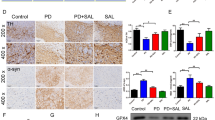
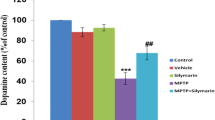
Abstract
Parkinson’s disease (PD) is one of the widely reported neurodegenerative disorders affecting more than ten million people worldwide. Due to therapeutic limitations and several adverse effects associated with currently used drugs, it is crucial to search for safe and effective options for treatment of PD. Oxidative stress, mitochondrial dysfunction, α-synuclein oligomeric aggregates, and glucocerebrosidase (GCase) deficiency are involved in PD pathogenesis. Rebamipide, an anti-ulcer drug, is a proven free-radical scavenger and antioxidant. The drug has shown neuroprotective effects in cultured SH-SY5Y cells. Therefore, we investigated the pharmacological effect of rebamipide in 6-hydroxydopamine (6-OHDA)-induced experimental PD model. Rebamipide was given to adult male albino rats of Charles-Foster strain in 20, 40, and 80 mg/kg (R-20, R-40, and R-80) oral dose twice daily for 24 days (day 4 to day 27) after 6-OHDA intrastriatal injection. The drug inhibited 6-OHDA-induced motor deficits and nigral α-synuclein aggregates in dose-dependent manner. R-40 and R-80 dose dependently increased striatal mitochondrial complex I, II, IV, and V activities; mitochondrial bioenergetics; and nigral GCase activity. 6-OHDA-induced lipid peroxidation was decreased. Highest dose (R-80) also decreased apoptotic proteins and upregulated striatal dopamine concentration in 6-OHDA-induced hemiparkinson’s rat model. Therefore, the anti-PD effect of rebamipide may involve stabilization of mitochondrial bioenergetics, enhancement of GCase enzymatic activity as well as decreased oxidative stress with α-synuclein pathology, and apoptosis in 6-OHDA-induced hemiparkinson’s rat model. Hence, preclinical evidence indicates rebamipide to be a potential drug for management of PD.








Similar content being viewed by others
Abbreviations
- 4-MU:
-
4-methylumbelliferone
- 6-OHDA:
-
6-hydroxydopamine
- α-Synuclein:
-
alpha-synuclein
- Aβ42:
-
amyloid-β 1–42
- ADP:
-
adenosine diphosphate
- ATP:
-
adenosine triphosphate
- β-actin:
-
beta-actin
- BSA:
-
bovine serum albumin
- CMC:
-
carboxymethylcellulose
- CNS:
-
central nervous system
- COMT:
-
catechol-O-methyltransferase
- DA:
-
dopamine
- DNA:
-
deoxyribonucleic acid
- DOPAC:
-
3,4-dihydroxyphenylacetic acid
- ECD:
-
electrochemical detector
- EGTA:
-
ethylene glycol-bis (β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid
- ELISA:
-
enzyme-linked immunosorbent assay
- ER:
-
endoplasmic reticulum
- ETC:
-
electron transport chain
- FAD:
-
flavin adenine dinucleotide
- FCCP:
-
carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone
- GC:
-
glucocerebroside
- GCase:
-
glucocerebrosidase
- h:
-
hours
- H+ :
-
Hydrogen ion
- H2O2 :
-
hydrogen peroxide
- HEPES:
-
4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid
- HPLC:
-
high-performance liquid chromatography
- HVA:
-
homovanillic acid
- iNOS:
-
inducible nitric oxide synthase
- kg:
-
kilogram
- KH2PO4 :
-
potassium phosphate monobasic anhydrous
- LPO:
-
lipid peroxide
- MAO B:
-
monoamine oxidase B
- MDA:
-
malondialdehyde
- mg:
-
milligram
- MgCl2 :
-
magnesium chloride
- Min:
-
minute
- mL:
-
milliliter
- mM:
-
millimolar
- mmol:
-
millimoles
- MMP:
-
mitochondrial membrane potential
- μL:
-
microliter
- μg:
-
microgram
- μmol:
-
micromoles
- NAD+ :
-
nicotinamide adenine dinucleotide (oxidized)
- NADH:
-
nicotinamide adenine dinucleotide (reduced)
- NBT:
-
nitroblue tetrazolium
- ng:
-
nanogram
- NIH:
-
