneration in diabetic retina and its potential drug targets. Curr Neuropharmacol 12:380–386
Ola MS, Berkich DA, Xu Y, King MT, Gardner TW, Simpson I, LaNoue KF (2006) Analysis of glucose metabolism in diabetic rat retinas. Am J Physiol Endocrinol Metab 290:E1057–E1067
Ola MS, Hosoya K, LaNoue KF (2011) Regulation of glutamate metabolism by hydrocortisone and branched chain keto acids in cultured rat retinal Müller cells (TR-MUL). Neurochem Int 59:656–663
Ola MS, Nawaz MI, Siddiquei MM, Al-Amro S, Abu El-Asrar AM (2012) Recent advances in understanding the biochemical and molecular mechanism of diabetic retinopathy. J Diabetes Complicat 26:56–64
Ola MS, Ahmed MM, Ahmad R, Abuohashish HM, Al-Rejaie SS, Alhomida AS (2015) Neuroprotective effects of rutin in streptozotocin-induced diabetic rat retina. J Mol Neurosci 56:440–448
Ola MS, Al-Dosari D, Alhomida AS (2018) Role of oxidative stress in diabetic retinopathy and the beneficial effects of flavonoids. Curr Pharm Des 24:2180–2187
Parke-Davis MP (2009) Product information: Neurontin (gabapentin). Pfizer, New York
Rossi AR, Angelo MF, Villarreal A, Lukin J, Ramos AJ (2013) Gabapentin administration reduces reactive gliosis and neurodegeneration after pilocarpine-induced status epilepticus. PLoS One 8:e78516. https://doi.org/10.1371/journal.pone.0078516
Rothstein JD, Kuncl RW (1995) Neuroprotective strategies in a model of chronic glutamate-mediated motor neuron toxicity. J Neurochem 65:643–651
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem 25:192–205
Shimoyama M, Shimoyama N, Hori Y (2000) Gabapentin affects glutamatergic excitatory neurotransmission in the rat dorsal horn. Pain 85:405–414
Sucher NJ, Lipton SA, Dreyer EB (1997) Molecular basis of glutamate toxicity in retinal ganglion cells. Vis Res 37:3483–3493
Sweatt AJ, Garcia-Espinosa MA, Wallin R, Hutson SM (2004) Branched-chain amino acids and neurotransmitter metabolism: expression of cytosolic branched-chain aminotransferase (BCATc) in the cerebellum and hippocampus. J Comp Neurol 477:360–370
Tönjes M, Barbus S, Park YJ, Wang W, Schlotter M, Lindroth AM, Pleier SV, Bai AHC, Karra D, Piro RM, Felsberg J, Addington A, Lemke D, Weibrecht I, Hovestadt V, Rolli CG, Campos B, Turcan S, Sturm D, Witt H, Chan TA, Herold-Mende C, Kemkemer R, König R, Schmidt K, Hull WE, Pfister SM, Jugold M, Hutson SM, Plass C, Okun JG, Reifenberger G, Lichter P, Radlwimmer B (2013) BCAT1 promotes cell proliferation through amino acid catabolism in gliomas carrying wild-type IDH1. Nat Med 19:901–908
Xu Y, Oz G, LaNoue KF, Keiger CJ, Berkich DA, Gruetter R, Hutson SH (2004) Whole-brain glutamate metabolism evaluated by steady-state kinetics using a double-isotope procedure: effects of gabapentin. J Neurochem 90:1104–1111
Acknowledgements
Authors would like to thank the funding support from King Abdul Aziz City for Science and Technology (KACST), grant number ARP 30-23.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ola, M.S., Alhomida, A.S. & LaNoue, K.F. Gabapentin Attenuates Oxidative Stress and Apoptosis in the Diabetic Rat Retina. Neurotox Res 36, 81–90 (2019). https://doi.org/10.1007/s12640-019-00018-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00018-w