National Institutes of Health Guide for the Care and Use of Laboratory Animals
- NMDA:
-
N-methyl-d-aspartate
- nmol:
-
nanomoles
- OFT:
-
open field test
- PD:
-
Parkinson’s disease
- pg:
-
picogram
- Pi:
-
inorganic phosphate
- p.o.:
-
per os
- R-20:
-
rebamipide 20 mg/kg
- R-40:
-
rebamipide 40 mg/kg
- R-80:
-
rebamipide 80 mg/kg
- RCR:
-
respiratory control ratio
- RNA:
-
ribonucleic acid
- ROS:
-
reactive oxygen species
- S.C.:
-
subcutaneous
- SD:
-
standard deviation
- SDS-PAGE:
-
sodium dodecyl sulfate polyacrylamide gel electrophoresis
- S:
-
seconds
- SOD:
-
superoxide dismutase
- SNc:
-
substantia nigra pars compacta
- TBARS:
-
thiobarbituric acid reactive substances
References
Ambrosi G, Kustrimovic N, Siani F, Rasini E, Cerri S, Ghezzi C, Dicorato G, Caputo S, Marino F, Cosentino M, Blandini F (2017) Complex changes in the innate and adaptive immunity accompany progressive degeneration of the nigrostriatal pathway induced by intrastriatal injection of 6-hydroxydopamine in the rat. Neurotox Res 32:71–81. https://doi.org/10.1007/s12640-017-9712-2
Athauda D, Maclagan K, Skene SS, Bajwa-Joseph M, Letchford D, Chowdhury K, Hibbert S, Budnik N, Zampedri L, Dickson J, Li Y, Aviles-Olmos I, Warner TT, Limousin P, Lees AJ, Greig NH, Tebbs S, Foltynie T (2017) Exenatide once weekly versus placebo in Parkinson’s disease: a randomised, double-blind, placebo-controlled trial. Lancet 390:1664–1675. https://doi.org/10.1016/S0140-6736(17)31585-4
Batool F, Haleem D (2008) Serotonin1A receptor agonism in the expression of behavioral dopaminergic supersensitivity in subchronic haloperidol treated rats. Pak J Pharm Sci 21:411–420
Bendikov-Bar I, Ron I, Filocamo M, Horowitz M (2011) Characterization of the ERAD process of the L444P mutant glucocerebrosidase variant. Blood Cells Mol Dis 46:4–10. https://doi.org/10.1016/j.bcmd.2010.10.012
Berger K, Przedborski S, Cadet JL (1991) Retrograde degeneration of nigrostriatal neurons induced by intrastriatal 6-hydroxydopamine injection in rats. Brain Res Bull 26:301–307. https://doi.org/10.1016/0361-9230(91)90242-C
Berman SB, Hastings TG (1999) Dopamine oxidation alters mitochondrial respiration and induces permeability transition in brain mitochondria. J Neurochem 73:1127–1137. https://doi.org/10.1046/j.1471-4159.1999.0731127.x
Blum D, Torch S, Lambeng N, Nissou M-F, Benabid A-L, Sadoul R, Verna J-M (2001) Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: contribution to the apoptotic theory in Parkinson’s disease. Prog Neurobiol 65:135–172. https://doi.org/10.1016/S0301-0082(01)00003-X
Bronstein PM (1972) Open-field behavior of the rat as a function of age: cross-sectional and longitudinal investigations. J Comp Physiol Psych 80:335–341. https://doi.org/10.1037/h0032986
Budi A, Heru S, Ahmad RA, Yusuf A (2012) Increase of oxidative stress and accumulation of α-synuclein in Wistar rat’s midbrain treated with rotenone. ITB J Sci 44 A:317–332. https://doi.org/10.5614/itbj.sci.2012.44.4.3
Burnette WN (1981) “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem 112:195–203. https://doi.org/10.1016/0003-2697(81)90281-5
Carrozzino D, Morberg BM, Siri C, Pezzoli G, Bech P (2018) Evaluating psychiatric symptoms in Parkinson’s disease by a clinimetric analysis of the Hopkins symptom checklist (SCL-90-R). Prog Neuro-Psychopharmacol Biol Psychiatry 81:131–137. https://doi.org/10.1016/j.pnpbp.2017.10.024
Carvey P et al (2005) 6-Hydroxydopamine-induced alterations in blood–brain barrier permeability. Eur J Neurosci 22:1158–1168. https://doi.org/10.1111/j.1460-9568.2005.04281.x
Cheng HC, Ulane CM, Burke RE (2010) Clinical progression in Parkinson disease and the neurobiology of axons. Ann Neurol 67:715–725. https://doi.org/10.1002/ana.21995
Choi HS, Lim JY, Chun HJ, Lee M, Kim ES, Keum B, Seo YS, Jeen YT, Um SH, Lee HS, Kim CD, Ryu HS, Sul D (2013) The effect of polaprezinc on gastric mucosal protection in rats with ethanol-induced gastric mucosal damage: comparison study with rebamipide. Life Sci 93:69–77. https://doi.org/10.1016/j.lfs.2013.05.019
Cleeter MW, Chau KY, Gluck C, Mehta A, Hughes DA, Duchen M, Wood NW, Hardy J, Mark Cooper J, Schapira AH (2013) Glucocerebrosidase inhibition causes mitochondrial dysfunction and free radical damage. Neurochem Int 62:1–7. https://doi.org/10.1016/j.neuint.2012.10.010
CoronelOliveros CM, PachecoCalderón R (2018) Prenatal exposure to ketamine in rats: implications on animal models of schizophrenia. Dev Psychobiol 60:30–42. https://doi.org/10.1002/dev.21586
Coulombe K, Saint-Pierre M, Cisbani G, St-Amour I, Gibrat C, Giguère-Rancourt A, Calon F, Cicchetti F (2016) Partial neurorescue effects of DHA following a 6-OHDA lesion of the mouse dopaminergic system. J Nutr Biochem 30:133–142
Creese B, Bell E, Johar I, Francis P, Ballard C, Aarsland D (2017) Glucocerebrosidase mutations and neuropsychiatric phenotypes in Parkinson’s disease and Lewy body dementias: review and meta-analyses. Am J Med Genet B Neuropsychiatr Genet 177:232–241. https://doi.org/10.1002/ajmg.b.32549
Czerniczyniec A, Bustamante J, Lores-Arnaiz S (2010) Dopamine modifies oxygen consumption and mitochondrial membrane potential in striatal mitochondria. Mol Cell Biochem 341:251–257. https://doi.org/10.1007/s11010-010-0456-z
Dabbeni-Sala F, Di Santo S, Franceschini D, Skaper SD, Giu P (2001) Melatonin protects against 6-OHDA-induced neurotoxicity in rats: a role for mitochondrial complex I activity. FASEB J 15:164–170. https://doi.org/10.1096/fj.00-0129com
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39:889–909. https://doi.org/10.1016/S0896-6273(03)00568-3
Denenberg VH (1969) Open-field behavior in the rat: what DOES it mean? Ann N Y Acad Sci 159:852–859. https://doi.org/10.1111/j.1749-6632.1969.tb12983.x
Di Maio R et al (2016) α-Synuclein binds to TOM20 and inhibits mitochondrial protein import in Parkinson’s disease. Sci Transl Med 8:342ra378. https://doi.org/10.1126/scitranslmed.aaf3634
Diao L et al (2012) Rebamipide suppresses diclofenac-induced intestinal permeability via mitochondrial protection in mice. World J Gastroenterol 18:1059–1066. https://doi.org/10.3748/wjg.v18.i10.1059
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35:495–516. https://doi.org/10.1080/01926230701320337
Fernandez A, De La Vega AG, Torres-Aleman I (1998) Insulin-like growth factor I restores motor coordination in a rat model of cerebellar ataxia. Proc Natl Acad Sci U S A 95:1253–1258. https://doi.org/10.1073/pnas.95.3.1253
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400
Fukui K, Yachi K, Yoshida H, Tanji K, Matsumiya T, Hayakari R, Tsuruga K, Tanaka H, Imaizumi T (2017) Rebamipide reduces amyloid-β 1–42 (Aβ42) production and ameliorates Aβ43-lowered cell viability in cultured SH-SY5Y human neuroblastoma cells. Neurosci Res 124:40–50. https://doi.org/10.1016/j.neures.2017.05.005
Garcia-Reitböck P, Anichtchik O, Bellucci A, Iovino M, Ballini C, Fineberg E, Ghetti B, Della Corte L, Spano PF, Tofaris GK, Goedert M, Spillantini MG (2010) SNARE protein redistribution and synaptic failure in a transgenic mouse model of Parkinson’s disease. Brain 133:2032–2044. https://doi.org/10.1093/brain/awq132
Geed M, Garabadu D, Ahmad A, Krishnamurthy S (2014) Silibinin pretreatment attenuates biochemical and behavioral changes induced by intrastriatal MPP+ injection in rats. Pharmacol Biochem Behav 117:92–103. https://doi.org/10.1016/j.pbb.2013.12.008
Gegg ME, Burke D, Heales SJ, Cooper JM, Hardy J, Wood NW, Schapira AH (2012) Glucocerebrosidase deficiency in substantia nigra of Parkinson disease brains. Ann Neurol 72:455–463. https://doi.org/10.1002/ana.23614
Gendy AM, Abdallah DM, El-Abhar HS (2017) The potential curative effect of rebamipide in hepatic ischemia/reperfusion injury. Naunyn Schmiedeberg's Arch Pharmacol 390:691–700. https://doi.org/10.1007/s00210-017-1370-7
Gilmer LK, Roberts KN, Joy K, Sullivan PG, Scheff SW (2009) Early mitochondrial dysfunction after cortical contusion injury. J Neurotrauma 26:1271–1280. https://doi.org/10.1089/neu.2008.0857
Griffiths DE, Houghton RL (1974) Studies on energy-linked reactions: modified mitochondrial ATPase of oligomycin-resistant mutants of Saccharomyces cerevisiae. Eur J Biochem 46:157–167. https://doi.org/10.1111/j.1432-1033.1974.tb03608.x
Guo C, Sun L, Chen X, Zhang D (2013) Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res 8:2003. https://doi.org/10.3969/j.issn.1673-5374.2013.21.009
Hu LF, Lu M, Tiong CX, Dawe GS, Hu G, Bian JS (2010) Neuroprotective effects of hydrogen sulfide on Parkinson’s disease rat models. Aging Cell 9:135–146. https://doi.org/10.1111/j.1474-9726.2009.00543.x
Ishihara T, Tanaka K-I, Tashiro S, Yoshida K, Mizushima T (2010) Protective effect of rebamipide against celecoxib-induced gastric mucosal cell apoptosis. Biochem Pharmacol 79:1622–1633. https://doi.org/10.1016/j.bcp.2010.01.030
Jun D-J, Kim J, Jung SY, Song R, Noh JH, Park YS, Ryu SH, Kim JH, Kong YY, Chung JM, Kim KT (2007) Extracellular ATP mediates necrotic cell swelling in SN4741 dopaminergic neurons through P2X7 receptors. J Biol Chem 282:37350–37358. https://doi.org/10.1074/jbc.M707915200
Kim C, Speisky M, Kharouba S (1987) Rapid and sensitive method for measuring norepinephrine, dopamine, 5-hydroxytryptamine and their major metabolites in rat brain by high-performance liquid chromatography: differential effect of probenecid, haloperidol and yohimbine on the concentrations of biogenic amines and metabolites in various regions of rat brain. J Chromatogr A 386:25–35. https://doi.org/10.1016/S0021-9673(01)94581-9
Kim CD, Hong KW (1995) Preventive effect of rebamipide on gastric lesions induced by ischemia-reperfusion in the rat. J Pharmacol Exp Ther 1(275):340–344
Kirik D, Rosenblad C, Björklund A (1998) Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat. Exp Neurol 152:259–277
Kowal SL, Dall TM, Chakrabarti R, Storm MV, Jain A (2013) The current and projected economic burden of Parkinson’s disease in the United States. Mov Disord 28:311–318. https://doi.org/10.1002/mds.25292
Krügel U, Kittner H, Franke H, Illes P (2003) Purinergic modulation of neuronal activity in the mesolimbic dopaminergic system in vivo. Synapse 47:134–142. https://doi.org/10.1002/syn.10162
Kumar A, Sharma N, Gupta A, Kalonia H, Mishra J (2012) Neuroprotective potential of atorvastatin and simvastatin (HMG-CoA reductase inhibitors) against 6-hydroxydopamine (6-OHDA) induced Parkinson-like symptoms. Brain Res 1471:13–22. https://doi.org/10.1016/j.brainres.2012.06.050
Kumar S, Mishra A, Krishnamurthy S (2017) Purinergic antagonism prevents mitochondrial dysfunction and behavioral deficits associated with dopaminergic toxicity induced by 6-OHDA in rats. Neurochem Res 42:1–17. https://doi.org/10.1007/s11064-017-2383-9
Kupsch A, Schmidt W, Gizatullina Z, Debska-Vielhaber G, Voges J, Striggow F, Panther P, Schwegler H, Heinze HJ, Vielhaber S, Gellerich FN (2014) 6-Hydroxydopamine impairs mitochondrial function in the rat model of Parkinson’s disease: respirometric, histological, and behavioral analyses. J Neural Transm (Vienna) 121:1245–1257. https://doi.org/10.1007/s00702-014-1185-3
Kwok JYY, Kwan JCY, Auyeung M, Mok VCT, Chan HYL (2017) The effects of yoga versus stretching and resistance training exercises on psychological distress for people with mild-to-moderate Parkinson’s disease: study protocol for a randomized controlled trial. Trials 18:509. https://doi.org/10.1186/s13063-017-2223-x
Lesage S, Brice A (2009) Parkinson’s disease: from monogenic forms to genetic susceptibility factors. Hum Mol Genet 18:R48–R59. https://doi.org/10.1093/hmg/ddp012
Lever C, Burton S, O’Keefe J (2006) Rearing on hind legs, environmental novelty and the hippocampal formation. Rev Neurosci 17:111–134. https://doi.org/10.1515/REVNEURO.2006.17.1-2.111
Lhommée E, Wojtecki L, Czernecki V, Witt K, Maier F, Tonder L, Timmermann L, Hälbig TD, Pineau F, Durif F, Witjas T, Pinsker M, Mehdorn M, Sixel-Döring F, Kupsch A, Krüger R, Elben S, Chabardès S, Thobois S, Brefel-Courbon C, Ory-Magne F, Regis JM, Maltête D, Sauvaget A, Rau J, Schnitzler A, Schüpbach M, Schade-Brittinger C, Deuschl G, Houeto JL, Krack P, Negovanska V, Welter ML, Corvol JC, Agid Y, Navarro S, Meier N, Hartmann A, Hesekamp H, Cornu P, Möller B, Nebel A, Raethjen J, Knudsen K, Volkmann J, Falk D, Paschen S, Meister I, Kuhn J, Donner K, Kessler J, Barbe M, Fink G, Maarouf M, Kühn A, Müller B, Faust K, Gruber D, Schneider GH, Seigneuret E, Pollak P, Fraix V, Kistner A, Rascol O, Arbus C, Danet L, Chaynes P, Groiss SJ, Hartmann C, Südmeyer M, Partowinia-Peters M, Vesper J, Ledily S, Damier P, Raoul S, Trenkwalder C, Richter-Dreske W, Wächter T, Weiss D, Eusebio A, Azulay JP, Polo G, Pinto S, Levin J, Dornier S, Pene F, Hourton D, Quintin M, Hoffart-Jourdain C, Brocvielle H, Balthasar K, Stein M, Harnisch S, Reuss A, Aminossadati B, Nasemann C, Oertel W, Bataille B, Hellwig D, Gharabaghi A, Amtage F, Mertens P, Kloss M, Post B, Speelman H (2018) Behavioural outcomes of subthalamic stimulation and medical therapy versus medical therapy alone for Parkinson’s disease with early motor complications (EARLYSTIM trial): secondary analysis of an open-label randomised trial. The Lancet Neurology 17:223–231. https://doi.org/10.1016/S1474-4422(18)30035-8
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mazzulli JR, Xu YH, Sun Y, Knight AL, McLean PJ, Caldwell GA, Sidransky E, Grabowski GA, Krainc D (2011) Gaucher disease glucocerebrosidase and α-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 146:37–52. https://doi.org/10.1016/j.cell.2011.06.001
Meyer OA, Tilson H, Byrd W, Riley M (1979) A method for the routine assessment of fore-and hindlimb grip strength of rats and mice. Neurobehav Toxicol 1:233–236
Mishra A, Chandravanshi LP, Trigun SK, Krishnamurthy S (2018) Ambroxol modulates 6-hydroxydopamine-induced temporal reduction in glucocerebrosidase (GCase) enzymatic activity and Parkinson’s disease symptoms. Biochem Pharmacol 155:479–493. https://doi.org/10.1016/j.bcp.2018.07.028
Moon S-J, Woo YJ, Jeong JH, Park MK, Oh HJ, Park JS, Kim EK, Cho ML, Park SH, Kim HY, Min JK (2012) Rebamipide attenuates pain severity and cartilage degeneration in a rat model of osteoarthritis by downregulating oxidative damage and catabolic activity in chondrocytes. Osteoarthr Cartil 20:1426–1438. https://doi.org/10.1016/j.joca.2012.08.002
Moore DJ, West AB, Dawson VL, Dawson TM (2005) Molecular pathophysiology of Parkinson’s disease. Annu Rev Neurosci 28:57–87. https://doi.org/10.1146/annurev.neuro.28.061604.135718
Müller T (2012) Drug therapy in patients with Parkinson’s disease. Transl Neurodegener 1:10. https://doi.org/10.1186/2047-9158-1-10
Murray MD, Callahan CM (2003) Improving medication use for older adults: an integrated research agenda. Ann Intern Med 139:425–429. https://doi.org/10.7326/0003-4819-139-5_Part_2-200309021-00009
Nagano Y, Matsui H, Muramatsu M, Shimokawa O, Shibahara T, Yanaka A, Nakahara A, Matsuzaki Y, Tanaka N, Nakamura Y (2005) Rebamipide significantly inhibits indomethacin-induced mitochondrial damage, lipid peroxidation, and apoptosis in gastric epithelial RGM-1 cells. Dig Dis Sci 50:S76–S83. https://doi.org/10.1007/s10620-005-2810-7
Nakabeppu Y, Tsuchimoto D, Yamaguchi H, Sakumi K (2007) Oxidative damage in nucleic acids and Parkinson’s disease. J Neurosci Res 85:919–934. https://doi.org/10.1002/jnr.21191
Nicholls DG, Ward MW (2000) Mitochondrial membrane potential and neuronal glutamate excitotoxicity: mortality and millivolts. Trends Neurosci 23:166–174. https://doi.org/10.1016/S0166-2236(99)01534-9
Ohashi Y, Aihara E, Takasuka H, Takahashi K, Takeuchi K (2009) Antral ulcers induced by alendronate, a nitrogen-containing bisphosphonate, in rat stomachs—prophylactic effect of rebamipide. J Physiol Pharmacol 60:85–93
Old SL, Johnson MA (1989) Methods of microphotometric assay of succinate dehydrogenase and cytochromec oxidase activities for use on human skeletal muscle. Histochem J 21:545–555. https://doi.org/10.1007/BF01753355
Osellame LD, Rahim AA, Hargreaves IP, Gegg ME, Richard-Londt A, Brandner S, Waddington SN, Schapira AHV, Duchen MR (2013) Mitochondria and quality control defects in a mouse model of Gaucher disease—links to Parkinson’s disease. Cell Metab 17:941–953. https://doi.org/10.1016/j.cmet.2013.04.014
Parker NF, Cameron CM, Taliaferro JP, Lee J, Choi JY, Davidson TJ, Daw ND, Witten IB (2016) Reward and choice encoding in terminals of midbrain dopamine neurons depends on striatal target. Nat Neurosci 19:845–854. https://doi.org/10.1038/nn.4287
Patki G, Lau Y-S (2011) Melatonin protects against neurobehavioral and mitochondrial deficits in a chronic mouse model of Parkinson’s disease. Pharmacol Biochem Behav 99:704–711. https://doi.org/10.1016/j.pbb.2011.06.026
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Pradhan SD, Brewer BR, Carvell GE, Sparto PJ, Delitto A, Matsuoka Y (2010) Assessment of fine motor control in individuals with Parkinson’s disease using force tracking with a secondary cognitive task. J Neurol Phys Ther 34:32–40. https://doi.org/10.1097/NPT.0b013e3181d055a6
Prajapati SK, Garabadu D, Krishnamurthy S (2017) Coenzyme Q10 prevents mitochondrial dysfunction and facilitates pharmacological activity of atorvastatin in 6-OHDA induced dopaminergic toxicity in rats. Neurotox Res 31:478–492. https://doi.org/10.1007/s12640-016-9693-6
Qian Y, Lei G, Castellanos FX, Forssberg H, Heijtz RD (2010) Deficits in fine motor skills in a genetic animal model of ADHD. Behav Brain Funct 6:51–51. https://doi.org/10.1186/1744-9081-6-51
Reczek D, Schwake M, Schröder J, Hughes H, Blanz J, Jin X, Brondyk W, van Patten S, Edmunds T, Saftig P (2007) LIMP-2 is a receptor for lysosomal mannose-6-phosphate-independent targeting of β-glucocerebrosidase. Cell 131:770–783. https://doi.org/10.1016/j.cell.2007.10.018
Reglodi D, Renaud J, Tamas A, Tizabi Y, Socías SB, Del-Bel E, Raisman-Vozari R (2017) Novel tactics for neuroprotection in Parkinson’s disease: role of antibiotics, polyphenols and neuropeptides. Prog Neurobiol 155:120–148. https://doi.org/10.1016/j.pneurobio.2015.10.004
Rocha EM, Smith GA, Park E, Cao H, Brown E, Hallett P, Isacson O (2015) Progressive decline of glucocerebrosidase in aging and Parkinson’s disease. Ann Clin Transl Neurol 2:433–438. https://doi.org/10.1002/acn3.177
Rozas G, Guerra M, Labandeira-Garcıa J (1997) An automated rotarod method for quantitative drug-free evaluation of overall motor deficits in rat models of parkinsonism. Brain Res Brain Res Protoc 2:75–84. https://doi.org/10.1016/S1385-299X(97)00034-2
Sakurai K, Osaka T, Yamasaki K (1998) Protection by rebamipide against acetic acid-induced colitis in rats: relationship with its antioxidative activity. Dig Dis Sci 43:125S–133S
Samaiya PK, Krishnamurthy S (2015) Characterization of mitochondrial bioenergetics in neonatal anoxic model of rats. J Bioenerg Biomembr 47:217–222. https://doi.org/10.1007/s10863-015-9603-2
Samaiya PK, Narayan G, Kumar A, Krishnamurthy S (2016) Neonatal anoxia leads to time dependent progression of mitochondrial linked apoptosis in rat cortex and associated long term sensorimotor deficits. Int J Dev Neurosci 52:55–65. https://doi.org/10.1016/j.ijdevneu.2016.05.005
Samaiya PK, Narayan G, Kumar A, Krishnamurthy S (2018) 2,4 Dinitrophenol attenuates mitochondrial dysfunction and improves neurobehavioral outcomes postanoxia in neonatal rats. Neurotox Res 34:121–136. https://doi.org/10.1007/s12640-018-9873-7
Sanberg PR, Bunsey MD, Giordano M, Norman AB (1988) The catalepsy test: its ups and downs. Behav Neurosci 102:748–759. https://doi.org/10.1037/0735-7044.102.5.748
Schapira A, Cooper J, Dexter D, Clark J, Jenner P, Marsden C (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54:823–827. https://doi.org/10.1111/j.1471-4159.1990.tb02325.x
Shapiro BL, Feigal RJ, Lam L (1979) Mitrochondrial NADH dehydrogenase in cystic fibrosis. Proc Natl Acad Sci U S A 76:2979–2983. https://doi.org/10.1073/pnas.76.6.2979
Shin BS, Kim CH, Jun YS, Yoon CH, Rho JI, Lee KC, Han HS, Yoo SD (2004) Oral absorption and pharmacokinetics of rebamipide and rebamipide lysinate in rats. Drug Dev Ind Pharm 30:869–876. https://doi.org/10.1081/DDC-200034577
Shioya Y, Kashiyama E, Okada K, Kusumoto N, Abe Y, Uchida M, Shimizu T (1989) Metabolic fate of the anti-ulcer agent,(±)-2-(4-chlorobenzoylamino)-3-[2 (1H)-quinolinon-4-yl] propionic acid (OPC-12759): absorption, distribution, and excretion in rats and dogs Iyakuhin. Kenkyu 20:522–533
Shioya Y, Shimizu T (1988) High-performance liquid chromatographic procedure for the determination of a new anti-gastric ulcer agent, 2-(4-chlorobenzoylamino)-3-[2 (1H)-quinlinon-4-yl] propionic acid, in human plasma and urine. J Chromatogr B Biomed Sci Appl 434:283–287. https://doi.org/10.1016/0378-4347(88)80089-6
Singh IN, Sullivan PG, Deng Y, Mbye LH, Hall ED (2006) Time course of post-traumatic mitochondrial oxidative damage and dysfunction in a mouse model of focal traumatic brain injury: implications for neuroprotective therapy. J Cereb Blood Flow Metab 26:1407–1418. https://doi.org/10.1038/sj.jcbfm.9600297
Smith Y, Wichmann T, Factor SA, DeLong MR (2012) Parkinson’s disease therapeutics: new developments and challenges since the introduction of levodopa. Neuropsychopharmacology 37:213–246. https://doi.org/10.1038/npp.2011.212
Smolinsky AN, Bergner CL, LaPorte JL, Kalueff AV (2009) Analysis of grooming behavior and its utility in studying animal stress, anxiety, and depression. In: Mood and anxiety related phenotypes in mice, vol 42. Springer, pp 21–36. https://doi.org/10.1007/978-1-60761-303-9_2
Sunderman F, Marzouk A, Hopfer S, Zaharia O, Reid M (1985) Increased lipid peroxidation in tissues of nickel chloride-treated rats. Ann Clin Lab Sci 15:229–236
Takeshita H, Yamamoto K, Nozato S, Inagaki T, Tsuchimochi H, Shirai M, Yamamoto R, Imaizumi Y, Hongyo K, Yokoyama S, Takeda M, Oguro R, Takami Y, Itoh N, Takeya Y, Sugimoto K, Fukada SI, Rakugi H (2017) Modified forelimb grip strength test detects aging-associated physiological decline in skeletal muscle function in male mice. Sci Rep 7:42323. https://doi.org/10.1038/srep42323
Uchiyama M, Mihara M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278. https://doi.org/10.1016/0003-2697(78)90342-1
Ungerstedt U (1971) Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiol Scand Suppl 82:69–93. https://doi.org/10.1111/j.1365-201X.1971.tb11000.x
Van Den Buuse M, Veldhuis HD, De Boer S, Versteeg DH, De Jong W (1986) Central 6-OHDA affects both open-field exploratory behaviour and the development of hypertension in SHR. Pharmacol Biochem Behav 24:15–21. https://doi.org/10.1016/0091-3057(86)90037-7
Walther S, Strik W (2012) Motor symptoms and schizophrenia. Neuropsychobiology 66:77–92. https://doi.org/10.1159/000339456.
Whishaw IQ, Tomie J-A, Ladowsky RL (1990) Red nucleus lesions do not affect limb preference or use, but exacerbate the effects of motor cortex lesions on grasping in the rat. Behav Brain Res 40:131–144. https://doi.org/10.1016/0166-4328(90)90005-Y
Yap TL, Gruschus JM, Velayati A, Westbroek W, Goldin E, Moaven N, Sidransky E, Lee JC (2011) α-Synuclein interacts with glucocerebrosidase providing a molecular link between Parkinson and Gaucher diseases. J Biol Chem 286:28080–28088. https://doi.org/10.1074/jbc.M111.237859
Zamzami N, Hirsch T, Dallaporta B, Petit PX, Kroemer G (1997) Mitochondrial implication in accidental and programmed cell death: apoptosis and necrosis. J Bioenerg Biomembr 29:185–193. https://doi.org/10.1023/a:1022694131572
Zhao X, Zhai S, An MS, Wang YH, Yang YF, Ge HQ, Liu JH, Pu XP (2013) Neuroprotective effects of protocatechuic aldehyde against neurotoxin-induced cellular and animal models of Parkinson’s disease. PLoS One 8:e78220. https://doi.org/10.1371/journal.pone.0078220
Acknowledgements
The authors wish to acknowledge Akums Drugs & Pharmaceuticals Ltd., New Delhi, India for providing rebamipide (active pharmaceutical ingredient) as gift sample. This work was supported by the teaching assistantship to Akanksha Mishra from Indian Institute of Technology (Banaras Hindu University), Varanasi-221005, U.P., India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All the procedures performed in the study were in accordance with the ethical standards of the Institutional animal ethical committee, Banaras Hindu University (Dean/2016/CAEC/33). The experiments were performed according to the principles of National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978) guidelines. The article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mishra, A., Krishnamurthy, S. Rebamipide Mitigates Impairments in Mitochondrial Function and Bioenergetics with α-Synuclein Pathology in 6-OHDA-Induced Hemiparkinson’s Model in Rats. Neurotox Res 35, 542–562 (2019). https://doi.org/10.1007/s12640-018-9983-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-018-9983-2